Maraba MG1 virus enhances natural killer cell function via conventional dendritic cells to reduce postoperative metastatic disease
- PMID: 24695102
- PMCID: PMC4088996
- DOI: 10.1038/mt.2014.60
Maraba MG1 virus enhances natural killer cell function via conventional dendritic cells to reduce postoperative metastatic disease
Abstract
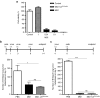
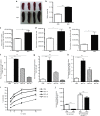
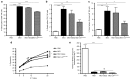
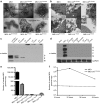
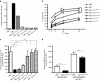
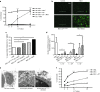
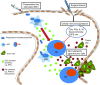
This study characterizes the ability of novel oncolytic rhabdoviruses (Maraba MG1) to boost natural killer (NK) cell activity. Our results demonstrate that MG1 activates NK cells via direct infection and maturation of conventional dendritic cells. Using NK depletion and conventional dendritic cells ablation studies in vivo, we established that both are required for MG1 efficacy. We further explored the efficacy of attenuated MG1 (nonreplicating MG1-UV(2min) and single-cycle replicating MG1-Gless) and demonstrated that these viruses activate conventional dendritic cells, although to a lesser extent than live MG1. This translates to equivalent abilities to remove tumor metastases only at the highest viral doses of attenuated MG1. In tandem, we characterized the antitumor ability of NK cells following preoperative administration of live and attenuated MG1. Our results demonstrates that a similar level of NK activation and reduction in postoperative tumor metastases was achieved with equivalent high viral doses concluding that viral replication is important, but not necessary for NK activation. Biochemical characterization of a panel of UV-inactivated MG1 (2-120 minutes) revealed that intact viral particle and target cell recognition are essential for NK cell-mediated antitumor responses. These findings provide mechanistic insight and preclinical rationale for safe perioperative virotherapy to effectively reduce metastatic disease following cancer surgery.
Figures

Similar articles
-
Combination of oncolytic Maraba virus with immune checkpoint blockade overcomes therapy resistance in an immunologically cold model of advanced melanoma with dysfunctional T-cell receptor signalling.J Immunother Cancer. 2024 Jul 25;12(7):e009443. doi: 10.1136/jitc-2024-009443. J Immunother Cancer. 2024. PMID: 39060020 Free PMC article.
-
Considerations for Clinical Translation of MG1 Maraba Virus.Methods Mol Biol. 2020;2058:285-293. doi: 10.1007/978-1-4939-9794-7_19. Methods Mol Biol. 2020. PMID: 31486046 Review.
-
Identification of genetically modified Maraba virus as an oncolytic rhabdovirus.Mol Ther. 2010 Aug;18(8):1440-9. doi: 10.1038/mt.2010.103. Epub 2010 Jun 15. Mol Ther. 2010. PMID: 20551913 Free PMC article.
-
Oncolytic Maraba Virus MG1 as a Treatment for Sarcoma.Int J Cancer. 2017 Sep 15;141(6):1257-1264. doi: 10.1002/ijc.30813. Epub 2017 Jun 21. Int J Cancer. 2017. PMID: 28568891
-
Development and applications of oncolytic Maraba virus vaccines.Oncolytic Virother. 2018 Nov 26;7:117-128. doi: 10.2147/OV.S154494. eCollection 2018. Oncolytic Virother. 2018. PMID: 30538968 Free PMC article. Review.
Cited by
-
Oncolytic Viro-Immunotherapy: An Emerging Option in the Treatment of Gliomas.Front Immunol. 2021 Oct 5;12:721830. doi: 10.3389/fimmu.2021.721830. eCollection 2021. Front Immunol. 2021. PMID: 34675919 Free PMC article. Review.
-
A Novel Chimeric Oncolytic Virus Mediates a Multifaceted Cellular Immune Response in a Syngeneic B16 Melanoma Model.Cancers (Basel). 2024 Oct 6;16(19):3405. doi: 10.3390/cancers16193405. Cancers (Basel). 2024. PMID: 39410025 Free PMC article.
-
Lipid accumulation impairs natural killer cell cytotoxicity and tumor control in the postoperative period.BMC Cancer. 2019 Aug 20;19(1):823. doi: 10.1186/s12885-019-6045-y. BMC Cancer. 2019. PMID: 31429730 Free PMC article.
-
Oncolytic virus therapy in cancer: A current review.World J Virol. 2021 Sep 25;10(5):229-255. doi: 10.5501/wjv.v10.i5.229. World J Virol. 2021. PMID: 34631474 Free PMC article. Review.
-
Attacking Postoperative Metastases using Perioperative Oncolytic Viruses and Viral Vaccines.Front Oncol. 2014 Aug 12;4:217. doi: 10.3389/fonc.2014.00217. eCollection 2014. Front Oncol. 2014. PMID: 25161958 Free PMC article. Review.
References
-
- Stojdl DF, Lichty BD, tenOever BR, Paterson JM, Power AT, Knowles S, et al. VSV strains with defects in their ability to shutdown innate immunity are potent systemic anti-cancer agents. Cancer Cell. 2003;4:263–275. - PubMed
-
- Boisgerault N, Tangy F, Gregoire M. New perspectives in cancer virotherapy: bringing the immune system into play. Immunotherapy. 2010;2:185–199. - PubMed
-
- Parato KA, Lichty BD, Bell JC. Diplomatic immunity: turning a foe into an ally. Curr Opin Mol Ther. 2009;11:13–21. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

