Gut Microbiota Interventions With Clostridium butyricum and Norfloxacin Modulate Immune Response in Experimental Autoimmune Encephalomyelitis Mice
- PMID: 31428083
- PMCID: PMC6689973
- DOI: 10.3389/fimmu.2019.01662
Gut Microbiota Interventions With Clostridium butyricum and Norfloxacin Modulate Immune Response in Experimental Autoimmune Encephalomyelitis Mice
Abstract
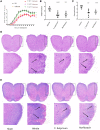
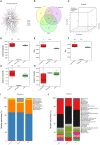
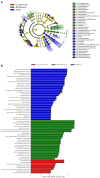
Gut microbiota has been proposed as an important environmental factor which can intervene and modulate central nervous system autoimmunity. Here, we altered the composition of gut flora with Clostridium butyricum and norfloxacin in experimental autoimmune encephalomyelitis (EAE), an animal model of multiple sclerosis. We found that appropriate C. butyricum (5.0 × 106 CFU/mL intragastrically daily, staring at weaning period of age) and norfloxacin (5 mg/kg intragastrically daily, 1 week prior to EAE induction) treatment could both ameliorate EAE although there are obvious differences in gut microbiota composition between these two interventions. C. butyricum increased while norfloxacin decreased the abundance and diversity of the gut microbiota in EAE mice, and both of the treatments decreased firmicutes/bacteroidetes ratio. In the genus level, C. butyricum treatment increased the abundance of Prevotella while Akkermansia and Allobaculum increased in norfloxacin treatment. Moreover, both interventions reduced Desulfovibroneceae and Ruminococcus species. Although there was discrepancy in the gut microbiota composition with the two interventions, C. butyricum and norfloxacin treatment both reduced Th17 response and increased Treg response in the gastrointestinal tract and extra-gastrointestinal organ systems in EAE mice. And the reduced activity of p38 mitogen-activated kinase and c-Jun N-terminal kinase signaling in spinal cord could be observed in the two interventions. The results suggested that manipulation of gut microbiota interventions should take factors such as timing, duration, and dosage into consideration. The discrepancy in the gut microbiota composition and the similar protective T cells response of C. butyricum and norfloxacin implies that achieving intestinal microecology balance by promoting and/or inhibiting the gut microbiota contribute to the well-being of immune response in EAE mice.
Keywords: Clostridium butyricum; EAE; Th17/Treg; gut microbiota; multiple sclerosis; norfloxacin.
Figures

Similar articles
-
Constipation induced gut microbiota dysbiosis exacerbates experimental autoimmune encephalomyelitis in C57BL/6 mice.J Transl Med. 2021 Jul 23;19(1):317. doi: 10.1186/s12967-021-02995-z. J Transl Med. 2021. PMID: 34301274 Free PMC article.
-
Strength Exercise Confers Protection in Central Nervous System Autoimmunity by Altering the Gut Microbiota.Front Immunol. 2021 Mar 16;12:628629. doi: 10.3389/fimmu.2021.628629. eCollection 2021. Front Immunol. 2021. PMID: 33796102 Free PMC article.
-
Prebiotic inulin controls Th17 cells mediated central nervous system autoimmunity through modulating the gut microbiota and short chain fatty acids.Gut Microbes. 2024 Jan-Dec;16(1):2402547. doi: 10.1080/19490976.2024.2402547. Epub 2024 Sep 17. Gut Microbes. 2024. PMID: 39287045 Free PMC article.
-
Gut Microbiota in Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis: Current Applications and Future Perspectives.Mediators Inflamm. 2018 Apr 2;2018:8168717. doi: 10.1155/2018/8168717. eCollection 2018. Mediators Inflamm. 2018. PMID: 29805314 Free PMC article. Review.
-
The Gut-Brain Axis in Multiple Sclerosis. Is Its Dysfunction a Pathological Trigger or a Consequence of the Disease?Front Immunol. 2021 Sep 21;12:718220. doi: 10.3389/fimmu.2021.718220. eCollection 2021. Front Immunol. 2021. PMID: 34621267 Free PMC article. Review.
Cited by
-
Mitigative Effects of Topical Norfloxacin on an Imiquimod-Induced Murine Model of Psoriasis.ACS Pharmacol Transl Sci. 2024 Aug 2;7(9):2739-2754. doi: 10.1021/acsptsci.4c00152. eCollection 2024 Sep 13. ACS Pharmacol Transl Sci. 2024. PMID: 39296262
-
The joint protective function of live- and dead-Lactobacillus plantarum GKD7 on anterior cruciate ligament transection induces osteoarthritis.Aging (Albany NY). 2024 Sep 5;16(18):12559-12573. doi: 10.18632/aging.206101. Epub 2024 Sep 5. Aging (Albany NY). 2024. PMID: 39237298 Free PMC article.
-
Relationship between gut microbiota and multiple sclerosis: a scientometric visual analysis from 2010 to 2023.Front Immunol. 2024 Aug 19;15:1451742. doi: 10.3389/fimmu.2024.1451742. eCollection 2024. Front Immunol. 2024. PMID: 39224586 Free PMC article.
-
Short-chain fatty acids suppresses astrocyte activation by amplifying Trp-AhR-AQP4 signaling in experimental autoimmune encephalomyelitis mice.Cell Mol Life Sci. 2024 Jul 8;81(1):293. doi: 10.1007/s00018-024-05332-x. Cell Mol Life Sci. 2024. PMID: 38976012 Free PMC article.
-
The role of the "gut microbiota-mitochondria" crosstalk in the pathogenesis of multiple sclerosis.Front Microbiol. 2024 Apr 29;15:1404995. doi: 10.3389/fmicb.2024.1404995. eCollection 2024. Front Microbiol. 2024. PMID: 38741740 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

