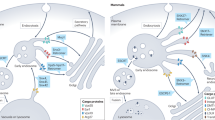
Abstract
Recent progress has been made in our understanding of the molecular mechanisms that regulate the biogenesis of multivesicular transport intermediates in the degradation pathway that leads to lysosomes. Here, we discuss recent work that uncovers some of the mechanisms that cause both membrane invagination within these newly forming intermediates and the detachment of these intermediates from early endosomal membranes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Trump, B. F., Goldblatt, P. J. & Stowell, R. E. Studies of necrosis in vitro of mouse hepatic parenchymal cells. Ultrastructural and cytochemical alterations of cytosomes, cytosegresomes, multivesicular bodies, and microbodies and their relation to the lysosome concept. Lab. Invest. 14, 1946–1968 (1965).
Helenius, A., Mellman, I., Wall, D. & Hubbard, A. Endosomes. Trends Biochem. Sci. 8, 245–250 (1983).
Geuze, H. J., Slot, J. W., Strous, G. J., Lodish, H. F. & Schwartz, A. L. Intracellular site of asialoglycoprotein receptor–ligand uncoupling: double-label immunoelectron microscopy during receptor-mediated endocytosis. Cell 32, 277–287 (1983).
Miller, K., Beardmore, J., Kanety, H., Schlessinger, J. & Hopkins, C. R. Localization of the epidermal growth factor (EGF) receptor within the endosome of EGF-stimulated epidermoid carcinoma (A431) cells. J. Cell Biol. 102, 500–509 (1986).
Mueller, S. C. & Hubbard, A. L. Receptor-mediated endocytosis of asialoglycoproteins by rat hepatocytes: receptor-positive and receptor-negative endosomes. J. Cell Biol. 102, 932–942 (1986).
Gruenberg, J., Griffiths, G. & Howell, K. E. Characterisation of the early endosome and putative endocytic carrier vesicles in vivo and with an assay of vesicle fusion in vitro. J. Cell Biol. 108, 1301–1316 (1989).
Griffiths, G., Hoflack, B., Simons, K., Mellman, I. & Kornfeld, S. The mannose-6-phosphate receptor and the biogenesis of lysosomes. Cell 52, 329–341 (1988).
Gruenberg, J. The endocytic pathway: a mosaic of domains. Nature Rev. Mol. Cell Biol. 2, 721–730 (2001).
Dunn, K. W., McGraw, T. E. & Maxfield, F. R. Iterative fractionation of recycling receptors from lysosomally destined ligands in an early sorting endosome. J. Cell Biol. 109, 3303–3314 (1989).
Yamashiro, D. J., Tycko, B., Fluss, S. R. & Maxfield, F. R. Segregation of transferrin to a mildly acidic (pH 6.4) para-Golgi compartment in the recycling pathway. Cell 37, 789–800 (1984).
Felder, S. et al. Kinase activity controls the sorting of the epidermal growth factor receptor within the multivesicular body. Cell 61, 623–634 (1990).
Katzmann, D. J., Odorizzi, G. & Emr, S. D. Receptor downregulation and multivesicular-body sorting. Nature Rev. Mol. Cell Biol. 3, 893–905 (2002).
Gruenberg, J. & Maxfield, F. Membrane transport in the endocytic pathway. Curr. Opin. Cell Biol. 7, 552–563 (1995).
Mukherjee, S. & Maxfield, F. R. Role of membrane organization and membrane domains in endocytic lipid trafficking. Traffic 1, 203–211 (2000).
Parton, R. G., Simons, K. & Dotti, C. G. Axonal and dentritic endocytic pathways in cultured neurons. J. Cell Biol. 119, 123–137 (1992).
Aniento, F., Emans, N., Griffiths, G. & Gruenberg, J. Cytoplasmic dynein-dependent vesicular transport from early to late endosomes. J. Cell Biol. 123, 1373–1388 (1993).
Bomsel, M., Parton, R., Kuznetsov, S. A., Schroer, T. A. & Gruenberg, J. Microtubule and motor dependent fusion in vitro between apical and basolateral endocytic vesicles from MDCK cells. Cell 62, 719–731 (1990).
