Single-cell resolution of MET- and EMT-like programs in osteoblasts during zebrafish fin regeneration
- PMID: 35169687
- PMCID: PMC8829776
- DOI: 10.1016/j.isci.2022.103784
Single-cell resolution of MET- and EMT-like programs in osteoblasts during zebrafish fin regeneration
Abstract
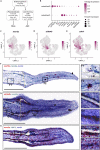
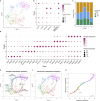
Zebrafish regenerate fin rays following amputation through epimorphic regeneration, a process that has been proposed to involve the epithelial-to-mesenchymal transition (EMT). We performed single-cell RNA sequencing (scRNA-seq) to elucidate osteoblastic transcriptional programs during zebrafish caudal fin regeneration. We show that osteoprogenitors are enriched with components associated with EMT and its reverse, mesenchymal-to-epithelial transition (MET), and provide evidence that the EMT markers cdh11 and twist2 are co-expressed in dedifferentiating cells at the amputation stump at 1 dpa, and in differentiating osteoblastic cells in the regenerate, the latter of which are enriched in EMT signatures. We also show that esrp1, a regulator of alternative splicing in epithelial cells that is associated with MET, is expressed in a subset of osteoprogenitors during outgrowth. This study provides a single cell resource for the study of osteoblastic cells during zebrafish fin regeneration, and supports the contribution of MET- and EMT-associated components to this process.
Keywords: Biological sciences; Developmental biology; Omics; Transcriptomics.
© 2022 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Suppressive effects of valproic acid on caudal fin regeneration in adult zebrafish.Anim Cells Syst (Seoul). 2020 Dec 24;24(6):349-358. doi: 10.1080/19768354.2020.1860126. Anim Cells Syst (Seoul). 2020. PMID: 33456719 Free PMC article.
-
Notch regulates blastema proliferation and prevents differentiation during adult zebrafish fin regeneration.Development. 2013 Apr;140(7):1402-11. doi: 10.1242/dev.087346. Epub 2013 Jan 23. Development. 2013. PMID: 23344707
-
In vivo electroporation of morpholinos into the regenerating adult zebrafish tail fin.J Vis Exp. 2012 Mar 29;(61):3632. doi: 10.3791/3632. J Vis Exp. 2012. PMID: 22491016 Free PMC article.
-
Molecular signaling networks that choreograph epimorphic fin regeneration in zebrafish - a mini-review.Gerontology. 2010;56(2):231-40. doi: 10.1159/000259327. Epub 2009 Nov 18. Gerontology. 2010. PMID: 19923791 Free PMC article. Review.
-
Zebrafish (Danio rerio) as a Model for Understanding the Process of Caudal Fin Regeneration.Zebrafish. 2020 Dec;17(6):359-372. doi: 10.1089/zeb.2020.1926. Epub 2020 Dec 1. Zebrafish. 2020. PMID: 33259770 Review.
Cited by
-
CUT&Tag applied to zebrafish adult tail fins reveals a return of embryonic H3K4me3 patterns during regeneration.Epigenetics Chromatin. 2024 Jul 20;17(1):22. doi: 10.1186/s13072-024-00547-5. Epigenetics Chromatin. 2024. PMID: 39033118 Free PMC article.
-
En1 and Lmx1b do not recapitulate embryonic dorsal-ventral limb patterning functions during mouse digit tip regeneration.Cell Rep. 2022 Nov 22;41(8):111701. doi: 10.1016/j.celrep.2022.111701. Cell Rep. 2022. PMID: 36417876 Free PMC article.
-
Insulin-like growth factor receptor / mTOR signaling elevates global translation to accelerate zebrafish fin regenerative outgrowth.Dev Biol. 2023 Oct;502:1-13. doi: 10.1016/j.ydbio.2023.05.008. Epub 2023 Jun 7. Dev Biol. 2023. PMID: 37290497 Free PMC article.
-
Genetically engineered zebrafish as models of skeletal development and regeneration.Bone. 2023 Feb;167:116611. doi: 10.1016/j.bone.2022.116611. Epub 2022 Nov 14. Bone. 2023. PMID: 36395960 Free PMC article. Review.
-
Loss of cped1 does not affect bone and lean tissue in zebrafish.bioRxiv [Preprint]. 2024 Aug 17:2024.07.10.601974. doi: 10.1101/2024.07.10.601974. bioRxiv. 2024. Update in: JBMR Plus. 2024 Dec 10;9(2):ziae159. doi: 10.1093/jbmrpl/ziae159. PMID: 39026892 Free PMC article. Updated. Preprint.
References
-
- Albano G., Dolder S., Siegrist M., Mercier-Zuber A., Auberson M., Stoudmann C., Hofstetter W., Bonny O., Fuster D.G. Increased bone resorption by osteoclast-specific deletion of the sodium/calcium exchanger isoform 1 (NCX1) Pflugers Arch. 2017;469:225–233. doi: 10.1007/s00424-016-1923-5. - DOI - PubMed
-
- Anbalagan S., Gordon L., Blechman J., Matsuoka R.L., Rajamannar P., Wircer E., Biran J., Reuveny A., Leshkowitz D., Stainier D.Y.R., Levkowitz G. Pituicyte cues regulate the development of permeable neuro-vascular interfaces. Dev.Cell. 2018;47:711–726.e715. doi: 10.1016/j.devcel.2018.10.017. - DOI - PubMed
-
- Ansieau S., Bastid J., Doreau A., Morel A.P., Bouchet B.P., Thomas C., Fauvet F., Puisieux I., Doglioni C., Piccinin S., et al. Induction of EMT by twist proteins as a collateral effect of tumor-promoting inactivation of premature senescence. Cancer Cell. 2008;14:79–89. doi: 10.1016/j.ccr.2008.06.005. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

