Immunotherapeutic approaches in EBV-associated nasopharyngeal carcinoma
- PMID: 36713430
- PMCID: PMC9875085
- DOI: 10.3389/fimmu.2022.1079515
Immunotherapeutic approaches in EBV-associated nasopharyngeal carcinoma
Abstract
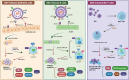
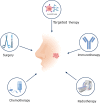
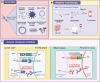
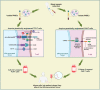
Epstein-Barr virus (EBV) was the first tumor virus in humans. Nasopharyngeal carcinoma (NPC) accounts for approximately 60% of the 200,000 new tumor cases caused by EBV infection worldwide each year. NPC has an insidious onset and is highly malignant, with more than 70% of patients having intermediate to advanced disease at the time of initial diagnosis, and is strongly implicated in epithelial cancers as well as malignant lymphoid and natural killer/T cell lymphomas. Over 90% of patients with confirmed undifferentiated NPC are infected with EBV. In recent decades, much progress has been made in understanding the molecular mechanisms of NPC and developing therapeutic approaches. Radiotherapy and chemotherapy are the main treatment options for NPC; however, they have a limited efficacy in patients with locally advanced or distant metastatic tumors. Tumor immunotherapy, including vaccination, adoptive cell therapy, and immune checkpoint blockade, represents a promising therapeutic approach for NPC. Significant breakthroughs have recently been made in the application of immunotherapy for patients with recurrent or metastatic NPC (RM-NPC), indicating a broad prospect for NPC immunotherapy. Here, we review important research findings regarding immunotherapy for NPC patients and provide insights for future research.
Keywords: EBV-associated cancer; EBV-directed vaccination; Epstein-Barr virus; TCR-T therapy; adoptive cell therapy; immune checkpoint inhibitors.
Copyright © 2023 Li, Duan, Chen, Zhan, Peng, Meng, Li, Li, Pang and Dou.
Conflict of interest statement
X-YL is an employee of OriCell Therapeutics. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be constructed as a potential conflict of interest.
Figures
Similar articles
-
Immunotherapy for nasopharyngeal carcinoma: Current status and prospects (Review).Int J Oncol. 2023 Aug;63(2):97. doi: 10.3892/ijo.2023.5545. Epub 2023 Jul 7. Int J Oncol. 2023. PMID: 37417358 Free PMC article. Review.
-
Immunotherapeutic approaches in nasopharyngeal carcinoma.Expert Opin Biol Ther. 2019 Nov;19(11):1165-1172. doi: 10.1080/14712598.2019.1650910. Epub 2019 Aug 13. Expert Opin Biol Ther. 2019. PMID: 31361154 Review.
-
Immunotherapy for EBV-Associated Nasopharyngeal Carcinoma.Crit Rev Oncog. 2018;23(3-4):219-234. doi: 10.1615/CritRevOncog.2018027528. Crit Rev Oncog. 2018. PMID: 30311576 Review.
-
Immunotherapy for the treatment of advanced nasopharyngeal carcinoma: a promising new era.J Cancer Res Clin Oncol. 2023 May;149(5):2071-2079. doi: 10.1007/s00432-022-04214-8. Epub 2022 Jul 25. J Cancer Res Clin Oncol. 2023. PMID: 35876949 Review.
-
Effective treatment of metastatic forms of Epstein-Barr virus-associated nasopharyngeal carcinoma with a novel adenovirus-based adoptive immunotherapy.Cancer Res. 2012 Mar 1;72(5):1116-25. doi: 10.1158/0008-5472.CAN-11-3399. Epub 2012 Jan 26. Cancer Res. 2012. PMID: 22282657 Clinical Trial.
Cited by
-
Dihydrotestosterone Enhances MICA-Mediated Immune Responses to Epstein-Barr Virus-Associated Gastric Carcinoma.Cancers (Basel). 2024 Sep 21;16(18):3219. doi: 10.3390/cancers16183219. Cancers (Basel). 2024. PMID: 39335190 Free PMC article.
-
Multi-Omics Exploration of the Mechanism of Curcumol to Reduce Invasion and Metastasis of Nasopharyngeal Carcinoma by Inhibiting NCL/EBNA1-Mediated UBE2C Upregulation.Biomolecules. 2024 Sep 9;14(9):1142. doi: 10.3390/biom14091142. Biomolecules. 2024. PMID: 39334908 Free PMC article.
-
The Impact of Oral Microbiome Dysbiosis on the Aetiology, Pathogenesis, and Development of Oral Cancer.Cancers (Basel). 2024 Aug 28;16(17):2997. doi: 10.3390/cancers16172997. Cancers (Basel). 2024. PMID: 39272855 Free PMC article. Review.
-
Single-cell resolution profiling of the immune microenvironment in primary and metastatic nasopharyngeal carcinoma.J Cancer Res Clin Oncol. 2024 Aug 19;150(8):391. doi: 10.1007/s00432-024-05900-5. J Cancer Res Clin Oncol. 2024. PMID: 39158776 Free PMC article.
-
Immune checkpoint inhibitor combined with chemotherapy versus chemotherapy alone in the first-line treatment for recurrent or metastatic nasopharyngeal carcinoma: a meta-analysis of random controlled trials.Eur Arch Otorhinolaryngol. 2024 Oct;281(10):5111-5118. doi: 10.1007/s00405-024-08768-w. Epub 2024 Jun 24. Eur Arch Otorhinolaryngol. 2024. PMID: 38914820 Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

