The Ebola Interferon Inhibiting Domains Attenuate and Dysregulate Cell-Mediated Immune Responses
- PMID: 27930745
- PMCID: PMC5145241
- DOI: 10.1371/journal.ppat.1006031
The Ebola Interferon Inhibiting Domains Attenuate and Dysregulate Cell-Mediated Immune Responses
Abstract
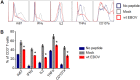
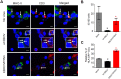
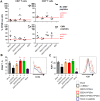
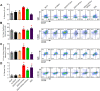
Ebola virus (EBOV) infections are characterized by deficient T-lymphocyte responses, T-lymphocyte apoptosis and lymphopenia. We previously showed that disabling of interferon-inhibiting domains (IIDs) in the VP24 and VP35 proteins effectively unblocks maturation of dendritic cells (DCs) and increases the secretion of cytokines and chemokines. Here, we investigated the role of IIDs in adaptive and innate cell-mediated responses using recombinant viruses carrying point mutations, which disabled IIDs in VP24 (EBOV/VP24m), VP35 (EBOV/VP35m) or both (EBOV/VP35m/VP24m). Peripheral blood mononuclear cells (PBMCs) from cytomegalovirus (CMV)-seropositive donors were inoculated with the panel of viruses and stimulated with CMV pp65 peptides. Disabling of the VP35 IID resulted in increased proliferation and higher percentages of CD4+ T cells secreting IFNγ and/or TNFα. To address the role of aberrant DC maturation in the IID-mediated suppression of T cell responses, CMV-stimulated DCs were infected with the panel of viruses and co-cultured with autologous T-lymphocytes. Infection with EBOV/VP35m infection resulted in a significant increase, as compared to wt EBOV, in proliferating CD4+ cells secreting IFNγ, TNFα and IL-2. Experiments with expanded CMV-specific T cells demonstrated their increased activation following co-cultivation with CMV-pulsed DCs pre-infected with EBOV/VP24m, EBOV/VP35m and EBOV/VP35m/VP24m, as compared to wt EBOV. Both IIDs were found to block phosphorylation of TCR complex-associated adaptors and downstream signaling molecules. Next, we examined the effects of IIDs on the function of B cells in infected PBMC. Infection with EBOV/VP35m and EBOV/VP35m/VP24m resulted in significant increases in the percentages of phenotypically distinct B-cell subsets and plasma cells, as compared to wt EBOV, suggesting inhibition of B cell function and differentiation by VP35 IID. Finally, infection with EBOV/VP35m increased activation of NK cells, as compared to wt EBOV. These results demonstrate a global suppression of cell-mediated responses by EBOV IIDs and identify the role of DCs in suppression of T-cell responses.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Different Temporal Effects of Ebola Virus VP35 and VP24 Proteins on Global Gene Expression in Human Dendritic Cells.J Virol. 2015 Aug;89(15):7567-83. doi: 10.1128/JVI.00924-15. Epub 2015 May 13. J Virol. 2015. PMID: 25972536 Free PMC article.
-
Transcriptional Analysis of Lymphoid Tissues from Infected Nonhuman Primates Reveals the Basis for Attenuation and Immunogenicity of an Ebola Virus Encoding a Mutant VP35 Protein.J Virol. 2021 Feb 24;95(6):e01995-20. doi: 10.1128/JVI.01995-20. Print 2021 Feb 24. J Virol. 2021. PMID: 33408171 Free PMC article.
-
A VP35 Mutant Ebola Virus Lacks Virulence but Can Elicit Protective Immunity to Wild-Type Virus Challenge.Cell Rep. 2019 Sep 17;28(12):3032-3046.e6. doi: 10.1016/j.celrep.2019.08.047. Cell Rep. 2019. PMID: 31533029 Free PMC article.
-
Insights into Ebola Virus VP35 and VP24 Interferon Inhibitory Functions and their Initial Exploitation as Drug Targets.Infect Disord Drug Targets. 2019;19(4):362-374. doi: 10.2174/1871526519666181123145540. Infect Disord Drug Targets. 2019. PMID: 30468131 Review.
-
Evasion of interferon responses by Ebola and Marburg viruses.J Interferon Cytokine Res. 2009 Sep;29(9):511-20. doi: 10.1089/jir.2009.0076. J Interferon Cytokine Res. 2009. PMID: 19694547 Free PMC article. Review.
Cited by
-
Antibody-Dependent Enhancement of Ebola Virus Infection by Human Antibodies Isolated from Survivors.Cell Rep. 2018 Aug 14;24(7):1802-1815.e5. doi: 10.1016/j.celrep.2018.07.035. Cell Rep. 2018. PMID: 30110637 Free PMC article.
-
Pathogenicity and Virulence of Ebolaviruses with Species- and Variant-specificity.Virulence. 2021 Dec;12(1):885-901. doi: 10.1080/21505594.2021.1898169. Virulence. 2021. PMID: 33734027 Free PMC article.
-
Role of Type I Interferons on Filovirus Pathogenesis.Vaccines (Basel). 2019 Feb 20;7(1):22. doi: 10.3390/vaccines7010022. Vaccines (Basel). 2019. PMID: 30791589 Free PMC article. Review.
-
Impact of Měnglà Virus Proteins on Human and Bat Innate Immune Pathways.J Virol. 2020 Jun 16;94(13):e00191-20. doi: 10.1128/JVI.00191-20. Print 2020 Jun 16. J Virol. 2020. PMID: 32295912 Free PMC article.
-
Ebola virus protein VP40 stimulates IL-12- and IL-18-dependent activation of human natural killer cells.JCI Insight. 2022 Aug 22;7(16):e158902. doi: 10.1172/jci.insight.158902. JCI Insight. 2022. PMID: 35862204 Free PMC article.
References
-
- CDC. 2014 Ebola Outbreak in West Africa—Case Counts 2016. http://www.cdc.gov/vhf/ebola/outbreaks/2014-west-africa/case-counts.html.
-
- Feldmann H, Sanchez A, Geisbert TW. Filoviridae: Marburg and Ebola Viruses. 6 ed Knipe DM, Howley PM, editors. Philadelphia: Lippincott Williams & Wilkins; 2013. 923–56 p.
-
- Geisbert TW, Hensley LE, Gibb TR, Steele KE, Jaax NK, Jahrling PB. Apoptosis induced in vitro and in vivo during infection by Ebola and Marburg viruses. Lab Invest. 2000;80(2):171–86. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

