NET formation can occur independently of RIPK3 and MLKL signaling
- PMID: 26549703
- PMCID: PMC4738457
- DOI: 10.1002/eji.201545615
NET formation can occur independently of RIPK3 and MLKL signaling
Abstract
The importance of neutrophil extracellular traps (NETs) in innate immunity is well established but the molecular mechanisms responsible for their formation are still a matter of scientific dispute. Here, we aim to characterize a possible role of the receptor-interacting protein kinase 3 (RIPK3) and the mixed lineage kinase domain-like (MLKL) signaling pathway, which are known to cause necroptosis, in NET formation. Using genetic and pharmacological approaches, we investigated whether this programmed form of necrosis is a prerequisite for NET formation. NETs have been defined as extracellular DNA scaffolds associated with the neutrophil granule protein elastase that are capable of killing bacteria. Neither Ripk3-deficient mouse neutrophils nor human neutrophils in which MLKL had been pharmacologically inactivated, exhibited abnormalities in NET formation upon physiological activation or exposure to low concentrations of PMA. These data indicate that NET formation occurs independently of both RIPK3 and MLKL signaling.
Keywords: MLKL; NET formation; NETosis; Necroptosis; Neutrophils; RIPK.
© 2015 The Authors. European Journal of Immunology published by WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures
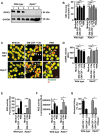
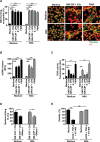
Comment in
-
Challenges in the characterization of neutrophil extracellular traps: The truth is in the details.Eur J Immunol. 2016 Jan;46(1):52-5. doi: 10.1002/eji.201546022. Eur J Immunol. 2016. PMID: 26635275
Similar articles
-
PMA and crystal-induced neutrophil extracellular trap formation involves RIPK1-RIPK3-MLKL signaling.Eur J Immunol. 2016 Jan;46(1):223-9. doi: 10.1002/eji.201545605. Epub 2015 Nov 30. Eur J Immunol. 2016. PMID: 26531064
-
Challenges in the characterization of neutrophil extracellular traps: The truth is in the details.Eur J Immunol. 2016 Jan;46(1):52-5. doi: 10.1002/eji.201546022. Eur J Immunol. 2016. PMID: 26635275
-
Receptor-Interacting Protein Kinases 1 and 3, and Mixed Lineage Kinase Domain-Like Protein Are Activated by Sublytic Complement and Participate in Complement-Dependent Cytotoxicity.Front Immunol. 2018 Feb 23;9:306. doi: 10.3389/fimmu.2018.00306. eCollection 2018. Front Immunol. 2018. PMID: 29527209 Free PMC article.
-
The diverse role of RIP kinases in necroptosis and inflammation.Nat Immunol. 2015 Jul;16(7):689-97. doi: 10.1038/ni.3206. Nat Immunol. 2015. PMID: 26086143 Review.
-
Necroptosis and Inflammation.Annu Rev Biochem. 2016 Jun 2;85:743-63. doi: 10.1146/annurev-biochem-060815-014830. Epub 2016 Feb 8. Annu Rev Biochem. 2016. PMID: 26865533 Review.
Cited by
-
Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018.Cell Death Differ. 2018 Mar;25(3):486-541. doi: 10.1038/s41418-017-0012-4. Epub 2018 Jan 23. Cell Death Differ. 2018. PMID: 29362479 Free PMC article. Review.
-
Formation of neutrophil extracellular traps in mitochondrial DNA-deficient cells.J Clin Biochem Nutr. 2020 Jan;66(1):15-23. doi: 10.3164/jcbn.19-77. Epub 2019 Nov 12. J Clin Biochem Nutr. 2020. PMID: 32001952 Free PMC article.
-
Dysregulation of neutrophil death in sepsis.Front Immunol. 2022 Aug 18;13:963955. doi: 10.3389/fimmu.2022.963955. eCollection 2022. Front Immunol. 2022. PMID: 36059483 Free PMC article. Review.
-
Klebsiella pneumoniae infection of murine neutrophils impairs their efferocytic clearance by modulating cell death machinery.PLoS Pathog. 2018 Oct 1;14(10):e1007338. doi: 10.1371/journal.ppat.1007338. eCollection 2018 Oct. PLoS Pathog. 2018. PMID: 30273394 Free PMC article.
-
Respiratory Syncytial Virus induces the classical ROS-dependent NETosis through PAD-4 and necroptosis pathways activation.Sci Rep. 2018 Sep 21;8(1):14166. doi: 10.1038/s41598-018-32576-y. Sci Rep. 2018. PMID: 30242250 Free PMC article.
References
-
- Simon, D. , Simon, H. U. and Yousefi, S. , Extracellular DNA traps in allergic, infectious, and autoimmune diseases. Allergy 2013. 68: 409–416. - PubMed
-
- Brinkmann, V. , Reichard, U. , Goosmann, C. , Fauler, B. , Uhlemann, Y. , Weiss, D. S. , Weinrauch, Y. et al, Neutrophil extracellular traps kill bacteria. Science 2004. 303: 1532–1535. - PubMed
-
- Urban, C. F. , Reichard, U. , Brinkmann, V. and Zychlinsky, A. , Neutrophil extracellular traps capture and kill Candida albicans yeast and hyphal forms. Cell. Microbiol. 2006. 8: 668–676. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

