SARS-CoV-2 Viral Load in the Nasopharynx at Time of First Infection Among Unvaccinated Individuals: A Secondary Cross-Protocol Analysis of 4 Randomized Trials
- PMID: 38780941
- PMCID: PMC11117088
- DOI: 10.1001/jamanetworkopen.2024.12835
SARS-CoV-2 Viral Load in the Nasopharynx at Time of First Infection Among Unvaccinated Individuals: A Secondary Cross-Protocol Analysis of 4 Randomized Trials
Abstract
Importance: SARS-CoV-2 viral load (VL) in the nasopharynx is difficult to quantify and standardize across settings, but it may inform transmission potential and disease severity.
Objective: To characterize VL at COVID-19 diagnosis among previously uninfected and unvaccinated individuals by evaluating the association of demographic and clinical characteristics, viral variant, and trial with VL, as well as the ability of VL to predict severe disease.
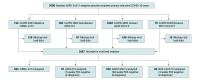
Design, setting, and participants: This secondary cross-protocol analysis used individual-level data from placebo recipients from 4 harmonized, phase 3 COVID-19 vaccine efficacy trials sponsored by Moderna, AstraZeneca, Janssen, and Novavax. Participants were SARS-CoV-2 negative at baseline and acquired COVID-19 during the blinded phase of the trials. The setting included the US, Brazil, South Africa, Colombia, Argentina, Peru, Chile, and Mexico; start dates were July 27, 2020, to December 27, 2020; data cutoff dates were March 26, 2021, to July 30, 2021. Statistical analysis was performed from November 2022 to June 2023.
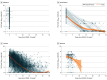
Main outcomes and measures: Linear regression was used to assess the association of demographic and clinical characteristics, viral variant, and trial with polymerase chain reaction-measured log10 VL in nasal and/or nasopharyngeal swabs taken at the time of COVID-19 diagnosis.
Results: Among 1667 participants studied (886 [53.1%] male; 995 [59.7%] enrolled in the US; mean [SD] age, 46.7 [14.7] years; 204 [12.2%] aged 65 years or older; 196 [11.8%] American Indian or Alaska Native, 150 [9%] Black or African American, 1112 [66.7%] White; 762 [45.7%] Hispanic or Latino), median (IQR) log10 VL at diagnosis was 6.18 (4.66-7.12) log10 copies/mL. Participant characteristics and viral variant explained only 5.9% of the variability in VL. The independent factor with the highest observed differences was trial: Janssen participants had 0.54 log10 copies/mL lower mean VL vs Moderna participants (95% CI, 0.20 to 0.87 log10 copies/mL lower). In the Janssen study, which captured the largest number of COVID-19 events and variants and used the most intensive post-COVID surveillance, neither VL at diagnosis nor averaged over days 1 to 28 post diagnosis was associated with COVID-19 severity.
Conclusions and relevance: In this study of placebo recipients from 4 randomized phase 3 trials, high variability was observed in SARS-CoV-2 VL at the time of COVID-19 diagnosis, and only a fraction was explained by individual participant characteristics or viral variant. These results suggest challenges for future studies of interventions seeking to influence VL and elevates the importance of standardized methods for specimen collection and viral load quantitation.
Conflict of interest statement
Figures


Similar articles
-
The effect of sample site and collection procedure on identification of SARS-CoV-2 infection.Cochrane Database Syst Rev. 2024 Dec 16;12(12):CD014780. doi: 10.1002/14651858.CD014780. Cochrane Database Syst Rev. 2024. PMID: 39679851 Free PMC article. Review.
-
RETRACTED: Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial.Int J Antimicrob Agents. 2020 Jul;56(1):105949. doi: 10.1016/j.ijantimicag.2020.105949. Epub 2020 Mar 20. Int J Antimicrob Agents. 2020. Retraction in: Int J Antimicrob Agents. 2025 Jan;65(1):107416. doi: 10.1016/j.ijantimicag.2024.107416. PMID: 32205204 Free PMC article. Retracted. Clinical Trial.
-
Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials.Lancet. 2021 Mar 6;397(10277):881-891. doi: 10.1016/S0140-6736(21)00432-3. Epub 2021 Feb 19. Lancet. 2021. PMID: 33617777 Free PMC article.
-
Antibody tests for identification of current and past infection with SARS-CoV-2.Cochrane Database Syst Rev. 2022 Nov 17;11(11):CD013652. doi: 10.1002/14651858.CD013652.pub2. Cochrane Database Syst Rev. 2022. PMID: 36394900 Free PMC article. Review.
-
Nirmatrelvir combined with ritonavir for preventing and treating COVID-19.Cochrane Database Syst Rev. 2023 Nov 30;11(11):CD015395. doi: 10.1002/14651858.CD015395.pub3. Cochrane Database Syst Rev. 2023. PMID: 38032024 Free PMC article. Review.
Cited by
-
Personal characteristics and transmission dynamics associated with SARS-CoV-2 semi-quantitative PCR test results: an observational study from Belgium, 2021-2022.Front Public Health. 2024 Sep 10;12:1429021. doi: 10.3389/fpubh.2024.1429021. eCollection 2024. Front Public Health. 2024. PMID: 39319296 Free PMC article.
References
-
- World Health Organization . WHO Coronavirus (COVID-19) Dashboard. Accessed March 6, 2024. https://covid19.who.int/
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
- UM1 AI069423/AI/NIAID NIH HHS/United States
- U01 AI069496/AI/NIAID NIH HHS/United States
- UM1 AI069476/AI/NIAID NIH HHS/United States
- UM1 AI148576/AI/NIAID NIH HHS/United States
- U01 AI069470/AI/NIAID NIH HHS/United States
- UM1 AI069534/AI/NIAID NIH HHS/United States
- UM1 AI069470/AI/NIAID NIH HHS/United States
- UM1 AI068614/AI/NIAID NIH HHS/United States
- UM1 AI068618/AI/NIAID NIH HHS/United States
- UM1 AI069496/AI/NIAID NIH HHS/United States
- UM1 AI068635/AI/NIAID NIH HHS/United States
- UM1 AI068617/AI/NIAID NIH HHS/United States
- UM1 AI148689/AI/NIAID NIH HHS/United States
- UM1 AI148450/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

