Adipocyte lipid chaperone AP2 is a secreted adipokine regulating hepatic glucose production
- PMID: 23663740
- PMCID: PMC3755450
- DOI: 10.1016/j.cmet.2013.04.012
Adipocyte lipid chaperone AP2 is a secreted adipokine regulating hepatic glucose production
Abstract
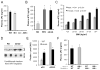
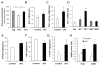
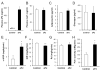
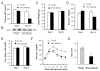
Proper control of hepatic glucose production is central to whole-body glucose homeostasis, and its disruption plays a major role in diabetes. Here, we demonstrate that although established as an intracellular lipid chaperone, aP2 is in fact actively secreted from adipocytes to control liver glucose metabolism. Secretion of aP2 from adipocytes is regulated by fasting- and lipolysis-related signals, and circulating aP2 levels are markedly elevated in mouse and human obesity. Recombinant aP2 stimulates glucose production and gluconeogenic activity in primary hepatocytes in vitro and in lean mice in vivo. In contrast, neutralization of secreted aP2 reduces glucose production and corrects the diabetic phenotype of obese mice. Hyperinsulinemic-euglycemic and pancreatic clamp studies upon aP2 administration or neutralization demonstrated actions of aP2 in liver. We conclude that aP2 is an adipokine linking adipocytes to hepatic glucose production and that neutralizing secreted aP2 may represent an effective therapeutic strategy against diabetes.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Secretion of fatty acid binding protein aP2 from adipocytes through a nonclassical pathway in response to adipocyte lipase activity.J Lipid Res. 2015 Feb;56(2):423-34. doi: 10.1194/jlr.M055798. Epub 2014 Dec 22. J Lipid Res. 2015. PMID: 25535287 Free PMC article.
-
Improved glucose and lipid metabolism in genetically obese mice lacking aP2.Endocrinology. 2000 Sep;141(9):3388-96. doi: 10.1210/endo.141.9.7637. Endocrinology. 2000. PMID: 10965911
-
Development of a therapeutic monoclonal antibody that targets secreted fatty acid-binding protein aP2 to treat type 2 diabetes.Sci Transl Med. 2015 Dec 23;7(319):319ra205. doi: 10.1126/scitranslmed.aac6336. Epub 2015 Dec 23. Sci Transl Med. 2015. PMID: 26702093
-
Adipocyte Fatty Acid-Binding Protein, Cardiovascular Diseases and Mortality.Front Immunol. 2021 Mar 19;12:589206. doi: 10.3389/fimmu.2021.589206. eCollection 2021. Front Immunol. 2021. PMID: 33815359 Free PMC article. Review.
-
Fatty Acid-Binding Protein 4 in Cardiovascular and Metabolic Diseases.J Atheroscler Thromb. 2019 Mar 1;26(3):216-232. doi: 10.5551/jat.48710. Epub 2019 Feb 7. J Atheroscler Thromb. 2019. PMID: 30726793 Free PMC article. Review.
Cited by
-
Possible Increase in Serum FABP4 Level Despite Adiposity Reduction by Canagliflozin, an SGLT2 Inhibitor.PLoS One. 2016 Apr 28;11(4):e0154482. doi: 10.1371/journal.pone.0154482. eCollection 2016. PLoS One. 2016. PMID: 27124282 Free PMC article.
-
p21-activated kinase 4 counteracts PKA-dependent lipolysis by phosphorylating FABP4 and HSL.Nat Metab. 2024 Jan;6(1):94-112. doi: 10.1038/s42255-023-00957-x. Epub 2024 Jan 12. Nat Metab. 2024. PMID: 38216738
-
Fatty Acid-Binding Protein 4 (FABP4): Pathophysiological Insights and Potent Clinical Biomarker of Metabolic and Cardiovascular Diseases.Clin Med Insights Cardiol. 2015 Feb 2;8(Suppl 3):23-33. doi: 10.4137/CMC.S17067. eCollection 2014. Clin Med Insights Cardiol. 2015. PMID: 25674026 Free PMC article. Review.
-
Plasma Levels of Fatty Acid-Binding Protein 4, Retinol-Binding Protein 4, High-Molecular-Weight Adiponectin, and Cardiovascular Mortality Among Men With Type 2 Diabetes: A 22-Year Prospective Study.Arterioscler Thromb Vasc Biol. 2016 Nov;36(11):2259-2267. doi: 10.1161/ATVBAHA.116.308320. Epub 2016 Sep 8. Arterioscler Thromb Vasc Biol. 2016. PMID: 27609367 Free PMC article.
-
Adipokines, Hepatokines and Myokines: Focus on Their Role and Molecular Mechanisms in Adipose Tissue Inflammation.Front Endocrinol (Lausanne). 2022 Jul 14;13:873699. doi: 10.3389/fendo.2022.873699. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35909571 Free PMC article. Review.
References
-
- Boden G. Effects of free fatty acids on gluconeogenesis and glycogenolysis. Life Sci. 2003;72:977–988. - PubMed
-
- Cabre A, Babio N, Lazaro I, Bullo M, Garcia-Arellano A, Masana L, Salas-Salvado J. FABP4 predicts atherogenic dyslipidemia development. The PREDIMED study. Atherosclerosis. 2012;222:229–234. - PubMed
-
- Cahill GF., Jr Fuel metabolism in starvation. Annu Rev Nutr. 2006;26:1–22. - PubMed
-
- Cao H, Maeda K, Gorgun CZ, Kim HJ, Park SY, Shulman GI, Kim JK, Hotamisligil GS. Regulation of metabolic responses by adipocyte/macrophage Fatty Acid-binding proteins in leptin-deficient mice. Diabetes. 2006;55:1915–1922. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