Kobayashi, T. et al. A lipid associated with the antiphospholipid syndrome regulates endosome structure/function. Nature 392, 193–197 (1998).
Matsuo, H. et al. Role of LBPA and Alix in multivesicular liposome formation and endosome organization. Science 303, 531–534 (2004).
Goda, Y. & Pfeffer, S. R. Selective recycling of the mannose 6-phosphate/IGF-II receptor to the TGN in vitro. Cell 55, 309–320 (1988).
Murk, J. L., Stoorvogel, W., Kleijmeer, M. J. & Geuze, H. J. The plasticity of multivesicular bodies and the regulation of antigen presentation. Semin. Cell Dev. Biol. 13, 303–311 (2002).
Prescianotto-Baschong, C. & Riezman, H. Ordering of compartments in the yeast endocytic pathway. Traffic 3, 37–49 (2002).
Escola, J. M. et al. Selective enrichment of tetraspan proteins on the internal vesicles of multivesicular endosomes and on exosomes secreted by human B-lymphocytes. J. Biol. Chem. 273, 20121–20127 (1998).
Umeda, A. et al. Distribution and trafficking of MPR300 is normal in cells with cholesterol accumulated in late endocytic compartments: evidence for early endosome-to-TGN trafficking of MPR300 in NPC fibroblasts. J. Lipid Res. 44, 1821–1832 (2003).
Seaman, M. N., Marcusson, E. G., Cereghino, J. L. & Emr, S. D. Endosome to Golgi retrieval of the vacuolar protein sorting receptor, Vps10p, requires the function of the VPS29, VPS30, and VPS35 gene products. J. Cell Biol. 137, 79–92 (1997).
Mukherjee, S., Soe, T. T. & Maxfield, F. R. Endocytic sorting of lipid analogues differing solely in the chemistry of their hydrophobic tails. J. Cell Biol. 144, 1271–1284 (1999).
Fivaz, M. et al. Differential sorting and fate of endocytosed GPI-anchored proteins. EMBO J. 21, 3989–4000 (2002).
Gillooly, D. J. et al. Localization of phosphatidylinositol 3-phosphate in yeast and mammalian cells. EMBO J. 19, 4577–4588 (2000).
Petiot, A., Fauré, J., Stenmark, H. & Gruenberg, J. PI3P signaling regulates receptor sorting but not transport in the endosomal pathway. J. Cell Biol. 162, 971–979 (2003).
Fernandez-Borja, M. et al. Multivesicular body morphogenesis requires phosphatidyl-inositol 3-kinase activity. Curr. Biol. 9, 55–58 (1999).
Futter, C. E., Collinson, L. M., Backer, J. M. & Hopkins, C. R. Human VPS34 is required for internal vesicle formation within multivesicular endosomes. J. Cell Biol. 155, 1251–1264 (2001).
Komada, M. & Soriano, P. Hrs, a FYVE finger protein localized to early endosomes, is implicated in vesicular traffic and required for ventral folding morphogenesis. Genes Dev. 13, 1475–1485 (1999).
Urbe, S., Mills, I. G., Stenmark, H., Kitamura, N. & Clague, M. J. Endosomal localization and receptor dynamics determine tyrosine phosphorylation of hepatocyte growth factor-regulated tyrosine kinase substrate. Mol. Cell Biol. 20, 7685–7692 (2000).
Raiborg, C. et al. FYVE and coiled-coil domains determine the specific localisation of Hrs to early endosomes. J. Cell Sci. 114, 2255–2263 (2001).
Raiborg, C., Bache, K. G., Mehlum, A., Stang, E. & Stenmark, H. Hrs recruits clathrin to early endosomes. EMBO J. 20, 5008–5021 (2001).
Raiborg, C. et al. Hrs sorts ubiquitinated proteins into clathrin-coated microdomains of early endosomes. Nature Cell Biol. 4, 394–398 (2002).
Sachse, M., Urbe, S., Oorschot, V., Strous, G. J. & Klumperman, J. Bilayered clathrin coats on endosomal vacuoles are involved in protein sorting toward lysosomes. Mol. Biol. Cell 13, 1313–1328 (2002).
