Galectin-9/TIM-3 interaction regulates virus-specific primary and memory CD8 T cell response
- PMID: 20463811
- PMCID: PMC2865527
- DOI: 10.1371/journal.ppat.1000882
Galectin-9/TIM-3 interaction regulates virus-specific primary and memory CD8 T cell response
Abstract
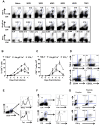
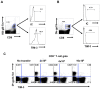
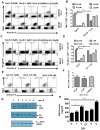
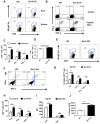
In this communication, we demonstrate that galectin (Gal)-9 acts to constrain CD8(+) T cell immunity to Herpes Simplex Virus (HSV) infection. In support of this, we show that animals unable to produce Gal-9, because of gene knockout, develop acute and memory responses to HSV that are of greater magnitude and better quality than those that occur in normal infected animals. Interestingly, infusion of normal infected mice with alpha-lactose, the sugar that binds to the carbohydrate-binding domain of Gal-9 limiting its engagement of T cell immunoglobulin and mucin (TIM-3) receptors, also caused a more elevated and higher quality CD8(+) T cell response to HSV particularly in the acute phase. Such sugar treated infected mice also had expanded populations of effector as well as memory CD8(+) T cells. The increased effector T cell responses led to significantly more efficient virus control. The mechanisms responsible for the outcome of the Gal-9/TIM-3 interaction in normal infected mice involved direct inhibitory effects on TIM-3(+) CD8(+) T effector cells as well as the promotion of Foxp3(+) regulatory T cell activity. Our results indicate that manipulating galectin signals, as can be achieved using appropriate sugars, may represent a convenient and inexpensive approach to enhance acute and memory responses to a virus infection.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

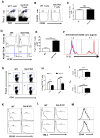

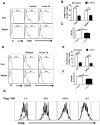
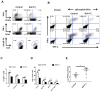
Similar articles
-
Influence of galectin-9/Tim-3 interaction on herpes simplex virus-1 latency.J Immunol. 2011 Dec 1;187(11):5745-55. doi: 10.4049/jimmunol.1102105. Epub 2011 Oct 21. J Immunol. 2011. PMID: 22021615 Free PMC article.
-
T cell immunoglobulin and mucin protein-3 (Tim-3)/Galectin-9 interaction regulates influenza A virus-specific humoral and CD8 T-cell responses.Proc Natl Acad Sci U S A. 2011 Nov 22;108(47):19001-6. doi: 10.1073/pnas.1107087108. Epub 2011 Nov 3. Proc Natl Acad Sci U S A. 2011. PMID: 22052881 Free PMC article.
-
Contrasting acute graft-versus-host disease effects of Tim-3/galectin-9 pathway blockade dependent upon the presence of donor regulatory T cells.Blood. 2012 Jul 19;120(3):682-90. doi: 10.1182/blood-2011-10-387977. Epub 2012 Jun 7. Blood. 2012. PMID: 22677125 Free PMC article.
-
The bitter side of sweet: the role of Galectin-9 in immunopathogenesis of viral infections.Rev Med Virol. 2015 May;25(3):175-86. doi: 10.1002/rmv.1832. Epub 2015 Mar 11. Rev Med Virol. 2015. PMID: 25760439 Review.
-
Emerging Tim-3 functions in antimicrobial and tumor immunity.Trends Immunol. 2011 Aug;32(8):345-9. doi: 10.1016/j.it.2011.05.003. Epub 2011 Jun 21. Trends Immunol. 2011. PMID: 21697013 Free PMC article. Review.
Cited by
-
Development and Characterization of a Nanobody against Human T-Cell Immunoglobulin and Mucin-3.Comput Math Methods Med. 2022 Jun 11;2022:2929605. doi: 10.1155/2022/2929605. eCollection 2022. Comput Math Methods Med. 2022. Retraction in: Comput Math Methods Med. 2023 Sep 27;2023:9803219. doi: 10.1155/2023/9803219 PMID: 35726228 Free PMC article. Retracted.
-
Tim-3 negatively regulates IL-12 expression by monocytes in HCV infection.PLoS One. 2011;6(5):e19664. doi: 10.1371/journal.pone.0019664. Epub 2011 May 26. PLoS One. 2011. PMID: 21637332 Free PMC article.
-
Galectin-9 is rapidly released during acute HIV-1 infection and remains sustained at high levels despite viral suppression even in elite controllers.AIDS Res Hum Retroviruses. 2014 Jul;30(7):654-64. doi: 10.1089/AID.2014.0004. Epub 2014 May 28. AIDS Res Hum Retroviruses. 2014. PMID: 24786365 Free PMC article.
-
Present and future perspectives on immunotherapy for advanced renal cell carcinoma: Going to the core or beating around the bush?J Kidney Cancer VHL. 2015 Apr 4;2(2):55-63. doi: 10.15586/jkcvhl.2015.24. eCollection 2015. J Kidney Cancer VHL. 2015. PMID: 28326259 Free PMC article.
-
IL-4 impairs wound healing potential in the skin by repressing fibronectin expression.J Allergy Clin Immunol. 2017 Jan;139(1):142-151.e5. doi: 10.1016/j.jaci.2016.07.012. Epub 2016 Aug 20. J Allergy Clin Immunol. 2017. PMID: 27554818 Free PMC article.
References
-
- Zinkernagel RM. Immunology taught by viruses. Science. 1996;271:173–178. - PubMed
-
- Ahmed R, Gray D. Immunological memory and protective immunity: understanding their relation. Science. 1996;272:54–60. - PubMed
-
- Gandhi RT, Walker BD. Immunologic control of HIV-1. Annu Rev Med. 2002;53:149–172. - PubMed
-
- Rabinovich GA, Toscano MA. Turning ‘sweet’ on immunity: galectin-glycan interactions in immune tolerance and inflammation. Nat Rev Immunol. 2009;9:338–352. - PubMed
-
- Mercier S, St-Pierre C, Pelletier I, Ouellet M, Tremblay MJ, et al. Galectin-1 promotes HIV-1 infectivity in macrophages through stabilization of viral adsorption. Virology. 2008;371:121–129. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

