Kif21a deficiency leads to impaired glomerular filtration barrier function
- PMID: 37932480
- PMCID: PMC10628293
- DOI: 10.1038/s41598-023-46270-1
Kif21a deficiency leads to impaired glomerular filtration barrier function
Abstract
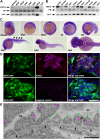
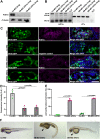
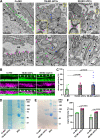
The renal glomerulus represents the major filtration body of the vertebrate nephron and is responsible for urine production and a number of other functions such as metabolic waste elimination and the regulation of water, electrolyte and acid-base balance. Podocytes are highly specialized epithelial cells that form a crucial part of the glomerular filtration barrier (GFB) by establishing a slit diaphragm for semipermeable plasma ultrafiltration. Defects of the GFB lead to proteinuria and impaired kidney function often resulting in end-stage renal failure. Although significant knowledge has been acquired in recent years, many aspects in podocyte biology are still incompletely understood. By using zebrafish as a vertebrate in vivo model, we report a novel role of the Kinesin-like motor protein Kif21a in glomerular filtration. Our studies demonstrate specific Kif21a localization to the podocytes. Its deficiency resulted in altered podocyte morphology leading to podocyte foot process effacement and altered slit diaphragm formation. Finally, we proved considerable functional consequences of Kif21a deficiency by demonstrating a leaky GFB resulting in severe proteinuria. Conclusively, our data identified a novel role of Kif21a for proper GFB function and adds another piece to the understanding of podocyte architecture and regulation.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Glomerular Endothelial Cell-Derived microRNA-192 Regulates Nephronectin Expression in Idiopathic Membranous Glomerulonephritis.J Am Soc Nephrol. 2021 Nov;32(11):2777-2794. doi: 10.1681/ASN.2020121699. J Am Soc Nephrol. 2021. PMID: 34716242 Free PMC article.
-
Knockdown of Tmem234 in zebrafish results in proteinuria.Am J Physiol Renal Physiol. 2015 Dec 1;309(11):F955-66. doi: 10.1152/ajprenal.00525.2014. Epub 2015 Sep 16. Am J Physiol Renal Physiol. 2015. PMID: 26377798
-
A reverse genetic screen in the zebrafish identifies crb2b as a regulator of the glomerular filtration barrier.Dev Biol. 2009 Oct 1;334(1):1-9. doi: 10.1016/j.ydbio.2009.04.017. Epub 2009 Apr 23. Dev Biol. 2009. PMID: 19393641
-
Role of the podocyte in proteinuria.Pediatr Nephrol. 2011 Oct;26(10):1775-80. doi: 10.1007/s00467-010-1725-5. Epub 2010 Dec 24. Pediatr Nephrol. 2011. PMID: 21184239 Free PMC article. Review.
-
Podocyte endocytosis in the regulation of the glomerular filtration barrier.Am J Physiol Renal Physiol. 2015 Sep 1;309(5):F398-405. doi: 10.1152/ajprenal.00136.2015. Epub 2015 Jun 17. Am J Physiol Renal Physiol. 2015. PMID: 26084928 Free PMC article. Review.
Cited by
-
β-Sitosterol Mitigates Apoptosis, Oxidative Stress and Inflammatory Response by Inactivating TLR4/NF-кB Pathway in Cell Models of Diabetic Nephropathy.Cell Biochem Biophys. 2024 Oct 19. doi: 10.1007/s12013-024-01559-4. Online ahead of print. Cell Biochem Biophys. 2024. PMID: 39424766
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