Hicke, L. Protein regulation by monoubiquitin. Nature Rev. Mol. Cell Biol. 2, 195–201 (2001).
Bilodeau, P. S., Urbanowski, J. L., Winistorfer, S. C. & Piper, R. C. The Vps27p Hse1p complex binds ubiquitin and mediates endosomal protein sorting. Nature Cell Biol. 4, 534–539 (2002).
Lloyd, T. E. et al. Hrs regulates endosome membrane invagination and tyrosine kinase receptor signaling in Drosophila. Cell 108, 261–269 (2002).
Bache, K. G., Brech, A., Mehlum, A. & Stenmark, H. Hrs regulates multivesicular body formation via ESCRT recruitment to endosomes. J. Cell Biol. 162, 435–442 (2003).
Hammond, D. E. et al. Endosomal dynamics of Met determine signaling output. Mol. Biol. Cell 14, 1346–1354 (2003).
Babst, M., Odorizzi, G., Estepa, E. J. & Emr, S. D. Mammalian tumor susceptibility gene 101 (TSG101) and the yeast homologue, Vps23p, both function in late endosomal trafficking. Traffic 1, 248–258 (2000).
Bishop, N., Horman, A. & Woodman, P. Mammalian class E vps proteins recognize ubiquitin and act in the removal of endosomal protein–ubiquitin conjugates. J. Cell Biol. 157, 91–101 (2002).
Pornillos, O. et al. HIV Gag mimics the Tsg101-recruiting activity of the human HRS protein. J. Cell Biol. 162, 425–434 (2003).
Katzmann, D. J., Stefan, C. J., Babst, M. & Emr, S. D. Vps27 recruits ESCRT machinery to endosomes during MVB sorting. J. Cell Biol. 162, 413–423 (2003).
Lu, Q., Hope, L. W., Brasch, M., Reinhard, C. & Cohen, S. N. TSG101 interaction with HRS mediates endosomal trafficking and receptor down-regulation. Proc. Natl Acad. Sci. USA 100, 7626–7631 (2003).
Martin-Serrano, J., Zang, T. & Bieniasz, P. D. HIV-1 and Ebola virus encode small peptide motifs that recruit Tsg101 to sites of particle assembly to facilitate egress. Nature Med. 7, 1313–1319 (2001).
Martin-Serrano, J., Zang, T. & Bieniasz, P. D. Role of ESCRT-I in retroviral budding. J. Virol. 77, 4794–4804 (2003).
Garrus, J. E. et al. Tsg101 and the vacuolar protein sorting pathway are essential for HIV-1 budding. Cell 107, 55–65 (2001).
Pelchen-Matthews, A., Kramer, B. & Marsh, M. Infectious HIV-1 assembles in late endosomes in primary macrophages. J. Cell Biol. 162, 443–455 (2003).
Sbrissa, D., Ikonomov, O. C. & Shisheva, A. Phosphatidylinositol 3-phosphate-interacting domains in PIKfyve. Binding specificity and role in PIKfyve. Endomembrane localization. J. Biol. Chem. 277, 6073–6079 (2002).
Odorizzi, G., Babst, M. & Emr, S. D. Fab1p PtdIns(3)P 5-kinase function essential for protein sorting in the multivesicular body. Cell 95, 847–858 (1998).
Ikonomov, O. C. et al. Functional dissection of lipid and protein kinase signals of PIKfyve reveals the role of PtdIns 3,5-P2 production for endomembrane integrity. J. Biol. Chem. 277, 9206–9211 (2002).
Reggiori, F. & Pelham, H. R. A transmembrane ubiquitin ligase required to sort membrane proteins into multivesicular bodies. Nature Cell Biol. 4, 117–123 (2002).
Whitley, P. et al. Identification of mammalian Vps24p as an effector of phosphatidylinositol 3,5 bisphosphate dependent endosome compartmentalization. J. Biol. Chem. 278, 38786–38795 (2003).
