A "Trojan horse" bispecific-antibody strategy for broad protection against ebolaviruses
- PMID: 27608667
- PMCID: PMC5647781
- DOI: 10.1126/science.aag3267
A "Trojan horse" bispecific-antibody strategy for broad protection against ebolaviruses
Abstract
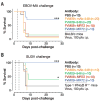
There is an urgent need for monoclonal antibody (mAb) therapies that broadly protect against Ebola virus and other filoviruses. The conserved, essential interaction between the filovirus glycoprotein, GP, and its entry receptor Niemann-Pick C1 (NPC1) provides an attractive target for such mAbs but is shielded by multiple mechanisms, including physical sequestration in late endosomes. Here, we describe a bispecific-antibody strategy to target this interaction, in which mAbs specific for NPC1 or the GP receptor-binding site are coupled to a mAb against a conserved, surface-exposed GP epitope. Bispecific antibodies, but not parent mAbs, neutralized all known ebolaviruses by coopting viral particles themselves for endosomal delivery and conferred postexposure protection against multiple ebolaviruses in mice. Such "Trojan horse" bispecific antibodies have potential as broad antifilovirus immunotherapeutics.
Copyright © 2016, American Association for the Advancement of Science.
Figures



Comment in
-
Hitting Ebola, to the power of two.Science. 2016 Oct 21;354(6310):284-285. doi: 10.1126/science.aaj2036. Science. 2016. PMID: 27846516 No abstract available.
Similar articles
-
Two Distinct Lysosomal Targeting Strategies Afford Trojan Horse Antibodies With Pan-Filovirus Activity.Front Immunol. 2021 Oct 14;12:729851. doi: 10.3389/fimmu.2021.729851. eCollection 2021. Front Immunol. 2021. PMID: 34721393 Free PMC article.
-
Host-Primed Ebola Virus GP Exposes a Hydrophobic NPC1 Receptor-Binding Pocket, Revealing a Target for Broadly Neutralizing Antibodies.mBio. 2016 Feb 23;7(1):e02154-15. doi: 10.1128/mBio.02154-15. mBio. 2016. PMID: 26908579 Free PMC article.
-
Macaque Monoclonal Antibodies Targeting Novel Conserved Epitopes within Filovirus Glycoprotein.J Virol. 2015 Oct 14;90(1):279-91. doi: 10.1128/JVI.02172-15. Print 2016 Jan 1. J Virol. 2015. PMID: 26468532 Free PMC article.
-
Achieving cross-reactivity with pan-ebolavirus antibodies.Curr Opin Virol. 2019 Feb;34:140-148. doi: 10.1016/j.coviro.2019.01.003. Epub 2019 Mar 15. Curr Opin Virol. 2019. PMID: 30884329 Free PMC article. Review.
-
Structural basis for differential neutralization of ebolaviruses.Viruses. 2012 Apr;4(4):447-70. doi: 10.3390/v4040447. Epub 2012 Apr 5. Viruses. 2012. PMID: 22590681 Free PMC article. Review.
Cited by
-
Generation of robust bispecific antibodies through fusion of single-domain antibodies on IgG scaffolds: a comprehensive comparison of formats.MAbs. 2023 Jan-Dec;15(1):2189432. doi: 10.1080/19420862.2023.2189432. MAbs. 2023. PMID: 36939220 Free PMC article.
-
A Pan-Ebolavirus Monoclonal Antibody Cocktail Provides Protection Against Ebola and Sudan Viruses.J Infect Dis. 2023 Nov 13;228(Suppl 7):S691-S700. doi: 10.1093/infdis/jiad205. J Infect Dis. 2023. PMID: 37288609 Free PMC article.
-
Fabs-in-tandem immunoglobulin is a novel and versatile bispecific design for engaging multiple therapeutic targets.MAbs. 2017 Oct;9(7):1118-1128. doi: 10.1080/19420862.2017.1345401. Epub 2017 Jul 10. MAbs. 2017. PMID: 28692328 Free PMC article.
-
Multimechanistic Monoclonal Antibodies (MAbs) Targeting Staphylococcus aureus Alpha-Toxin and Clumping Factor A: Activity and Efficacy Comparisons of a MAb Combination and an Engineered Bispecific Antibody Approach.Antimicrob Agents Chemother. 2017 Jul 25;61(8):e00629-17. doi: 10.1128/AAC.00629-17. Print 2017 Aug. Antimicrob Agents Chemother. 2017. PMID: 28584141 Free PMC article.
-
Virus inoculation and treatment regimens for evaluating anti-filovirus monoclonal antibody efficacy in vivo.Biosaf Health. 2019 Jun;1(1):6-13. doi: 10.1016/j.bsheal.2019.02.004. Epub 2019 Mar 1. Biosaf Health. 2019. PMID: 32835206 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

