HDAC11 promotes both NLRP3/caspase-1/GSDMD and caspase-3/GSDME pathways causing pyroptosis via ERG in vascular endothelial cells
- PMID: 35279683
- PMCID: PMC8918356
- DOI: 10.1038/s41420-022-00906-9
HDAC11 promotes both NLRP3/caspase-1/GSDMD and caspase-3/GSDME pathways causing pyroptosis via ERG in vascular endothelial cells
Abstract
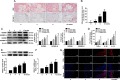
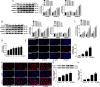
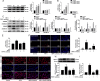
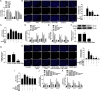
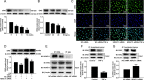
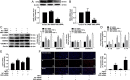
Histone deacetylase 11 (HDAC11), a sole member of the class IV HDAC subfamily, participates in various cardiovascular diseases. Recent evidence showed that pyroptosis was a form of inflammatory programmed cell death and is critical for atherosclerosis (AS). However, little is known about the effect of HDAC11 on endothelial cell pyroptosis in AS. Thus, this study aims to investigate the role of HDAC11 in vascular endothelial cell pyroptosis and its molecular mechanism. Firstly, we found that HDAC11 expression was up-regulated and pyroptosis occurred in the aorta of ApoE-/- mice fed with a high-fat diet (HFD) for 8 or 12 weeks. Then, in vitro study found the treatment of human umbilical vein endothelial cells (HUVECs) with tumor necrosis factor-α (TNF-α) resulted in pyroptosis, as evidenced by activation of caspase-1 and caspase-3 activation, cleavage of downstream gasdermin D (GSDMD) and gasdermin E (GSDME/DFNA5), the release of pro-inflammatory cytokines interleukin (IL)-1β, IL-6 and IL-18, as well as elevation of LDH activity and increase of propidium iodide (PI)-positive cells. Besides, TNF-α increased HDAC11 expression and induced pyroptosis via TNFR1 in HUVECs. HDAC11 knockdown mitigated pyroptosis by suppressing both NLRP3/caspase-1/GSDMD and caspase-3/GSDME pathways in TNF-α-induced HUVECs. Moreover, GSDME knockdown by siRNA significantly decreased pyroptosis and inflammatory response, while treatment with disulfiram or necrosulfonamide (NSA) further augmented the inhibitory effects of GSDME siRNA on pyroptosis and inflammatory response. Further studies found HDAC11 formed a complex with ERG and decreased the acetylation levels of ERG. More importantly, ERG knockdown augmented vascular endothelial cell pyroptosis in TNF-α-induced HUVECs. Taken together, our study suggests that HDAC11 might promote both NLRP3/caspase-1/GSDMD and caspase-3/GSDME pathways leading to pyroptosis via regulation of ERG acetylation in HUVECs. Modulation of HDAC11 may serve as a potential target for therapeutic strategies of AS.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Hydroxytyrosol Acetate Inhibits Vascular Endothelial Cell Pyroptosis via the HDAC11 Signaling Pathway in Atherosclerosis.Front Pharmacol. 2021 Apr 23;12:656272. doi: 10.3389/fphar.2021.656272. eCollection 2021. Front Pharmacol. 2021. PMID: 33967800 Free PMC article.
-
Effects of amyloid β (Aβ)42 and Gasdermin D on the progression of Alzheimer's disease in vitro and in vivo through the regulation of astrocyte pyroptosis.Aging (Albany NY). 2023 Nov 2;15(21):12209-12224. doi: 10.18632/aging.205174. Epub 2023 Nov 2. Aging (Albany NY). 2023. PMID: 37921870 Free PMC article.
-
Gasdermin E-mediated keratinocyte pyroptosis participates in the pathogenesis of psoriasis by promoting skin inflammation.Br J Dermatol. 2024 Aug 14;191(3):385-396. doi: 10.1093/bjd/ljae179. Br J Dermatol. 2024. PMID: 38655652
-
Mini-Review: GSDME-Mediated Pyroptosis in Diabetic Nephropathy.Front Pharmacol. 2021 Nov 16;12:780790. doi: 10.3389/fphar.2021.780790. eCollection 2021. Front Pharmacol. 2021. PMID: 34867412 Free PMC article. Review.
-
The pyroptotic role of Caspase-3/GSDME signalling pathway among various cancer: A Review.Int J Biol Macromol. 2023 Jul 1;242(Pt 2):124832. doi: 10.1016/j.ijbiomac.2023.124832. Epub 2023 May 15. Int J Biol Macromol. 2023. PMID: 37196719 Review.
Cited by
-
Regulation of cardiovascular diseases by histone deacetylases and NADPH oxidases.Redox Biol. 2024 Oct 4;77:103379. doi: 10.1016/j.redox.2024.103379. Online ahead of print. Redox Biol. 2024. PMID: 39378612 Free PMC article. Review.
-
Histone deacetylases: Regulation of vascular homeostasis via endothelial cells and vascular smooth muscle cells and the role in vascular pathogenesis.Genes Dis. 2024 Jan 22;11(6):101216. doi: 10.1016/j.gendis.2024.101216. eCollection 2024 Nov. Genes Dis. 2024. PMID: 39281836 Free PMC article. Review.
-
Miltirone induces GSDME-dependent pyroptosis in colorectal cancer by activating caspase 3.Heliyon. 2024 Aug 20;10(16):e36603. doi: 10.1016/j.heliyon.2024.e36603. eCollection 2024 Aug 30. Heliyon. 2024. PMID: 39262975 Free PMC article.
-
Role of gasdermin D in inflammatory diseases: from mechanism to therapeutics.Front Immunol. 2024 Aug 26;15:1456244. doi: 10.3389/fimmu.2024.1456244. eCollection 2024. Front Immunol. 2024. PMID: 39253076 Free PMC article. Review.
-
Epigenetic regulation of diverse regulated cell death modalities in cardiovascular disease: Insights into necroptosis, pyroptosis, ferroptosis, and cuproptosis.Redox Biol. 2024 Oct;76:103321. doi: 10.1016/j.redox.2024.103321. Epub 2024 Aug 19. Redox Biol. 2024. PMID: 39186883 Free PMC article. Review.
References
-
- Fan XD, Wan LL, Duan M, Lu S. HDAC11 deletion reduces fructose-induced cardiac dyslipidemia, apoptosis and inflammation by attenuating oxidative stress injury. Biochem Biophys Res Commun. 2018;503:444–51. - PubMed
-
- Zhou B, Zeng S, Li N, Yu L, Yang G, Yang Y, et al. Angiogenic factor with G patch and FHA domains 1 is a novel regulator of vascular injury. Arterioscler Thromb Vasc Biol. 2017;37:675–84. - PubMed
-
- Zhang R, Ge J. Proteinase-activated receptor-2 modulates Ve-cadherin expression to affect human vascular endothelial barrier function. J Cell Biochem. 2017;118:4587–93. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

