RARα1 control of mammary gland ductal morphogenesis and wnt1-tumorigenesis
- PMID: 20923554
- PMCID: PMC3096972
- DOI: 10.1186/bcr2724
RARα1 control of mammary gland ductal morphogenesis and wnt1-tumorigenesis
Abstract
Introduction: Retinoic acid signaling pathways are disabled in human breast cancer suggesting a controlling role in normal mammary growth that might be lost in tumorigenesis. We tested a single receptor isotype, RARα1, for its role in mouse mammary gland morphogenesis and MMTV-wnt1-induced oncogenesis.
Methods: The role of RARα1 in mammary morphogenesis was tested in RARα1-knockout (KO) mice and in mammary tumorigenesis in bi-genic (RARα1/KO crossed with MMTV-wnt1) mice. We used whole mounts analysis, stem cells/progenitor quantification, mammary gland repopulation, Q-PCR, test of tumor-free survival, tumor fragments and cell transplantation.
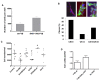
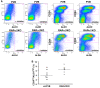
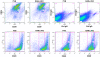
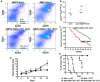
Results: In 2 genetic backgrounds (129/Bl-6 and FVB) the neo-natal RARα1/KO-mammary epithelial tree was 2-fold larger and the pubertal tree had 2-fold more branch points and 5-fold more mature end buds, a phenotype that was predominantly epithelial cell autonomous. The stem/progenitor compartment of the RARα1/KO mammary, defined as CD24(low)/ALDH(high activity) was increased by a median 1.7 fold, but the mammary stem cell (MaSC)-containing compartment, (CD24(low)/CD29(high)), was larger (~1.5 fold) in the wt-glands, and the mammary repopulating ability of the wt-gland epithelium was ~2-fold greater. In MMTV-wnt1 transgenic glands the progenitor (CD24(low)/ALDH(high activity)) content was 2.6-fold greater than in the wt and was further increased in the RARα1/KO-wnt1 glands. The tumor-free survival of RARα1/KO-wnt1 mice was significantly (p=0.0002, Kaplan Meier) longer, the in vivo growth of RARα1/KO-wnt1 transplanted tumor fragments was significantly (p=0.01) slower and RARα1/KO-wnt1 tumors cell suspension produced tumors after much longer latency.
Conclusions: In vitamin A-replete mice, RARα1 is required to maintain normal mammary morphogenesis, but paradoxically, also efficient tumorigenesis. While its loss increases the density of the mammary epithelial tree and the content of luminal mammary progenitors, it appears to reduce the size of the MaSC-containing compartment, the mammary repopulating activity, and to delay significantly the MMTV-wnt1-mammary tumorigenesis. Whether the delay in tumorigenesis is solely due to a reduction in wnt1 target cells or due to additional mechanisms remains to be determined. These results reveal the intricate nature of the retinoid signaling pathways in mammary development and carcinogenesis and suggest that a better understanding will be needed before retinoids can join the armament of effective anti- breast cancer therapies.
Figures


Similar articles
-
Autophagy regulator BECN1 suppresses mammary tumorigenesis driven by WNT1 activation and following parity.Autophagy. 2014;10(11):2036-52. doi: 10.4161/auto.34398. Epub 2014 Oct 30. Autophagy. 2014. PMID: 25483966 Free PMC article.
-
Chromatin effector Pygo2 regulates mammary tumor initiation and heterogeneity in MMTV-Wnt1 mice.Oncogene. 2014 Jan 30;33(5):632-42. doi: 10.1038/onc.2012.620. Epub 2013 Jan 21. Oncogene. 2014. PMID: 23334328 Free PMC article.
-
Cooperative signaling between Wnt1 and integrin-linked kinase induces accelerated breast tumor development.Breast Cancer Res. 2010;12(3):R38. doi: 10.1186/bcr2592. Epub 2010 Jun 21. Breast Cancer Res. 2010. PMID: 20565980 Free PMC article.
-
TGF beta regulation of cell proliferation.Princess Takamatsu Symp. 1994;24:250-63. Princess Takamatsu Symp. 1994. PMID: 8983080 Review.
-
Key signaling nodes in mammary gland development and cancer: β-catenin.Breast Cancer Res. 2010;12(6):213. doi: 10.1186/bcr2723. Epub 2010 Nov 3. Breast Cancer Res. 2010. PMID: 21067528 Free PMC article. Review.
Cited by
-
Enhanced expression of retinoic acid receptor alpha (RARA) induces epithelial-to-mesenchymal transition and disruption of mammary acinar structures.Mol Oncol. 2015 Feb;9(2):355-64. doi: 10.1016/j.molonc.2014.09.005. Epub 2014 Sep 22. Mol Oncol. 2015. PMID: 25300573 Free PMC article.
-
Left-right analysis of mammary gland development in retinoid X receptor-α+/- mice.Philos Trans R Soc Lond B Biol Sci. 2016 Dec 19;371(1710):20150416. doi: 10.1098/rstb.2015.0416. Philos Trans R Soc Lond B Biol Sci. 2016. PMID: 27821527 Free PMC article.
-
Blocking the PAH2 domain of Sin3A inhibits tumorigenesis and confers retinoid sensitivity in triple negative breast cancer.Oncotarget. 2016 Jul 12;7(28):43689-43702. doi: 10.18632/oncotarget.9905. Oncotarget. 2016. PMID: 27286261 Free PMC article.
-
Macrophages orchestrate breast cancer early dissemination and metastasis.Nat Commun. 2018 Jan 2;9(1):21. doi: 10.1038/s41467-017-02481-5. Nat Commun. 2018. PMID: 29295986 Free PMC article.
-
Cellular and micro-environmental responses influencing the antitumor activity of all-trans retinoic acid in breast cancer.Cell Commun Signal. 2024 Feb 15;22(1):127. doi: 10.1186/s12964-024-01492-2. Cell Commun Signal. 2024. PMID: 38360674 Free PMC article. Review.
References
-
- Fitzgerald P, Teng M, Chandraratna RA, Heyman RA, Allegretto EA. Retinoic acid receptor alpha expression correlates with retinoid-induced growth inhibition of human breast cancer cells regardless of estrogen receptor status. Cancer Res. 1997;57:2642–2650. - PubMed
-
- Toma S, Isnardi L, Raffo P, Dastoli G, De Francisci E, Riccardi L, Palumbo R, Bollag W. Effects of all-trans-retinoic acid and 13-cis-retinoic acid on breast-cancer cell lines: growth inhibition and apoptosis induction. Int J Cancer. 1997;70:619–627. doi: 10.1002/(SICI)1097-0215(19970304)70:5<619::AID-IJC21>3.0.CO;2-6. - DOI - PubMed
-
- Fontana JA, Hobbs PD, Dawson MI. Inhibition of mammary carcinoma growth by retinoidal benzoic acid derivatives. Exp Cell Biol. 1988;56:254–263. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

