Monoclonal antibodies for cancer immunotherapy
- PMID: 19304016
- PMCID: PMC2677705
- DOI: 10.1016/S0140-6736(09)60251-8
Monoclonal antibodies for cancer immunotherapy
Abstract
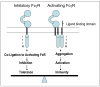
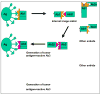
Monoclonal antibodies are effective treatments for many malignant diseases. However, the ability of antibodies to initiate tumour-antigen-specific immune responses has received less attention than have other mechanisms of antibody action. We describe the rationale and evidence for the development of antibodies that can stimulate host tumour-antigen-specific immune responses. Such responses can be induced through the induction of antibody-dependent cellular cytotoxicity, promotion of antibody-targeted cross-presentation of tumour antigens, or by triggering of the idiotypic network. Future treatment modifications or combinations might be able to prolong, amplify, and shape these immune responses to increase the clinical benefits of antibody therapy for human cancer.
Figures


Similar articles
-
Using antibodies in tumour immunotherapy.Expert Opin Biol Ther. 2004 Aug;4(8):1265-84. doi: 10.1517/14712598.4.8.1265. Expert Opin Biol Ther. 2004. PMID: 15268661 Review.
-
Tumor associated antigen (TAA) mimicry and immunotherapy of malignant diseases from anti-idiotypic antibodies to peptide mimics.Cancer Chemother Biol Response Modif. 2001;19:309-26. Cancer Chemother Biol Response Modif. 2001. PMID: 11686020 Review. No abstract available.
-
The anti-idiotype vaccines for immunotherapy.Curr Opin Mol Ther. 2001 Feb;3(1):63-9. Curr Opin Mol Ther. 2001. PMID: 11249733 Review.
-
NGcGM3 ganglioside: a privileged target for cancer vaccines.Clin Dev Immunol. 2010;2010:814397. doi: 10.1155/2010/814397. Epub 2010 Oct 27. Clin Dev Immunol. 2010. PMID: 21048926 Free PMC article. Review.
-
Biological therapy: approaches in colorectal cancer. Strategies to enhance carcinoembryonic antigen (CEA) as an immunogenic target.Br J Cancer. 1998 Mar;77(5):683-93. doi: 10.1038/bjc.1998.114. Br J Cancer. 1998. PMID: 9514045 Free PMC article. Review. No abstract available.
Cited by
-
Self-Assembly as a Molecular Strategy to Improve Immunotherapy.Acc Chem Res. 2020 Nov 17;53(11):2534-2545. doi: 10.1021/acs.accounts.0c00438. Epub 2020 Oct 19. Acc Chem Res. 2020. PMID: 33074649 Free PMC article.
-
Cripto-1 as a Potential Target of Cancer Stem Cells for Immunotherapy.Cancers (Basel). 2021 May 20;13(10):2491. doi: 10.3390/cancers13102491. Cancers (Basel). 2021. PMID: 34065315 Free PMC article. Review.
-
Tumor antigen-specific monoclonal antibodies and induction of T-cell immunity.Semin Oncol. 2014 Oct;41(5):678-84. doi: 10.1053/j.seminoncol.2014.08.003. Epub 2014 Aug 12. Semin Oncol. 2014. PMID: 25440612 Free PMC article. Review.
-
Monoclonal antibodies: From magic bullet to precision weapon.Mol Biomed. 2024 Oct 11;5(1):47. doi: 10.1186/s43556-024-00210-1. Mol Biomed. 2024. PMID: 39390211 Free PMC article. Review.
-
Advances in epidermal growth factor receptor specific immunotherapy: lessons to be learned from armed antibodies.Oncotarget. 2020 Sep 22;11(38):3531-3557. doi: 10.18632/oncotarget.27730. eCollection 2020 Sep 22. Oncotarget. 2020. PMID: 33014289 Free PMC article. Review.
References
-
- McLaughlin P, Grillo-Lopez AJ, Link BK, et al. Rituximab chimeric anti-CD20 monoclonal antibody therapy for relapsed indolent lymphoma: half of patients respond to a four-dose treatment program. J Clin Oncol. 1998;16:2825–33. - PubMed
-
- Kaminski MS, Estes J, Zasadny KR, et al. Radioimmunotherapy with iodine (131)I tositumomab for relapsed or refractory B-cell non-Hodgkin lymphoma: updated results and long-term follow-up of the University of Michigan experience. Blood. 2000;96:1259–66. - PubMed
-
- Coiffier B. Rituximab in combination with CHOP improves survival in elderly patients with aggressive non-Hodgkin’s lymphoma. Semin Oncol. 2002;29(2 Suppl 6):18–22. - PubMed
-
- Witzig TE, Gordon LI, Cabanillas F, et al. Randomized controlled trial of yttrium-90-labeled ibritumomab tiuxetan radioimmunotherapy versus rituximab immunotherapy for patients with relapsed or refractory low-grade, follicular, or transformed B-cell non-Hodgkin’s lymphoma. J Clin Oncol. 2003;20:2453–63. - PubMed
-
- Sievers EL, Appelbaum FR, Spielberger RT, et al. Selective ablation of acute myeloid leukemia using antibody-targeted chemotherapy: a phase I study of an anti-CD33 calicheamicin immunoconjugate. Blood. 1999;93:3678–84. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 CA121033/CA/NCI NIH HHS/United States
- R01 CA105500/CA/NCI NIH HHS/United States
- P30 CA051008-16/CA/NCI NIH HHS/United States
- R01 CA106802/CA/NCI NIH HHS/United States
- R01 CA109465/CA/NCI NIH HHS/United States
- R01 CA050633-16/CA/NCI NIH HHS/United States
- R01 CA050633/CA/NCI NIH HHS/United States
- R01 CA109465-03/CA/NCI NIH HHS/United States
- P30 CA051008/CA/NCI NIH HHS/United States
- R01 CA121033-02/CA/NCI NIH HHS/United States
- R01 CA106802-04/CA/NCI NIH HHS/United States
- R01 CA105500-04/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

