Evaluation of post-vaccination immunoglobulin G antibodies and T-cell immune response after inoculation with different types and doses of SARS-CoV-2 vaccines: A retrospective cohort study
- PMID: 36703898
- PMCID: PMC9871809
- DOI: 10.3389/fmed.2022.1092646
Evaluation of post-vaccination immunoglobulin G antibodies and T-cell immune response after inoculation with different types and doses of SARS-CoV-2 vaccines: A retrospective cohort study
Abstract
Introduction: The induction and speed of production of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) immune biomarkers may vary by type and number of inoculated vaccine doses. This study aimed to explore variations in SARS-CoV-2 anti-spike (anti-S), anti-nucleocapsid (anti-N), and neutralizing immunoglobulin G (IgG) antibodies, and T-cell response by type and number of SARS-CoV-2 vaccine doses received.
Methods: In a naturally exposed and SARS-CoV-2-vaccinated population, we quantified the anti-S, anti-N, and neutralizing IgG antibody concentration and assessed T-cell response. Data on socio-demographics, medical history, and history of SARS-CoV-2 infection and vaccination were collected. Furthermore, nasal swabs were collected to test for SARS-CoV-2 infection. Confounder-adjusted association between having equal or more than a median concentration of the three IgG antibodies and T-cell response by number and type of the inoculated vaccines was quantified.
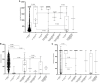
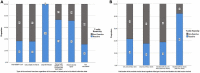
Results: We surveyed 952 male participants with a mean age of 35.5 years ± 8.4 standard deviations. Of them, 52.6% were overweight/obese, and 11.7% had at least one chronic comorbidity. Of the participants, 1.4, 0.9, 20.2, 75.2, and 2.2% were never vaccinated, primed with only one dose, primed with two doses, boosted with only one dose, and boosted with two doses, respectively. All were polymerase chain reaction-negative to SARS-CoV-2. BBIBP-CorV (Sinopharm) was the most commonly used vaccine (92.1%), followed by rAd26-S + rAd5-S (Sputnik V Gam-COVID-Vac) (1.5%) and BNT162b2 (Pfizer-BioNTech) (0.3%). Seropositivity to anti-S, anti-N, and neutralizing IgG antibodies was detected in 99.7, 99.9, and 99.3% of the study participants, respectively. The T-cell response was detected in 38.2% of 925 study participants. Every additional vaccine dose was significantly associated with increased odds of having ≥median concentration of anti-S [adjusted odds ratio (aOR), 1.34; 95% confidence interval (CI): 1.02-1.76], anti-N (aOR, 1.35; 95% CI: 1.03-1.75), neutralizing IgG antibodies (aOR, 1.29; 95% CI: 1.00-1.66), and a T-cell response (aOR, 1.48; 95% CI: 1.12-1.95). Compared with boosting with only one dose, boosting with two doses was significantly associated with increased odds of having ≥median concentration of anti-S (aOR, 13.8; 95% CI: 1.78-106.5), neutralizing IgG antibodies (aOR, 13.2; 95% CI: 1.71-101.9), and T-cell response (aOR, 7.22; 95% CI: 1.99-26.5) although not with anti-N (aOR, 0.41; 95% CI: 0.16-1.08). Compared with priming and subsequently boosting with BBIBP-CorV, all participants who were primed with BBIBP-CorV and subsequently boosted with BNT162b2 had ≥median concentration of anti-S and neutralizing IgG antibodies and 14.6-time increased odds of having a T-cell response (aOR, 14.63; 95% CI: 1.78-120.5). Compared with priming with two doses, boosting with the third dose was not associated, whereas boosting with two doses was significantly associated with having ≥median concentration of anti-S (aOR, 14.20; 95% CI: 1.85-109.4), neutralizing IgG (aOR, 13.6; 95% CI: 1.77-104.3), and T-cell response (aOR, 7.62; 95% CI: 2.09-27.8).
Conclusion: Achieving and maintaining a high blood concentration of protective immune biomarkers that predict vaccine effectiveness is very critical to limit transmission and contain outbreaks. In this study, boosting with only one dose or with only BBIBP-CorV after priming with BBIBP-CorV was insufficient, whereas boosting with two doses, particularly boosting with the mRNA-based vaccine, was shown to be associated with having a high concentration of anti-S, anti-N, and neutralizing IgG antibodies and producing an efficient T-cell response.
Keywords: COVID-19; SARS-CoV-2; coronavirus; vaccination; vaccine.
