Role of mucosal immunity and epithelial-vascular barrier in modulating gut homeostasis
- PMID: 37402104
- PMCID: PMC10504119
- DOI: 10.1007/s11739-023-03329-1
Role of mucosal immunity and epithelial-vascular barrier in modulating gut homeostasis
Abstract
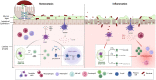
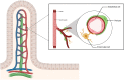
The intestinal mucosa represents the most extensive human barrier having a defense function against microbial and food antigens. This barrier is represented externally by a mucus layer, consisting mainly of mucins, antimicrobial peptides, and secretory immunoglobulin A (sIgA), which serves as the first interaction with the intestinal microbiota. Below is placed the epithelial monolayer, comprising enterocytes and specialized cells, such as goblet cells, Paneth cells, enterochromaffin cells, and others, each with a specific protective, endocrine, or immune function. This layer interacts with both the luminal environment and the underlying lamina propria, where mucosal immunity processes primarily take place. Specifically, the interaction between the microbiota and an intact mucosal barrier results in the activation of tolerogenic processes, mainly mediated by FOXP3+ regulatory T cells, underlying intestinal homeostasis. Conversely, the impairment of the mucosal barrier function, the alteration of the normal luminal microbiota composition (dysbiosis), or the imbalance between pro- and anti-inflammatory mucosal factors may result in inflammation and disease. Another crucial component of the intestinal barrier is the gut-vascular barrier, formed by endothelial cells, pericytes, and glial cells, which regulates the passage of molecules into the bloodstream. The aim of this review is to examine the various components of the intestinal barrier, assessing their interaction with the mucosal immune system, and focus on the immunological processes underlying homeostasis or inflammation.
Keywords: Chronic inflammation; Epithelial monolayer; Gut–vascular barrier; Microbiota; Mucosal immunity.
© 2023. The Author(s).
Conflict of interest statement
None to disclose.
Figures

Similar articles
-
Mucosal innate immune cells regulate both gut homeostasis and intestinal inflammation.Eur J Immunol. 2013 Dec;43(12):3108-15. doi: 10.1002/eji.201343782. Epub 2013 Oct 23. Eur J Immunol. 2013. PMID: 24414823 Review.
-
Secretory IgA in Intestinal Mucosal Secretions as an Adaptive Barrier against Microbial Cells.Int J Mol Sci. 2020 Dec 4;21(23):9254. doi: 10.3390/ijms21239254. Int J Mol Sci. 2020. PMID: 33291586 Free PMC article. Review.
-
Interaction between the gut microbiome and mucosal immune system.Mil Med Res. 2017 Apr 27;4:14. doi: 10.1186/s40779-017-0122-9. eCollection 2017. Mil Med Res. 2017. PMID: 28465831 Free PMC article. Review.
-
Goblet cells: multifaceted players in immunity at mucosal surfaces.Mucosal Immunol. 2018 Nov;11(6):1551-1557. doi: 10.1038/s41385-018-0039-y. Epub 2018 Jun 4. Mucosal Immunol. 2018. PMID: 29867079 Free PMC article. Review.
-
Role of Goblet Cells in Intestinal Barrier and Mucosal Immunity.J Inflamm Res. 2021 Jul 13;14:3171-3183. doi: 10.2147/JIR.S318327. eCollection 2021. J Inflamm Res. 2021. PMID: 34285541 Free PMC article. Review.
Cited by
-
New insights into the intestinal barrier through "gut-organ" axes and a glimpse of the microgravity's effects on intestinal barrier.Front Physiol. 2024 Oct 10;15:1465649. doi: 10.3389/fphys.2024.1465649. eCollection 2024. Front Physiol. 2024. PMID: 39450142 Free PMC article. Review.
-
Dietary astragalin confers protection against lipopolysaccharide-induced intestinal mucosal barrier damage through mitigating inflammation and modulating intestinal microbiota.Front Nutr. 2024 Oct 2;11:1481203. doi: 10.3389/fnut.2024.1481203. eCollection 2024. Front Nutr. 2024. PMID: 39421621 Free PMC article.
-
Comparative analysis of the effects of cyclophosphamide and dexamethasone on intestinal immunity and microbiota in delayed hypersensitivity mice.PLoS One. 2024 Oct 17;19(10):e0312147. doi: 10.1371/journal.pone.0312147. eCollection 2024. PLoS One. 2024. PMID: 39418230 Free PMC article.
-
Successful Treatment of Complicated Pyogenic Spondylitis Due to Advanced Rectosigmoid Cancer Utilizing Vigorous Antibiotic Therapy and Minimally Invasive Robotic Colorectal Surgery: A Case Report.Cureus. 2024 Aug 22;16(8):e67536. doi: 10.7759/cureus.67536. eCollection 2024 Aug. Cureus. 2024. PMID: 39310526 Free PMC article.
-
Probiotics in miRNA-Mediated Regulation of Intestinal Immune Homeostasis in Pigs: A Physiological Narrative.Microorganisms. 2024 Aug 7;12(8):1606. doi: 10.3390/microorganisms12081606. Microorganisms. 2024. PMID: 39203448 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

