Ghrelin octanoylation by ghrelin O-acyltransferase: protein acylation impacting metabolic and neuroendocrine signalling
- PMID: 34315274
- PMCID: PMC8316800
- DOI: 10.1098/rsob.210080
Ghrelin octanoylation by ghrelin O-acyltransferase: protein acylation impacting metabolic and neuroendocrine signalling
Abstract
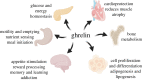
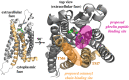
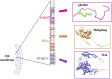
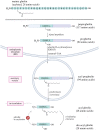
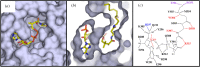
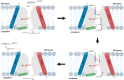
The acylated peptide hormone ghrelin impacts a wide range of physiological processes but is most well known for controlling hunger and metabolic regulation. Ghrelin requires a unique posttranslational modification, serine octanoylation, to bind and activate signalling through its cognate GHS-R1a receptor. Ghrelin acylation is catalysed by ghrelin O-acyltransferase (GOAT), a member of the membrane-bound O-acyltransferase (MBOAT) enzyme family. The ghrelin/GOAT/GHS-R1a system is defined by multiple unique aspects within both protein biochemistry and endocrinology. Ghrelin serves as the only substrate for GOAT within the human proteome and, among the multiple hormones involved in energy homeostasis and metabolism such as insulin and leptin, acts as the only known hormone in circulation that directly stimulates appetite and hunger signalling. Advances in GOAT enzymology, structural modelling and inhibitor development have revolutionized our understanding of this enzyme and offered new tools for investigating ghrelin signalling at the molecular and organismal levels. In this review, we briefly summarize the current state of knowledge regarding ghrelin signalling and ghrelin/GOAT enzymology, discuss the GOAT structural model in the context of recently reported MBOAT enzyme superfamily member structures, and highlight the growing complement of GOAT inhibitors that offer options for both ghrelin signalling studies and therapeutic applications.
Keywords: GHS-R1a; ghrelin; ghrelin O-acyltransferase; membrane-bound O-acyltransferase; neuroendocrinology; protein acylation.
Figures




Similar articles
-
Ghrelin octanoylation by ghrelin O-acyltransferase: Unique protein biochemistry underlying metabolic signaling.Biochem Soc Trans. 2019 Feb 28;47(1):169-178. doi: 10.1042/BST20180436. Epub 2019 Jan 9. Biochem Soc Trans. 2019. PMID: 30626708 Review.
-
Functional group and stereochemical requirements for substrate binding by ghrelin O-acyltransferase revealed by unnatural amino acid incorporation.Bioorg Chem. 2018 Sep;79:98-106. doi: 10.1016/j.bioorg.2018.04.009. Epub 2018 Apr 21. Bioorg Chem. 2018. PMID: 29738973
-
Biochemical Assays for Ghrelin Acylation and Inhibition of Ghrelin O-Acyltransferase.Methods Mol Biol. 2019;2009:227-241. doi: 10.1007/978-1-4939-9532-5_18. Methods Mol Biol. 2019. PMID: 31152408
-
An overview of ghrelin O-acyltransferase inhibitors: a literature and patent review for 2010-2019.Expert Opin Ther Pat. 2020 Aug;30(8):581-593. doi: 10.1080/13543776.2020.1776263. Epub 2020 Jun 21. Expert Opin Ther Pat. 2020. PMID: 32564644 Free PMC article. Review.
-
Structure-activity analysis of human ghrelin O-acyltransferase reveals chemical determinants of ghrelin selectivity and acyl group recognition.Biochemistry. 2015 Feb 3;54(4):1100-10. doi: 10.1021/bi5010359. Epub 2015 Jan 21. Biochemistry. 2015. PMID: 25562443
Cited by
-
The Impact of Gastrointestinal Hormones on Human Adipose Tissue Function.Nutrients. 2024 Sep 25;16(19):3245. doi: 10.3390/nu16193245. Nutrients. 2024. PMID: 39408213 Free PMC article. Review.
-
Detection of natural autoimmunity to ghrelin in diabetes mellitus.Front Med Technol. 2024 Jul 12;6:1407409. doi: 10.3389/fmedt.2024.1407409. eCollection 2024. Front Med Technol. 2024. PMID: 39070294 Free PMC article.
-
Emerging Relevance of Ghrelin in Programmed Cell Death and Its Application in Diseases.Int J Mol Sci. 2023 Dec 8;24(24):17254. doi: 10.3390/ijms242417254. Int J Mol Sci. 2023. PMID: 38139082 Free PMC article. Review.
-
Methylation analysis by targeted bisulfite sequencing in large for gestational age (LGA) newborns: the LARGAN cohort.Clin Epigenetics. 2023 Dec 13;15(1):191. doi: 10.1186/s13148-023-01612-8. Clin Epigenetics. 2023. PMID: 38093359 Free PMC article.
-
Anti-Obesity Medications and Investigational Agents: An Obesity Medicine Association (OMA) Clinical Practice Statement (CPS) 2022.Obes Pillars. 2022 Apr 15;2:100018. doi: 10.1016/j.obpill.2022.100018. eCollection 2022 Jun. Obes Pillars. 2022. PMID: 37990711 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

