Nuclear poly(ADP-ribose) activity is a therapeutic target in amyotrophic lateral sclerosis
- PMID: 30157956
- PMCID: PMC6114235
- DOI: 10.1186/s40478-018-0586-1
Nuclear poly(ADP-ribose) activity is a therapeutic target in amyotrophic lateral sclerosis
Abstract
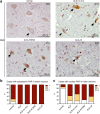
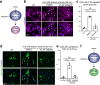
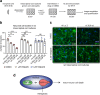
Amyotrophic lateral sclerosis (ALS) is a devastating and fatal motor neuron disease. Diagnosis typically occurs in the fifth decade of life and the disease progresses rapidly leading to death within ~ 2-5 years of symptomatic onset. There is no cure, and the few available treatments offer only a modest extension in patient survival. A protein central to ALS is the nuclear RNA/DNA-binding protein, TDP-43. In > 95% of ALS patients, TDP-43 is cleared from the nucleus and forms phosphorylated protein aggregates in the cytoplasm of affected neurons and glia. We recently defined that poly(ADP-ribose) (PAR) activity regulates TDP-43-associated toxicity. PAR is a posttranslational modification that is attached to target proteins by PAR polymerases (PARPs). PARP-1 and PARP-2 are the major enzymes that are active in the nucleus. Here, we uncovered that the motor neurons of the ALS spinal cord were associated with elevated nuclear PAR, suggesting elevated PARP activity. Veliparib, a small-molecule inhibitor of nuclear PARP-1/2, mitigated the formation of cytoplasmic TDP-43 aggregates in mammalian cells. In primary spinal-cord cultures from rat, Veliparib also inhibited TDP-43-associated neuronal death. These studies uncover that PAR activity is misregulated in the ALS spinal cord, and a small-molecular inhibitor of PARP-1/2 activity may have therapeutic potential in the treatment of ALS and related disorders associated with abnormal TDP-43 homeostasis.
Keywords: ABT-888/Veliparib; Motor neuron disease, primary neuron; PAR; PARylation; Parp; Poly(ADP-ribose); TDP-43.
Conflict of interest statement
Ethics approval and consent to participate
Informed consent was obtained from next of kin in accordance with institutional review board guidelines of the University of Pennsylvania.
Consent for publication
All authors consent to publication.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Poly(ADP-Ribosylation) in Age-Related Neurological Disease.Trends Genet. 2019 Aug;35(8):601-613. doi: 10.1016/j.tig.2019.05.004. Epub 2019 Jun 7. Trends Genet. 2019. PMID: 31182245 Free PMC article. Review.
-
Poly(ADP-Ribose) Prevents Pathological Phase Separation of TDP-43 by Promoting Liquid Demixing and Stress Granule Localization.Mol Cell. 2018 Sep 6;71(5):703-717.e9. doi: 10.1016/j.molcel.2018.07.002. Epub 2018 Aug 9. Mol Cell. 2018. PMID: 30100264 Free PMC article.
-
Poly-A binding protein-1 localization to a subset of TDP-43 inclusions in amyotrophic lateral sclerosis occurs more frequently in patients harboring an expansion in C9orf72.J Neuropathol Exp Neurol. 2014 Sep;73(9):837-45. doi: 10.1097/NEN.0000000000000102. J Neuropathol Exp Neurol. 2014. PMID: 25111021 Free PMC article.
-
GPNMB ameliorates mutant TDP-43-induced motor neuron cell death.J Neurosci Res. 2017 Aug;95(8):1647-1665. doi: 10.1002/jnr.23999. Epub 2016 Dec 9. J Neurosci Res. 2017. PMID: 27935101
-
Emerging small-molecule therapeutic approaches for amyotrophic lateral sclerosis and frontotemporal dementia.Bioorg Med Chem Lett. 2020 Feb 15;30(4):126942. doi: 10.1016/j.bmcl.2019.126942. Epub 2019 Dec 30. Bioorg Med Chem Lett. 2020. PMID: 31926785 Review.
Cited by
-
Poly(ADP-Ribosylation) in Age-Related Neurological Disease.Trends Genet. 2019 Aug;35(8):601-613. doi: 10.1016/j.tig.2019.05.004. Epub 2019 Jun 7. Trends Genet. 2019. PMID: 31182245 Free PMC article. Review.
-
Therapeutics-how to treat phase separation-associated diseases.Emerg Top Life Sci. 2020 Dec 11;4(3):307-318. doi: 10.1042/ETLS20190176. Emerg Top Life Sci. 2020. PMID: 32364240 Free PMC article. Review.
-
Poly ADP-ribose signaling is dysregulated in Huntington disease.Proc Natl Acad Sci U S A. 2024 Oct;121(40):e2318098121. doi: 10.1073/pnas.2318098121. Epub 2024 Sep 27. Proc Natl Acad Sci U S A. 2024. PMID: 39331414
-
Poly(ADP-ribose) Engages the TDP-43 Nuclear-Localization Sequence to Regulate Granulo-Filamentous Aggregation.Biochemistry. 2018 Dec 26;57(51):6923-6926. doi: 10.1021/acs.biochem.8b00910. Epub 2018 Dec 17. Biochemistry. 2018. PMID: 30540446 Free PMC article.
-
ADP-ribosylation signalling and human disease.Open Biol. 2019 Apr 26;9(4):190041. doi: 10.1098/rsob.190041. Open Biol. 2019. PMID: 30991935 Free PMC article. Review.
References
-
- Abdelkarim GE, Gertz K, Harms C, Katchanov J, Dirnagl U, Szabo C, Endres M. Protective effects of PJ34, a novel, potent inhibitor of poly(ADP-ribose) polymerase (PARP) in in vitro and in vivo models of stroke. Int J Mol Med. 2001;7:255–260. - PubMed
-
- Andrabi SA, Kang HC, Haince JF, Lee YI, Zhang J, Chi Z, West AB, Koehler RC, Poirier GG, Dawson TM, et al. Iduna protects the brain from glutamate excitotoxicity and stroke by interfering with poly(ADP-ribose) polymer-induced cell death. Nat Med. 2011;17:692–699. doi: 10.1038/nm.2387. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

