An arms race between 5'ppp-RNA virus and its alternative recognition receptor MDA5 in RIG-I-lost teleost fish
- PMID: 39347580
- PMCID: PMC11441976
- DOI: 10.7554/eLife.94898
An arms race between 5'ppp-RNA virus and its alternative recognition receptor MDA5 in RIG-I-lost teleost fish
Abstract
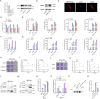
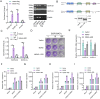
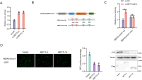
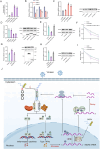
The incessant arms race between viruses and hosts has led to numerous evolutionary innovations that shape life's evolution. During this process, the interactions between viral receptors and viruses have garnered significant interest since viral receptors are cell surface proteins exploited by viruses to initiate infection. Our study sheds light on the arms race between the MDA5 receptor and 5'ppp-RNA virus in a lower vertebrate fish, Miichthys miiuy. Firstly, the frequent and independent loss events of RIG-I in vertebrates prompted us to search for alternative immune substitutes, with homology-dependent genetic compensation response (HDGCR) being the main pathway. Our further analysis suggested that MDA5 of M. miiuy and Gallus gallus, the homolog of RIG-I, can replace RIG-I in recognizing 5'ppp-RNA virus, which may lead to redundancy of RIG-I and loss from the species genome during evolution. Secondly, as an adversarial strategy, 5'ppp-RNA SCRV can utilize the m6A methylation mechanism to degrade MDA5 and weaken its antiviral immune ability, thus promoting its own replication and immune evasion. In summary, our study provides a snapshot into the interaction and coevolution between vertebrate and virus, offering valuable perspectives on the ecological and evolutionary factors that contribute to the diversity of the immune system.
Keywords: 5'ppp-RNA; MDA5; RIG-I; chicken; evolutionary biology; genetic compensation response; immune evasion; immunology; inflammation; pattern recognition receptors.
Plain language summary
Before the immune system can eliminate a bacterium, virus or other type of pathogen, it needs to be able to recognize these foreign elements. To achieve this, cells in the immune system have proteins called pattern recognition receptors (PRRs) which can identify distinct molecular features of certain pathogens. One specific group of PRRs is a family of retinoic acid-induced RIG-I-like receptors (RLRs), which help immune cells detect different types of viruses. Members of this family recognize distinct motifs on the genetic material of viruses known as RNA. For instance, RIG-I recognizes a marker known as 5’ppp on the end of single-stranded RNA molecules, whereas MDA5 recognizes long strands of double-stranded RNA. Many vertebrates – including various mammals, birds, and fish – lost the RIG-I receptor over the course of evolution. However, Geng et al. predicted that some animals lacking the RIG-I receptor may still be able to activate an immune response against viruses that contain the 5’ppp-RNA motif. To investigate this possibility, Geng et al. studied chickens and miiuy croakers (a type of ray-finned fish) which no longer have a RIG-I receptor. They found that both animals can still sense and eliminate two 5’ppp-RNA viruses called VSV and SCRV. Further experiments revealed that these two viruses are detected by a modified MDA5 receptor that had evolved to bind to 5’-ppp and activate the antiviral response. Viruses are also continuously evolving new ways to escape the immune system. This led Geng et al. to investigate whether SCRV, which causes serious harm to marine fish, has evolved a way to evade the MDA5 protection mechanism. Using miiuy croakers as a model, they found that SCRV causes the transcripts that produce the MDA5 protein to contain more molecules of m6a. This molecular tag degrades the transcript, leading to lower levels of MDA5, reducing the antiviral response against SCRV. The findings of Geng et al. offer valuable perspectives on how the immune system adapts over the course of evolution, and highlight the diversity of antiviral responses in vertebrates. Chickens and miiuy croakers are commonly farmed animals, and further work investigating how viruses invade these species could prevent illnesses from spreading and having a negative impact on the economy.
© 2024, Geng et al.
Conflict of interest statement
SG, XL, WZ, TX No competing interests declared
Figures




Update of
- doi: 10.1101/2024.01.10.575132
- doi: 10.7554/eLife.94898.1
- doi: 10.7554/eLife.94898.2
- doi: 10.7554/eLife.94898.3
Similar articles
-
Comparative Structure and Function Analysis of the RIG-I-Like Receptors: RIG-I and MDA5.Front Immunol. 2019 Jul 17;10:1586. doi: 10.3389/fimmu.2019.01586. eCollection 2019. Front Immunol. 2019. PMID: 31379819 Free PMC article. Review.
-
5'PPP-RNA induced RIG-I activation inhibits drug-resistant avian H5N1 as well as 1918 and 2009 pandemic influenza virus replication.Virol J. 2010 May 21;7:102. doi: 10.1186/1743-422X-7-102. Virol J. 2010. PMID: 20492658 Free PMC article.
-
Structural Analysis of dsRNA Binding to Anti-viral Pattern Recognition Receptors LGP2 and MDA5.Mol Cell. 2016 May 19;62(4):586-602. doi: 10.1016/j.molcel.2016.04.021. Mol Cell. 2016. PMID: 27203181 Free PMC article.
-
The anticancer functions of RIG-I-like receptors, RIG-I and MDA5, and their applications in cancer therapy.Transl Res. 2017 Dec;190:51-60. doi: 10.1016/j.trsl.2017.08.004. Epub 2017 Aug 31. Transl Res. 2017. PMID: 28917654 Review.
-
Solution structures of cytosolic RNA sensor MDA5 and LGP2 C-terminal domains: identification of the RNA recognition loop in RIG-I-like receptors.J Biol Chem. 2009 Jun 26;284(26):17465-74. doi: 10.1074/jbc.M109.007179. Epub 2009 Apr 20. J Biol Chem. 2009. PMID: 19380577 Free PMC article.
Cited by
-
How the Structure of Signaling Regulation Evolves: Insights from an Evolutionary Model.bioRxiv [Preprint]. 2024 Oct 25:2024.10.23.619883. doi: 10.1101/2024.10.23.619883. bioRxiv. 2024. PMID: 39484560 Free PMC article. Preprint.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