Mayran, M., Parton, R. G. & Gruenberg, J. Annexin II regulates multivesicular endosome biogenesis in the degradation pathway of animal cells. EMBO J. 13, 3242–3253 (2003).
Zobiack, N., Rescher, U., Ludwig, C., Zeuschner, D. & Gerke, V. The annexin 2/S100A10 complex controls the distribution of transferrin receptor-containing recycling endosomes. Mol. Biol. Cell 14, 4896–4908 (2003).
Emans, N. et al. Annexin II is a major component of fusogenic endosomal vesicles. J. Cell Biol. 120, 1357–1370 (1993).
Harder, T. & Gerke, V. The subcellular distribution of early endosomes is affected by the annexin II2p11(2) complex. J. Cell Biol. 123, 1119–1132 (1993).
Gerke, V. & Moss, S. E. Annexins: from structure to function. Physiol. Rev. 82, 331–371 (2002).
Babiychuk, E. B. & Draeger, A. Annexins in cell membrane dynamics. Ca(2+)-regulated association of lipid microdomains. J. Cell Biol. 150, 1113–1124 (2000).
Harder, T., Kellner, R., Parton, R. G. & Gruenberg, J. Specific release of membrane-bound annexin II and cortical cytoskeletal elements by sequestration of membrane cholesterol. Mol. Biol. Cell 8, 533–545 (1997).
Oling, F., Bergsma-Schutter, W. & Brisson, A. Trimers, dimers of trimers, and trimers of trimers are common building blocks of annexin a5 two-dimensional crystals. J. Struct. Biol. 133, 55–63 (2001).
Oliferenko, S. et al. Analysis of CD44-containing lipid rafts: recruitment of annexin II and stabilization by the actin cytoskeleton. J. Cell Biol. 146, 843–854 (1999).
Steinman, R. M., Mellman, I. S., Muller, W. A. & Cohn, Z. A. Endocytosis and the recycling of plasma membrane. J. Cell Biol. 96, 1–27 (1983).
Lafont, F., Lecat, S., Verkade, P. & Simons, K. Annexin XIIIb associates with lipid microdomains to function in apical delivery. J. Cell Biol. 142, 1413–1427 (1998).
Aniento, F., Gu, F., Parton, R. & Gruenberg, J. An endosomal βcop is involved in the pH-dependent formation of transport vesicles destined for late endosomes. J. Cell Biol. 133, 29–41 (1996).
Gu, F., Aniento, F., Parton, R. & Gruenberg, J. Functional dissection of COP-I subunits in the biogenesis of multivesicular endosomes. J. Cell Biol. 139, 1183–1195 (1997).
Gu, F. & Gruenberg, J. ARF1 regulates pH-dependent COP functions in the early endocytic pathway. J. Biol. Chem. 275, 8154–8160 (2000).
Daro, E., Sheff, D., Gomez, M., Kreis, T. & Mellman, I. Inhibition of endosome function in CHO cells bearing a temperature-sensitive defect in the coatomer (COPI) component εCOP. J. Cell Biol. 139, 1747–1759 (1997).
Whitney, J. A., Gomez, M., Sheff, D., Kreis, T. E. & Mellman, I. Cytoplasmic coat proteins involved in endosome function. Cell 83, 703–713 (1995).
Press, B., Feng, Y., Hoflack, B. & Wandinger-Ness, A. Mutant Rab7 causes the accumulation of cathepsin D and cation-independent mannose 6-phosphate receptor in an early endocytic compartment. J. Cell Biol. 140, 1075–1089 (1998).
Zerial, M. & McBride, H. Rab proteins as membrane organizers. Nature Rev. Mol. Cell Biol. 2, 107–117 (2001).
Acknowledgements
H.S. is supported by the Norwegian Cancer Society and the Research Council of Norway, and J.G. by the Swiss National Science Foundation and the Human Frontier Science Programme Organization.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Gruenberg, J., Stenmark, H. The biogenesis of multivesicular endosomes. Nat Rev Mol Cell Biol 5, 317–323 (2004). https://doi.org/10.1038/nrm1360
Issue Date:
DOI: https://doi.org/10.1038/nrm1360