Abstract
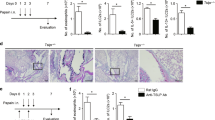
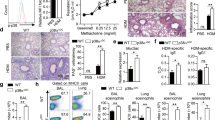
Members of the suppressor of cytokine signaling (SOCS) family are involved in the pathogenesis of many inflammatory diseases. SOCS-3 is predominantly expressed in T-helper type 2 (TH2) cells, but its role in TH2-related allergic diseases remains to be investigated. In this study we provide a strong correlation between SOCS-3 expression and the pathology of asthma and atopic dermatitis, as well as serum IgE levels in allergic human patients. SOCS-3 transgenic mice showed increased TH2 responses and multiple pathological features characteristic of asthma in an airway hypersensitivity model system. In contrast, dominant-negative mutant SOCS-3 transgenic mice, as well as mice with a heterozygous deletion of Socs3, had decreased TH2 development. These data indicate that SOCS-3 has an important role in regulating the onset and maintenance of TH2-mediated allergic immune disease, and suggest that SOCS-3 may be a new therapeutic target for the development of antiallergic drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wuthrich, B. Serum IgE in atopic dermatitis: relationship to severity of cutaneous involvement and course of disease as well as coexistence of atopic respiratory diseases. Clin. Allergy 8, 241–248 (1978).
Kuhn, R., Rajewsky, K. & Muller, W. Generation and analysis of interleukin-4 deficient mice. Science 254, 707–710 (1991).
Seder, R.A. et al. The presence of interleukin 4 during in vitro priming determines the lymphokine-producing potential of CD4+ T cells from T cell receptor transgenic mice. J. Exp. Med. 176, 1091–1098 (1992).
Kopf, M. et al. Disruption of the murine IL-4 gene blocks Th2 cytokine responses. Nature 362, 245–248 (1993).
Yasukawa, H., Sasaki, A. & Yoshimura, A. Negative regulation of cytokine signaling pathways. Annu. Rev. Immunol. 18, 143–164 (2000).
Krebs, D.L. & Hilton, D.J. SOCS: physiological suppressors of cytokine signaling. J. Cell. Sci. 113, 2813–2819 (2000).
Yoshimura, A. et al. A novel cytokine-inducible gene CIS encodes an SH2-containing protein that binds to tyrosine-phosphorylated interleukin 3 and erythropoietin receptors. EMBO J. 14, 2816–2826 (1995).
Starr, R. et al. A family of cytokine-inducible inhibitors of signalling. Nature 387, 917–921 (1997).
Endo, T.A. et al. A new protein containing an SH2 domain that inhibits JAK kinases. Nature 387, 921–924 (1997).
Naka, T. et al. Structure and function of a new STAT-induced STAT inhibitor. Nature 387, 924–929 (1997).
Alexander, W.S. Suppressors of cytokine signalling (SOCS) in the immune system. Nat. Rev. Immunol. 2, 410–416 (2002).
Hilton, D.J. et al. Twenty proteins containing a C-terminal SOCS box form five structural classes. Proc. Natl. Acad. Sci. USA 95, 114–119 (1998).
Masuhara, M. et al. Cloning and characterization of novel CIS family genes. Biochem. Biophys. Res. Commun. 239, 439–446 (1997).
Adams, T.E. et al. Growth hormone preferentially induces the rapid, transient expression of SOCS-3, a novel inhibitor of cytokine receptor signaling. J. Biol. Chem. 273, 1285–1287 (1998).
Bjorbaek, C., Elmquist, J.K., Frantz, J.D., Shoelson, S.E. & Flier, J.S. Identification of SOCS-3 as a potential mediator of central leptin resistance. Mol. Cell 1, 619–625 (1998).
Duval, D., Reinhardt, B., Kedinger, C. & Boeuf, H. Role of suppressors of cytokine signaling (Socs) in leukemia inhibitory factor (LIF)-dependent embryonic stem cell survival. FASEB J. 14, 1577–1584 (2000).
Suzuki, A. et al. CIS3/SOCS3/SSI3 plays a negative regulatory role in STAT3 activation and intestinal inflammation. J. Exp. Med. 193, 471–481 (2001).
Shouda, T. et al. Induction of the cytokine signal regulator SOCS3/CIS3 as a therapeutic strategy for treating inflammatory arthritis. J. Clin. Invest. 108, 1781–1788 (2001).
Marine, J.C. et al. SOCS3 is essential in the regulation of fetal liver erythropoiesis. Cell 98, 617–627 (1999).
Roberts, A.W. et al. Placental defects and embryonic lethality in mice lacking suppressor of cytokine signaling 3. Proc. Natl. Acad. Sci. USA 98, 9324–9329 (2001).
