Akt mediates cytoprotection of endothelial cells by vascular endothelial growth factor in an anchorage-dependent manner
- PMID: 10347193
- PMCID: PMC3624707
- DOI: 10.1074/jbc.274.23.16349
Akt mediates cytoprotection of endothelial cells by vascular endothelial growth factor in an anchorage-dependent manner
Abstract
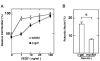
Regulation of endothelial cell apoptosis is a critical modulator of normal and pathological angiogenesis. In this study, we examined the role of the protein kinase Akt/PKB in endothelial cell survival in response to growth factor and matrix attachment signals. Vascular endothelial growth factor(VEGF)-induced cytoprotection of endothelial cell monolayers correlated with the wortmannin-sensitive induction of Akt activity. Transfection of an adenovirus expressing a dominant-negative Akt mutant decreased endothelial cell viability in the presence of VEGF. Conversely, adenoviral transduction of wild-type Akt facilitated the cell survival effects of VEGF, whereas transduction of constitutively active Akt conferred endothelial cell survival in the absence of VEGF. Constitutively active Akt also conferred survival to endothelial cells in suspension culture, whereas stimulation with VEGF did not. In suspension cultures, VEGF stimulation was unable to activate Akt, and Akt protein levels were repressed in cells undergoing anoikis. These data suggest that cross-talk between growth factor- and anchorage-dependent signaling pathways are essential for Akt activation and endothelial cell survival.
Figures

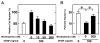
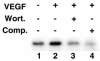





Similar articles
-
Akt down-regulation of p38 signaling provides a novel mechanism of vascular endothelial growth factor-mediated cytoprotection in endothelial cells.J Biol Chem. 2001 Aug 10;276(32):30359-65. doi: 10.1074/jbc.M009698200. Epub 2001 May 31. J Biol Chem. 2001. PMID: 11387313
-
Vascular endothelial growth factor induces protein kinase C (PKC)-dependent Akt/PKB activation and phosphatidylinositol 3'-kinase-mediates PKC delta phosphorylation: role of PKC in angiogenesis.Cell Biol Int. 2002;26(9):751-9. doi: 10.1016/s1065-6995(02)90926-1. Cell Biol Int. 2002. PMID: 12377207
-
Vascular endothelial growth factor regulates endothelial cell survival through the phosphatidylinositol 3'-kinase/Akt signal transduction pathway. Requirement for Flk-1/KDR activation.J Biol Chem. 1998 Nov 13;273(46):30336-43. doi: 10.1074/jbc.273.46.30336. J Biol Chem. 1998. PMID: 9804796
-
Signaling transduction mechanisms mediating biological actions of the vascular endothelial growth factor family.Cardiovasc Res. 2001 Feb 16;49(3):568-81. doi: 10.1016/s0008-6363(00)00268-6. Cardiovasc Res. 2001. PMID: 11166270 Review.
-
[Effects of statins on angiogenesis and vasculogenesis].Rev Esp Cardiol. 2002 Aug;55(8):838-44. doi: 10.1016/s0300-8932(02)76713-4. Rev Esp Cardiol. 2002. PMID: 12199980 Review. Spanish.
Cited by
-
ZO-1 interacts with YB-1 in endothelial cells to regulate stress granule formation during angiogenesis.Nat Commun. 2024 May 23;15(1):4405. doi: 10.1038/s41467-024-48852-7. Nat Commun. 2024. PMID: 38782923 Free PMC article.
-
Human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) alleviate excessive autophagy of ovarian granular cells through VEGFA/PI3K/AKT/mTOR pathway in premature ovarian failure rat model.J Ovarian Res. 2023 Sep 30;16(1):198. doi: 10.1186/s13048-023-01278-z. J Ovarian Res. 2023. PMID: 37777781 Free PMC article.
-
CYD0281, a Bcl-2 BH4 domain antagonist, inhibits tumor angiogenesis and breast cancer tumor growth.BMC Cancer. 2023 May 26;23(1):479. doi: 10.1186/s12885-023-10974-4. BMC Cancer. 2023. PMID: 37237269 Free PMC article.
-
Mitochondrial Hydrogen Peroxide Activates PTEN and Inactivates Akt Leading to Autophagy Inhibition-Dependent Cell Death in Neuronal Models of Parkinson's Disease.Mol Neurobiol. 2023 Jun;60(6):3345-3364. doi: 10.1007/s12035-023-03286-y. Epub 2023 Feb 28. Mol Neurobiol. 2023. PMID: 36853430 Free PMC article.
-
Extracellular Vesicles from Human Umbilical Cord-Derived MSCs Affect Vessel Formation In Vitro and Promote VEGFR2-Mediated Cell Survival.Cells. 2022 Nov 24;11(23):3750. doi: 10.3390/cells11233750. Cells. 2022. PMID: 36497011 Free PMC article.
References
-
- Risau W. Nature. 1997;386:671–674. - PubMed
-
- de Vries C, Escobedo JA, Ueno H, Houck KA, Ferrara N, Williams LT. Science. 1992;255:989–991. - PubMed
-
- Millauer B, Wizigmann-Voos S, Schnurch H, Martinez R, Moller NPH, Risau W, Ulrich A. Cell. 1993;72:835–846. - PubMed
-
- Carmeliet P, Ferreira V, Breier G, Pollefeyt S, Kieckens L, Gertsenstein M, Fahrig M, Vandenhoeck A, Harpal K, Eberhardt C, Declercq C, Pawling J, Moons L, Collen D, Risau W, Nagy A. Nature. 1996;380:435–439. - PubMed
-
- Ferrara N, Carver-Moore K, Chen H, Dowd M, Lu L, O’Shea K, Powell-Braxton L, Hillan KJ, Moore MW. Nature. 1996;380:439–442. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

