A pan-cancer mycobiome analysis reveals fungal involvement in gastrointestinal and lung tumors
- PMID: 36179671
- PMCID: PMC9564002
- DOI: 10.1016/j.cell.2022.09.015
A pan-cancer mycobiome analysis reveals fungal involvement in gastrointestinal and lung tumors
Abstract
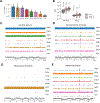
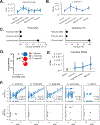
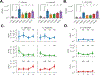
Fungal microorganisms (mycobiota) comprise a small but immunoreactive component of the human microbiome, yet little is known about their role in human cancers. Pan-cancer analysis of multiple body sites revealed tumor-associated mycobiomes at up to 1 fungal cell per 104 tumor cells. In lung cancer, Blastomyces was associated with tumor tissues. In stomach cancers, high rates of Candida were linked to the expression of pro-inflammatory immune pathways, while in colon cancers Candida was predictive of metastatic disease and attenuated cellular adhesions. Across multiple GI sites, several Candida species were enriched in tumor samples and tumor-associated Candida DNA was predictive of decreased survival. The presence of Candida in human GI tumors was confirmed by external ITS sequencing of tumor samples and by culture-dependent analysis in an independent cohort. These data implicate the mycobiota in the pathogenesis of GI cancers and suggest that tumor-associated fungal DNA may serve as diagnostic or prognostic biomarkers.
Keywords: Blastomyces; Candida; Malassezia; cancer; colon cancer; lung cancer; mycobiome; stomach cancer; trans-kingdom interactions; tumor-associated fungi.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests X.S. is the co-founder and CEO of Xilis, Inc. This study and its findings do not have any overlap or implications over Xilis’ commercial interests. A.B.D., X.L. and I.D.I. are inventors on a US provisional patent application, covering inventions described in this manuscript. The authors declare that they have no conflicts of interest with the contents of this article.
Figures




Comment in
-
The tumor mycobiome: A paradigm shift in cancer pathogenesis.Cell. 2022 Sep 29;185(20):3648-3651. doi: 10.1016/j.cell.2022.09.013. Cell. 2022. PMID: 36179665 Free PMC article.
-
Fungi in cancer: not such a 'fun-guy'.Nat Rev Cancer. 2022 Dec;22(12):659. doi: 10.1038/s41568-022-00533-7. Nat Rev Cancer. 2022. PMID: 36323799 No abstract available.
Similar articles
-
Host Factors Associated with Gut Mycobiome Structure.mSystems. 2023 Apr 27;8(2):e0098622. doi: 10.1128/msystems.00986-22. Epub 2023 Feb 14. mSystems. 2023. PMID: 36786595 Free PMC article.
-
The gut mycobiome of the Human Microbiome Project healthy cohort.Microbiome. 2017 Nov 25;5(1):153. doi: 10.1186/s40168-017-0373-4. Microbiome. 2017. PMID: 29178920 Free PMC article.
-
Breastmilk and NICU surfaces are potential sources of fungi for infant mycobiomes.Fungal Genet Biol. 2019 Jul;128:29-35. doi: 10.1016/j.fgb.2019.03.008. Epub 2019 Mar 21. Fungal Genet Biol. 2019. PMID: 30905830 Free PMC article.
-
Critically Appraising the Significance of the Oral Mycobiome.J Dent Res. 2021 Feb;100(2):133-140. doi: 10.1177/0022034520956975. Epub 2020 Sep 13. J Dent Res. 2021. PMID: 32924741 Free PMC article. Review.
-
Unveiling the hidden players: exploring the role of gut mycobiome in cancer development and treatment dynamics.Gut Microbes. 2024 Jan-Dec;16(1):2328868. doi: 10.1080/19490976.2024.2328868. Epub 2024 Mar 14. Gut Microbes. 2024. PMID: 38485702 Free PMC article. Review.
Cited by
-
Magnetic Nanobead Paper-Based Biosensors for Colorimetric Detection of Candida albicans.ACS Omega. 2024 Oct 7;9(42):43005-43015. doi: 10.1021/acsomega.4c05941. eCollection 2024 Oct 22. ACS Omega. 2024. PMID: 39464470 Free PMC article.
-
Gastrointestinal Microbiota in Gastric Cancer: Potential Mechanisms and Clinical Applications-A Literature Review.Cancers (Basel). 2024 Oct 21;16(20):3547. doi: 10.3390/cancers16203547. Cancers (Basel). 2024. PMID: 39456641 Free PMC article. Review.
-
Tertiary Lymphoid Structures in Microorganism-Related Cancer.Cancers (Basel). 2024 Oct 12;16(20):3464. doi: 10.3390/cancers16203464. Cancers (Basel). 2024. PMID: 39456558 Free PMC article. Review.
-
Effects of intratumoral microbiota on tumorigenesis, anti-tumor immunity, and microbe-based cancer therapy.Front Oncol. 2024 Sep 26;14:1429722. doi: 10.3389/fonc.2024.1429722. eCollection 2024. Front Oncol. 2024. PMID: 39391251 Free PMC article. Review.
-
Intratumoral microbiota in colorectal cancer: focus on specific distribution and potential mechanisms.Cell Commun Signal. 2024 Sep 26;22(1):455. doi: 10.1186/s12964-024-01831-3. Cell Commun Signal. 2024. PMID: 39327582 Free PMC article. Review.
References
-
- Benedict K, Roy M, Chiller T, and Davis JP (2012). Epidemiologic and Ecologic Features of Blastomycosis: A Review. Current Fungal Infection Reports 6, 327–335.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

