Homeostasis and effector function of lymphopenia-induced "memory-like" T cells in constitutively T cell-depleted mice
- PMID: 18354198
- PMCID: PMC2670614
- DOI: 10.4049/jimmunol.180.7.4742
Homeostasis and effector function of lymphopenia-induced "memory-like" T cells in constitutively T cell-depleted mice
Abstract
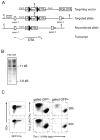
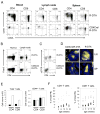
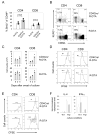
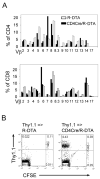
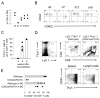
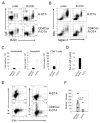
Naive T lymphocytes acquire a phenotype similar to Ag-experienced memory T cells as a result of proliferation under lymphopenic conditions. Such "memory-like" T (T(ML)) cells constitute a large fraction of the peripheral T cell pool in patients recovering from T cell ablative therapies, HIV patients under highly active antiretroviral therapy, and in the elderly population. To generate a model that allows characterization of T(ML) cells without adoptive transfer, irradiation, or thymectomy, we developed genetically modified mice that express diphtheria toxin A under control of a loxP-flanked stop cassette (R-DTA mice). Crossing these mice to CD4Cre mice resulted in efficient ablation of CD4 single-positive thymocytes, whereas double-positive and CD8 single-positive thymocytes were only partially affected. In the periphery the pool of naive (CD44(low)CD62L(high)) T cells was depleted. However, some T cells were resistant to Cre activity, escaped deletion in the thymus, and underwent lymphopenia-induced proliferation resulting in a pool of T(ML) cells that was similar in size and turnover to the pool of CD44(high)CD62L(low) "memory phenotype" T cells in control mice. CD4Cre/R-DTA mice remained lymphopenic despite the large available immunological "space" and normal Ag-induced T cell proliferation. CD4Cre/R-DTA mice showed a biased TCR repertoire indicating oligoclonal T cell expansion. Infection with the helminth Nippostrongylus brasiliensis resulted in diminished effector cell recruitment and impaired worm expulsion, demonstrating that T(ML) cells are not sufficient to mediate an effective immune response.
Figures


Similar articles
-
Memory-like CD8+ and CD4+ T cells cooperate to break peripheral tolerance under lymphopenic conditions.Proc Natl Acad Sci U S A. 2008 Dec 9;105(49):19414-9. doi: 10.1073/pnas.0807743105. Epub 2008 Nov 25. Proc Natl Acad Sci U S A. 2008. PMID: 19033460 Free PMC article.
-
Mimicking homeostatic proliferation in vitro generates T cells with high anti-tumor function in non-lymphopenic hosts.Cancer Immunol Immunother. 2013 Mar;62(3):503-15. doi: 10.1007/s00262-012-1350-1. Epub 2012 Sep 23. Cancer Immunol Immunother. 2013. PMID: 23001162 Free PMC article.
-
Differential lymphopenia-induced homeostatic proliferation for CD4+ and CD8+ T cells following septic injury.J Leukoc Biol. 2009 Mar;85(3):382-90. doi: 10.1189/jlb.0808491. Epub 2008 Dec 30. J Leukoc Biol. 2009. PMID: 19088177 Free PMC article.
-
Homeostatic proliferation and survival of naïve and memory T cells.Eur J Immunol. 2009 Aug;39(8):2088-94. doi: 10.1002/eji.200939444. Eur J Immunol. 2009. PMID: 19637200 Review.
-
Cytokine signals in T-cell homeostasis.J Immunother. 2005 Jul-Aug;28(4):289-94. doi: 10.1097/01.cji.0000165356.03924.e7. J Immunother. 2005. PMID: 16000945 Review.
Cited by
-
Oculomotor nerve guidance and terminal branching requires interactions with differentiating extraocular muscles.Dev Biol. 2021 Aug;476:272-281. doi: 10.1016/j.ydbio.2021.04.006. Epub 2021 Apr 24. Dev Biol. 2021. PMID: 33905720 Free PMC article.
-
IgE Effector Mechanisms, in Concert with Mast Cells, Contribute to Acquired Host Defense against Staphylococcusaureus.Immunity. 2020 Oct 13;53(4):793-804.e9. doi: 10.1016/j.immuni.2020.08.002. Epub 2020 Sep 9. Immunity. 2020. PMID: 32910906 Free PMC article.
-
Microglial phagocytosis of living photoreceptors contributes to inherited retinal degeneration.EMBO Mol Med. 2015 Sep;7(9):1179-97. doi: 10.15252/emmm.201505298. EMBO Mol Med. 2015. PMID: 26139610 Free PMC article.
-
Roles of basophils and mast cells in cutaneous inflammation.Semin Immunopathol. 2016 Sep;38(5):563-70. doi: 10.1007/s00281-016-0570-4. Epub 2016 May 11. Semin Immunopathol. 2016. PMID: 27170045 Review.
-
Sensory nerve niche regulates mesenchymal stem cell homeostasis via FGF/mTOR/autophagy axis.Nat Commun. 2023 Jan 20;14(1):344. doi: 10.1038/s41467-023-35977-4. Nat Commun. 2023. PMID: 36670126 Free PMC article.
References
-
- Fadel S, Sarzotti M. Cellular immune responses in neonates. Int Rev Immunol. 2000;19:173–193. - PubMed
-
- Modigliani Y, Coutinho G, Burlen-Defranoux O, Coutinho A, Bandeira A. Differential contribution of thymic outputs and peripheral expansion in the development of peripheral T cell pools. Eur J Immunol. 1994;24:1223–1227. - PubMed
-
- Schonland SO, Zimmer JK, Lopez-Benitez CM, Widmann T, Ramin KD, Goronzy JJ, Weyand CM. Homeostatic control of T-cell generation in neonates. Blood. 2003;102:1428–1434. - PubMed
-
- Steinmann GG, Klaus B, Muller-Hermelink HK. The involution of the ageing human thymic epithelium is independent of puberty. A morphometric study. Scand J Immunol. 1985;22:563–575. - PubMed
-
- Goronzy JJ, Weyand CM. T cell development and receptor diversity during aging. Curr Opin Immunol. 2005;17:468–475. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