Seki, Y. et al. Expression of the suppressor of cytokine signaling-5 (SOCS5) negatively regulates IL-4-dependent STAT6 activation and Th2 differentiation. Proc. Natl. Acad. Sci. USA 99, 13003–13008 (2002).
Egwuagu, C.E. et al. Suppressors of cytokine signaling proteins are differentially expressed in Th1 and Th2 cells: implications Th cell lineage commitment and maintenance. J. Immunol. 168, 3181–3187 (2002).
Makino, S. Asthma prevention and management guidelines. Int. Arch. Allergy Appl. Immunol. 121 (suppl. 1), 1–14 (2000).
Matsumoto, Y. et al. Identification of highly expressed genes in peripheral blood T cells from patients with atopic dermatitis. Int. Arch. Allergy Appl. Immunol. (in the press).
Kibe, A. et al. Differential regulation by glucocorticoid of IL-13-induced eosinophilia, hyperresponsiveness, and goblet cell hyperplasia in mouse airways. Am. J. Respir. Crit. Care Med. 167, 50–56 (2003).
Szabo, S.J. et al. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 100, 655–669 (2000).
Szabo, S.J., Dighe, A.S., Gubler, U. & Murphy, K.M. Regulation of the interleukin (IL)-12R beta 2 subunit expression in developing T helper 1 (Th1) and Th2 cells. J. Exp. Med. 185, 817–824 (1997).
Afkarian, M. et al. T-bet is a STAT1-induced regulator of IL-12R expression in naive CD4+ T cells. Nat. Immunol. 3, 549–557 (2002).
Sasaki, A. et al. CIS3/SOCS-3 suppresses erythropoietin (EPO) signaling by binding the EPO receptor and JAK2. J. Biol. Chem. 275, 29338–29347 (2000).
Matsumoto, A. et al. A role of suppressor of cytokine signaling-3 (SOCS3/CIS3/SSI3) is CD28 mediated IL-2 production. J. Exp. Med. 197, 425–436 (2003).
Ring, J., Darsow, U. & Behrendt, H. Atopic eczema and allergy. Curr. Allergy Rep. 1, 39–43 (2001).
Kubo, M. et al. CD28 costimulation accelerates IL-4 receptor sensitivity and IL-4-mediated Th2 differentiation. J. Immunol. 163, 2432–2442 (1999).
Bour-Jordan, H. et al. CTLA-4 regulates the requirement for cytokine-induced signals in TH2 lineage commitment. Nat. Immunol. 4, 182–188 (2003).
Hanada, T. et al. A mutant form of JAB/SOCS1 augments the cytokine-induced JAK/STAT pathway by accelerating degradation of wild-type JAB/CIS family proteins through the SOCS-box. J. Biol. Chem. 276, 40746–40754 (2001).
Nomura, I. et al. Evaluation of the staphylococcal exotoxins and their specific IgE in childhood atopic dermatitis. J. Allergy Clin. Immunol. 104, 441–446 (1999).
Ferber, I.A. et al. GATA-3 significantly downregulates IFN-gamma production from developing Th1 cells in addition to inducing IL-4 and IL-5 levels. Clin. Immunol. 91, 134–144 (1999).
Barner, M., Mohrs, M., Brombacher, F. & Kopf, M. Differences between IL-4R alpha-deficient and IL-4-deficient mice reveal a role for IL-13 in the regulation of Th2 responses. Curr. Biol. 8, 669–672 (1998).
Acknowledgements
This work was supported by a Grant in Aid for Scientific Research on Priority Areas of the Ministry of Education, Culture, Sports, Science and Technology (Japan). Y.S. is a research fellow of the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Seki, Yi., Inoue, H., Nagata, N. et al. SOCS-3 regulates onset and maintenance of TH2-mediated allergic responses. Nat Med 9, 1047–1054 (2003). https://doi.org/10.1038/nm896
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm896
This article is cited by
-
IL-6 prevents Th2 cell polarization by promoting SOCS3-dependent suppression of IL-2 signaling
Cellular & Molecular Immunology (2023)
-
Haematological indices and immune response profiles in dogs naturally infected and co-infected with Dirofilaria repens and Babesia canis
Scientific Reports (2023)
-
Genes related to allergen exposure in allergic rhinitis: a gene-chip-based study in a mouse model
BMC Medical Genomics (2022)
-
Cytokine signaling pathway in cystic fibrosis: expression of SOCS and STATs genes in different clinical phenotypes of the disease
Molecular and Cellular Biochemistry (2021)
-
Igalan from Inula helenium (L.) suppresses the atopic dermatitis-like response in stimulated HaCaT keratinocytes via JAK/STAT3 signaling
Inflammation Research (2020)