Melanocytes derived from patients with Hermansky-Pudlak Syndrome types 1, 2, and 3 have distinct defects in cargo trafficking
- PMID: 15675963
- PMCID: PMC1635963
- DOI: 10.1111/j.0022-202X.2004.23585.x
Melanocytes derived from patients with Hermansky-Pudlak Syndrome types 1, 2, and 3 have distinct defects in cargo trafficking
Abstract
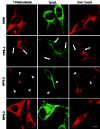
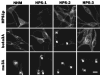
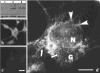
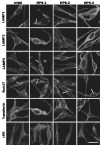
Hermansky-Pudlak Syndrome (HPS) is a genetically heterogeneous disorder in which mutations in one of several genes interrupts biogenesis of melanosomes, platelet dense bodies, and lysosomes. Affected patients have oculocutaneous albinism, a bleeding diathesis, and sometimes develop granulomatous colitis or pulmonary fibrosis. In order to assess the role of HPS genes in melanosome biogenesis, melanocytes cultured from patients with HPS subtypes 1, 2, or 3 were assessed for the localization of various melanocyte proteins. Tyrosinase, Tyrp1, and Dct/Tyrp2 were atypically and distinctly expressed in HPS-1 and HPS-3 melanocytes, whereas only tyrosinase showed an atypical distribution in HPS-2 melanocytes. The HPS1 and AP3B1 (i.e., HPS-2) gene products showed no expression in HPS-1 and HPS-2 melanocytes, respectively, whereas HPS-3 melanocytes exhibited normal expression for both proteins. In normal human melanocytes, the HPS1 protein was expressed as an approximately 80 kDa molecule with both granular and reticular intracellular profiles. In HPS-1, lysosome associated membrane protein 1 (LAMP1), and LAMP3 were localized to abnormal large granules; in HPS-2, all LAMPs exhibited a normal granular expression; and in HPS-3, LAMP1, and LAMP3 exhibited a distinct less granular and more floccular pattern. In contrast, the expressions of Rab 27, transferrin, and cKit were unaffected in all three HPS genotypes. These data demonstrate that the three initially identified subtypes of human HPS exhibit distinct defects in the trafficking of various melanocyte-specific proteins.
Figures

Similar articles
-
Improper trafficking of melanocyte-specific proteins in Hermansky-Pudlak syndrome type-5.J Invest Dermatol. 2007 Jun;127(6):1471-8. doi: 10.1038/sj.jid.5700737. Epub 2007 Feb 15. J Invest Dermatol. 2007. PMID: 17301833 Free PMC article.
-
AP-3 mediates tyrosinase but not TRP-1 trafficking in human melanocytes.Mol Biol Cell. 2001 Jul;12(7):2075-85. doi: 10.1091/mbc.12.7.2075. Mol Biol Cell. 2001. PMID: 11452004 Free PMC article.
-
Membranous complexes characteristic of melanocytes derived from patients with Hermansky-Pudlak syndrome type 1 are macroautophagosomal entities of the lysosomal compartment.Pigment Cell Res. 2005 Dec;18(6):417-26. doi: 10.1111/j.1600-0749.2005.00265.x. Pigment Cell Res. 2005. PMID: 16280007 Free PMC article.
-
Hermansky-Pudlak syndrome: a disease of protein trafficking and organelle function.Pigment Cell Res. 2006 Feb;19(1):19-42. doi: 10.1111/j.1600-0749.2005.00289.x. Pigment Cell Res. 2006. PMID: 16420244 Review.
-
The molecular machinery for the biogenesis of lysosome-related organelles: lessons from Hermansky-Pudlak syndrome.Semin Cell Dev Biol. 2002 Aug;13(4):271-8. doi: 10.1016/s1084952102000563. Semin Cell Dev Biol. 2002. PMID: 12243726 Review.
Cited by
-
BLOC-2 targets recycling endosomal tubules to melanosomes for cargo delivery.J Cell Biol. 2015 May 25;209(4):563-77. doi: 10.1083/jcb.201410026. J Cell Biol. 2015. PMID: 26008744 Free PMC article.
-
ELANE mutations in cyclic and severe congenital neutropenia: genetics and pathophysiology.Hematol Oncol Clin North Am. 2013 Feb;27(1):19-41, vii. doi: 10.1016/j.hoc.2012.10.004. Epub 2012 Nov 7. Hematol Oncol Clin North Am. 2013. PMID: 23351986 Free PMC article. Review.
-
Genetic modifiers of abnormal organelle biogenesis in a Drosophila model of BLOC-1 deficiency.Hum Mol Genet. 2010 Mar 1;19(5):861-78. doi: 10.1093/hmg/ddp555. Epub 2009 Dec 16. Hum Mol Genet. 2010. PMID: 20015953 Free PMC article.
-
Melanosomes--dark organelles enlighten endosomal membrane transport.Nat Rev Mol Cell Biol. 2007 Oct;8(10):786-97. doi: 10.1038/nrm2258. Nat Rev Mol Cell Biol. 2007. PMID: 17878918 Free PMC article. Review.
-
AP-3 complex subunit delta gene, ap3d1, regulates melanogenesis and melanophore survival via autophagy in zebrafish (Danio rerio).Pigment Cell Melanoma Res. 2022 Sep;35(5):495-505. doi: 10.1111/pcmr.13055. Pigment Cell Melanoma Res. 2022. PMID: 35816398 Free PMC article.
References
-
- Anderson PD, Huizing M, Claassen DA, White J, Gahl WA. Hermansky–Pudlak syndrome type 4 (HPS-4): Clinical and molecular characteristics. Hum Genet. 2003;113:10–17. - PubMed
-
- Anikster Y, Huizing M, White J, Bale S, Gahl WA, Toro J. Mutation of a new gene causes a unique form of Hermansky–Pudlak syndrome in genetic isolate of central Puerto Rico. Nat Genet. 2001;28:376–380. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

