Lipoprotein Lipase: Is It a Magic Target for the Treatment of Hypertriglyceridemia
- PMID: 36065644
- PMCID: PMC9449100
- DOI: 10.3803/EnM.2022.402
Lipoprotein Lipase: Is It a Magic Target for the Treatment of Hypertriglyceridemia
Abstract
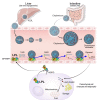
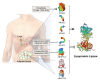
High levels of triglycerides (TG) and triglyceride-rich lipoproteins (TGRLs) confer a residual risk of cardiovascular disease after optimal low-density lipoprotein cholesterol (LDL-C)-lowering therapy. Consensus has been made that LDL-C is a non-arguable primary target for lipid lowering treatment, but the optimization of TGRL for reducing the remnant risk of cardiovascular diseases is urged. Omega-3 fatty acids and fibrates are used to reduce TG levels, but many patients still have high TG and TGRL levels combined with low high-density lipoprotein concentration that need to be ideally treated. Lipoprotein lipase (LPL) is a key regulator for TGs that hydrolyzes TGs to glycerol and free fatty acids in lipoprotein particles for lipid storage and consumption in peripheral organs. A deeper understanding of human genetics has enabled the identification of proteins regulating the LPL activity, which include the apolipoproteins and angiopoietin-like families. Novel therapeutic approach such as antisense oligonucleotides and monoclonal antibodies that regulate TGs have been developed in recent decades. In this article, we focus on the biology of LPL and its modulators and review recent clinical application, including genetic studies and clinical trials of novel therapeutics. Optimization of LPL activity to lower TG levels could eventually reduce incident atherosclerotic cardiovascular disease in conjunction with successful LDL-C reduction.
Keywords: Angiopoietin-like protein 3; Apolipoproteins; Cardiovascular diseases; Cholesterol; Lipoprotein lipase; Oligonucleotides, antisense; Triglycerides.
Conflict of interest statement
No potential conflict of interest relevant to this article was reported.
Figures
Similar articles
-
Identification of a small molecule that stabilizes lipoprotein lipase in vitro and lowers triglycerides in vivo.Biochem Biophys Res Commun. 2014 Jul 25;450(2):1063-9. doi: 10.1016/j.bbrc.2014.06.114. Epub 2014 Jun 28. Biochem Biophys Res Commun. 2014. PMID: 24984153
-
The Forgotten Lipids: Triglycerides, Remnant Cholesterol, and Atherosclerotic Cardiovascular Disease Risk.Endocr Rev. 2019 Apr 1;40(2):537-557. doi: 10.1210/er.2018-00184. Endocr Rev. 2019. PMID: 30312399 Free PMC article. Review.
-
Long-chain omega-3 fatty acids, fibrates and niacin as therapeutic options in the treatment of hypertriglyceridemia: a review of the literature.Atherosclerosis. 2015 Oct;242(2):647-56. doi: 10.1016/j.atherosclerosis.2015.06.012. Epub 2015 Jun 11. Atherosclerosis. 2015. PMID: 26296750 Review.
-
2017 George Lyman Duff Memorial Lecture: Fat in the Blood, Fat in the Artery, Fat in the Heart: Triglyceride in Physiology and Disease.Arterioscler Thromb Vasc Biol. 2018 Apr;38(4):700-706. doi: 10.1161/ATVBAHA.117.309666. Epub 2018 Feb 1. Arterioscler Thromb Vasc Biol. 2018. PMID: 29419410 Free PMC article. Review.
-
Omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and their mechanisms of action on apolipoprotein B-containing lipoproteins in humans: a review.Lipids Health Dis. 2017 Aug 10;16(1):149. doi: 10.1186/s12944-017-0541-3. Lipids Health Dis. 2017. PMID: 28797250 Free PMC article. Review.
Cited by
-
Lipoprotein lipase as a target for obesity/diabetes related cardiovascular disease.J Pharm Pharm Sci. 2024 Jul 16;27:13199. doi: 10.3389/jpps.2024.13199. eCollection 2024. J Pharm Pharm Sci. 2024. PMID: 39081272 Free PMC article. Review.
-
Dyslipidemia in Patients with Chronic Kidney Disease: An Updated Overview.Diabetes Metab J. 2023 Sep;47(5):612-629. doi: 10.4093/dmj.2023.0067. Epub 2023 Jul 24. Diabetes Metab J. 2023. PMID: 37482655 Free PMC article. Review.
-
Peroxisome Proliferator-Activated Receptor α in Lipoprotein Metabolism and Atherosclerotic Cardiovascular Disease.Biomedicines. 2023 Oct 3;11(10):2696. doi: 10.3390/biomedicines11102696. Biomedicines. 2023. PMID: 37893070 Free PMC article. Review.
-
Sugar and Dyslipidemia: A Double-Hit, Perfect Storm.J Clin Med. 2023 Aug 31;12(17):5660. doi: 10.3390/jcm12175660. J Clin Med. 2023. PMID: 37685728 Free PMC article. Review.
-
Angiopoietin-like Proteins and Lipoprotein Lipase: The Waltz Partners That Govern Triglyceride-Rich Lipoprotein Metabolism? Impact on Atherogenesis, Dietary Interventions, and Emerging Therapies.J Clin Med. 2024 Sep 4;13(17):5229. doi: 10.3390/jcm13175229. J Clin Med. 2024. PMID: 39274442 Free PMC article.
References
-
- Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: executive summary: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J Am Coll Cardiol. 2019;73:3168–209. - PubMed
-
- Santamarina-Fojo S. The familial chylomicronemia syndrome. Endocrinol Metab Clin North Am. 1998;27:551–67. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

