Calpain activity and muscle wasting in sepsis
- PMID: 18492780
- PMCID: PMC2575901
- DOI: 10.1152/ajpendo.90226.2008
Calpain activity and muscle wasting in sepsis
Abstract
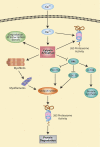
Muscle wasting in sepsis reflects activation of multiple proteolytic mechanisms, including lyosomal and ubiquitin-proteasome-dependent protein breakdown. Recent studies suggest that activation of the calpain system also plays an important role in sepsis-induced muscle wasting. Perhaps the most important consequence of calpain activation in skeletal muscle during sepsis is disruption of the sarcomere, allowing for the release of myofilaments (including actin and myosin) that are subsequently ubiquitinated and degraded by the 26S proteasome. Other important consequences of calpain activation that may contribute to muscle wasting during sepsis include degradation of certain transcription factors and nuclear cofactors, activation of the 26S proteasome, and inhibition of Akt activity, allowing for downstream activation of Foxo transcription factors and GSK-3beta. The role of calpain activation in sepsis-induced muscle wasting suggests that the calpain system may be a therapeutic target in the prevention and treatment of muscle wasting during sepsis. Furthermore, because calpain activation may also be involved in muscle wasting caused by other conditions, including different muscular dystrophies and cancer, calpain inhibitors may be beneficial not only in the treatment of sepsis-induced muscle wasting but in other conditions causing muscle atrophy as well.
Figures
Similar articles
-
Treatment of rats with calpain inhibitors prevents sepsis-induced muscle proteolysis independent of atrogin-1/MAFbx and MuRF1 expression.Am J Physiol Regul Integr Comp Physiol. 2006 Jun;290(6):R1589-97. doi: 10.1152/ajpregu.00668.2005. Epub 2006 Feb 2. Am J Physiol Regul Integr Comp Physiol. 2006. PMID: 16455766
-
Novel aspects on the regulation of muscle wasting in sepsis.Int J Biochem Cell Biol. 2005 Oct;37(10):2156-68. doi: 10.1016/j.biocel.2005.01.017. Epub 2005 Mar 13. Int J Biochem Cell Biol. 2005. PMID: 16125115 Review.
-
Muscle wasting in a rat model of long-lasting sepsis results from the activation of lysosomal, Ca2+ -activated, and ubiquitin-proteasome proteolytic pathways.J Clin Invest. 1996 Apr 1;97(7):1610-7. doi: 10.1172/JCI118586. J Clin Invest. 1996. PMID: 8601625 Free PMC article.
-
Sepsis stimulates release of myofilaments in skeletal muscle by a calcium-dependent mechanism.FASEB J. 1999 Aug;13(11):1435-43. doi: 10.1096/fasebj.13.11.1435. FASEB J. 1999. PMID: 10428767
-
Muscle cachexia: current concepts of intracellular mechanisms and molecular regulation.Ann Surg. 2001 Jan;233(1):9-17. doi: 10.1097/00000658-200101000-00003. Ann Surg. 2001. PMID: 11141219 Free PMC article. Review.
Cited by
-
Intensive care unit-acquired weakness: Recent insights.J Intensive Med. 2023 Aug 30;4(1):73-80. doi: 10.1016/j.jointm.2023.07.002. eCollection 2024 Jan. J Intensive Med. 2023. PMID: 38263973 Free PMC article. Review.
-
Activation of Calpain Contributes to Mechanical Ventilation-Induced Depression of Protein Synthesis in Diaphragm Muscle.Cells. 2022 Mar 18;11(6):1028. doi: 10.3390/cells11061028. Cells. 2022. PMID: 35326479 Free PMC article.
-
Protective Effects of Glutamine and Leucine Supplementation on Sepsis-Induced Skeletal Muscle Injuries.Int J Mol Sci. 2021 Nov 30;22(23):13003. doi: 10.3390/ijms222313003. Int J Mol Sci. 2021. PMID: 34884807 Free PMC article.
-
Out of Control: The Role of the Ubiquitin Proteasome System in Skeletal Muscle during Inflammation.Biomolecules. 2021 Sep 8;11(9):1327. doi: 10.3390/biom11091327. Biomolecules. 2021. PMID: 34572540 Free PMC article. Review.
-
Differential Protein Metabolism and Regeneration in Gastrocnemius Muscles in High-fat Diet Fed Mice and Pre-hibernation Daurian Ground Squirrels: A Comparison between Pathological and Healthy Obesity.Zool Stud. 2021 Mar 8;60:e6. doi: 10.6620/ZS.2021.60-06. eCollection 2021. Zool Stud. 2021. PMID: 34386092 Free PMC article.
References
-
- Agarkova I, Perriard JC. The M-band: an elastic web that crosslinks thick filaments in the center of the sarcomere. Trends Cell Biol 15: 477–485, 2005. - PubMed
-
- Aizawa H, Kawahara H, Tanaka K, Yokosawa H. Activation of the proteasome during Xenopus egg activation imploes a link between proteasome activation and intracellular calcium release. Biochem Biophys Res Commun 218: 224–228, 1996. - PubMed
-
- Auvin S, Pignol B, Navet E, Pons D, Marin JG, Bigg D, Chabrier PE. Novel dual inhibitors of calpain and lipid peroxidation. Bioorg Med Chem 14: 3825–3828, 2004. - PubMed
-
- Baar K, Nader G, Bodine S. Resistance exercise, muscle loading/unloading and the control of muscle mass. Essays Biochem 42: 61–74, 2006. - PubMed
-
- Baracos V, Greenberg RE, Goldberg AL. Influence of calcium and other divalent cations on protein turnover in rat skeletal muscle. Am J Physiol Endocrinol Metab 250: E702–E710, 1986. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