Copyright © 2023 Al-Rifai, Alhosani, Abuyadek, Atef, Donnelly, Leinberger-Jabari, Ahmed, Altrabulsi, Alatoom, Alsuwaidi and AbdelWareth.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Comparative immunogenicity and safety of Gam-COVID-Vac and Sinopharm BBIBP-CorV vaccines: results of a pilot clinical study.Heliyon. 2023 Nov 7;9(11):e21877. doi: 10.1016/j.heliyon.2023.e21877. eCollection 2023 Nov. Heliyon. 2023. PMID: 38027828 Free PMC article.
-
Comparison of immune activation of the COVID vaccines: ChAdOx1, BNT162b2, mRNA-1273, BBIBP-CorV, and Gam-COVID-Vac from serological human samples in Hungary showed higher protection after mRNA-based immunization.Eur Rev Med Pharmacol Sci. 2022 Jul;26(14):5297-5306. doi: 10.26355/eurrev_202207_29321. Eur Rev Med Pharmacol Sci. 2022. PMID: 35916830
-
Immunogenicity, safety, and reactogenicity of a half- versus full-dose BNT162b2 (Pfizer-BioNTech) booster following a two-dose ChAdOx1 nCoV-19, BBIBP-CorV, or Gam-COVID-Vac priming schedule in Mongolia: a randomised, controlled, non-inferiority trial.Lancet Reg Health West Pac. 2023 Nov 21;42:100953. doi: 10.1016/j.lanwpc.2023.100953. eCollection 2024 Jan. Lancet Reg Health West Pac. 2023. PMID: 38357398 Free PMC article.
-
Immunity after COVID-19 vaccination in people with higher risk of compromised immune status: a scoping review.Cochrane Database Syst Rev. 2022 Aug 9;8(8):CD015021. doi: 10.1002/14651858.CD015021. Cochrane Database Syst Rev. 2022. PMID: 35943061 Free PMC article. Review.
-
The Efficacy of COVID-19 Vaccines in Chronic Kidney Disease and Kidney Transplantation Patients: A Narrative Review.Vaccines (Basel). 2021 Aug 10;9(8):885. doi: 10.3390/vaccines9080885. Vaccines (Basel). 2021. PMID: 34452010 Free PMC article. Review.
Cited by
-
Natural infection versus hybrid (natural and vaccination) humoral immune response to SARS-CoV-2: a comparative paired analysis.Front Immunol. 2023 Sep 1;14:1230974. doi: 10.3389/fimmu.2023.1230974. eCollection 2023. Front Immunol. 2023. PMID: 37720219 Free PMC article.
-
Anti-S and Anti-N Antibody Responses of COVID-19 Vaccine Recipients.Vaccines (Basel). 2023 Aug 22;11(9):1398. doi: 10.3390/vaccines11091398. Vaccines (Basel). 2023. PMID: 37766076 Free PMC article.
-
Longitudinal Follow-Up of the Immunity to SARS-CoV-2 in Health Care Workers in Argentina: Persistence of Humoral Response and Neutralizing Capacity after Sputnik V Vaccination.mSphere. 2023 Jun 22;8(3):e0066222. doi: 10.1128/msphere.00662-22. Epub 2023 Apr 18. mSphere. 2023. PMID: 37070983 Free PMC article.
-
A longitudinal analysis of humoral, T cellular response and influencing factors in a cohort of healthcare workers: Implications for personalized SARS-CoV-2 vaccination strategies.Front Immunol. 2023 Mar 14;14:1130802. doi: 10.3389/fimmu.2023.1130802. eCollection 2023. Front Immunol. 2023. PMID: 36999012 Free PMC article.
-
Genomic characteristics, disease outcome and heterologous vaccine effectiveness among cases with SARS CoV-2 infection.BMC Infect Dis. 2024 Nov 8;24(1):1266. doi: 10.1186/s12879-024-10124-6. BMC Infect Dis. 2024. PMID: 39516737 Free PMC article.
References
-
- Worldometer. COVID-19 coronavirus pandemic. Available online at: https://www.worldometers.info/coronavirus/ (Accessed December 3, 2022).
-
- Gavi VaccinesWork. COVID-19 vaccines have saved 20 million lives so far, study estimates. (2022). Available online at: https://www.gavi.org/vaccineswork/covid-19-vaccines-have-saved-20-millio... (Accessed December 4, 2022).
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

