Transmitting pain and itch messages: a contemporary view of the spinal cord circuits that generate gate control
- PMID: 24811377
- PMCID: PMC4492533
- DOI: 10.1016/j.neuron.2014.01.018
Transmitting pain and itch messages: a contemporary view of the spinal cord circuits that generate gate control
Abstract
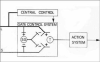
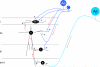
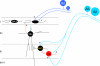
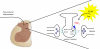
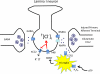
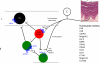
The original formulation of Gate Control Theory (GCT) proposed that the perception of pain produced by spinal cord signaling to the brain depends on a balance of activity generated in large (nonnociceptive)- and small (nociceptive)-diameter primary afferent fibers. The theory proposed that activation of the large-diameter afferent "closes" the gate by engaging a superficial dorsal horn interneuron that inhibits the firing of projection neurons. Activation of the nociceptors "opens" the gate through concomitant excitation of projection neurons and inhibition of the inhibitory interneurons. Sixty years after publication of the GCT, we are faced with an ever-growing list of morphologically and neurochemically distinct spinal cord interneurons. The present Review highlights the complexity of superficial dorsal horn circuitry and addresses the question whether the premises outlined in GCT still have relevance today. By examining the dorsal horn circuits that underlie the transmission of "pain" and "itch" messages, we also address the extent to which labeled lines can be incorporated into a contemporary view of GCT.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Spinal Circuits Transmitting Mechanical Pain and Itch.Neurosci Bull. 2018 Feb;34(1):186-193. doi: 10.1007/s12264-017-0136-z. Epub 2017 May 8. Neurosci Bull. 2018. PMID: 28484964 Free PMC article. Review.
-
Transgenic Mouse Models for the Tracing of “Pain” Pathways.In: Kruger L, Light AR, editors. Translational Pain Research: From Mouse to Man. Boca Raton (FL): CRC Press/Taylor & Francis; 2010. Chapter 7. In: Kruger L, Light AR, editors. Translational Pain Research: From Mouse to Man. Boca Raton (FL): CRC Press/Taylor & Francis; 2010. Chapter 7. PMID: 21882471 Free Books & Documents. Review.
-
Spinal presynaptic inhibition in pain control.Neuroscience. 2014 Dec 26;283:95-106. doi: 10.1016/j.neuroscience.2014.09.032. Epub 2014 Sep 22. Neuroscience. 2014. PMID: 25255936 Review.
-
Neonatal Injury Evokes Persistent Deficits in Dynorphin Inhibitory Circuits within the Adult Mouse Superficial Dorsal Horn.J Neurosci. 2020 May 13;40(20):3882-3895. doi: 10.1523/JNEUROSCI.0029-20.2020. Epub 2020 Apr 14. J Neurosci. 2020. PMID: 32291327 Free PMC article.
-
Spinal Circuits for Touch, Pain, and Itch.Annu Rev Physiol. 2018 Feb 10;80:189-217. doi: 10.1146/annurev-physiol-022516-034303. Epub 2017 Sep 27. Annu Rev Physiol. 2018. PMID: 28961064 Free PMC article. Review.
Cited by
-
Possible implications of animal models for the assessment of visceral pain.Animal Model Exp Med. 2020 Aug 10;3(3):215-228. doi: 10.1002/ame2.12130. eCollection 2020 Sep. Animal Model Exp Med. 2020. PMID: 33024943 Free PMC article. Review.
-
Neuronal GRK2 regulates microglial activation and contributes to electroacupuncture analgesia on inflammatory pain in mice.Biol Res. 2022 Feb 3;55(1):5. doi: 10.1186/s40659-022-00374-6. Biol Res. 2022. PMID: 35115050 Free PMC article.
-
Microglia in Pain: Detrimental and Protective Roles in Pathogenesis and Resolution of Pain.Neuron. 2018 Dec 19;100(6):1292-1311. doi: 10.1016/j.neuron.2018.11.009. Neuron. 2018. PMID: 30571942 Free PMC article. Review.
-
Targeted ubiquitination of sensory neuron calcium channels reduces the development of neuropathic pain.Proc Natl Acad Sci U S A. 2022 May 17;119(20):e2118129119. doi: 10.1073/pnas.2118129119. Epub 2022 May 13. Proc Natl Acad Sci U S A. 2022. PMID: 35561213 Free PMC article.
-
Peripheral and Central Mechanisms of Itch.Neuron. 2018 May 2;98(3):482-494. doi: 10.1016/j.neuron.2018.03.023. Neuron. 2018. PMID: 29723501 Free PMC article. Review.
References
-
- Ahmadi S, Lippross S, Neuhuber WL, Zeilhofer HU. PGE(2) selectively blocks inhibitory glycinergic neurotransmission onto rat superficial dorsal horn neurons. Nat Neurosci. 2002;5:34–40. - PubMed
-
- Albuquerque C, Lee CJ, Jackson AC, MacDermott AB. Subpopulations of GABAergic and non-GABAergic rat dorsal horn neurons express Ca2+-permeable AMPA receptors. Eur J Neurosci. 1999;11:2758–2766. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

