Diversity and robustness of bone morphogenetic protein pattern formation
- PMID: 33795238
- PMCID: PMC8034876
- DOI: 10.1242/dev.192344
Diversity and robustness of bone morphogenetic protein pattern formation
Abstract
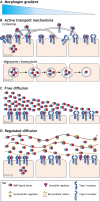
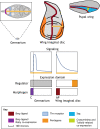
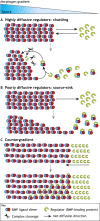
Pattern formation by bone morphogenetic proteins (BMPs) demonstrates remarkable plasticity and utility in several contexts, such as early embryonic development, tissue patterning and the maintenance of stem cell niches. BMPs pattern tissues over many temporal and spatial scales: BMP gradients as short as 1-2 cell diameters maintain the stem cell niche of the Drosophila germarium over a 24-h cycle, and BMP gradients of several hundred microns establish dorsal-ventral tissue specification in Drosophila, zebrafish and Xenopus embryos in timescales between 30 min and several hours. The mechanisms that shape BMP signaling gradients are also incredibly diverse. Although ligand diffusion plays a dominant role in forming the gradient, a cast of diffusible and non-diffusible regulators modulate gradient formation and confer robustness, including scale invariance and adaptability to perturbations in gene expression and growth. In this Review, we document the diverse ways that BMP gradients are formed and refined, and we identify the core principles that they share to achieve reliable performance.
Keywords: Axis formation; BMP patterning; Drosophila; Morphogen gradient; Zebrafish.
© 2021. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures
Similar articles
-
Extracellular modulation of BMP activity in patterning the dorsoventral axis.Birth Defects Res C Embryo Today. 2006 Sep;78(3):224-42. doi: 10.1002/bdrc.20079. Birth Defects Res C Embryo Today. 2006. PMID: 17061292 Review.
-
Computational analysis of BMP gradients in dorsal-ventral patterning of the zebrafish embryo.J Theor Biol. 2007 Oct 21;248(4):579-89. doi: 10.1016/j.jtbi.2007.05.026. Epub 2007 Jun 6. J Theor Biol. 2007. PMID: 17673236 Free PMC article.
-
Scale invariance of BMP signaling gradients in zebrafish.Sci Rep. 2019 Apr 1;9(1):5440. doi: 10.1038/s41598-019-41840-8. Sci Rep. 2019. PMID: 30932076 Free PMC article.
-
SNW1 is a critical regulator of spatial BMP activity, neural plate border formation, and neural crest specification in vertebrate embryos.PLoS Biol. 2011 Feb 15;9(2):e1000593. doi: 10.1371/journal.pbio.1000593. PLoS Biol. 2011. PMID: 21358802 Free PMC article.
-
Gradients and thresholds: BMP response gradients unveiled in Drosophila embryos.Trends Genet. 2003 Dec;19(12):701-8. doi: 10.1016/j.tig.2003.10.009. Trends Genet. 2003. PMID: 14642751 Review.
Cited by
-
An RNA interference approach for functional studies in the sea urchin and its use in analysis of Nodal signaling gradients.bioRxiv [Preprint]. 2024 Jul 9:2024.06.20.599930. doi: 10.1101/2024.06.20.599930. bioRxiv. 2024. Update in: Dev Biol. 2024 Dec;516:59-70. doi: 10.1016/j.ydbio.2024.08.002. PMID: 38979202 Free PMC article. Updated. Preprint.
-
Modelling the structure of Short Gastrulation and generation of a toolkit for studying its function in Drosophila.Biol Open. 2022 Jun 15;11(6):bio059199. doi: 10.1242/bio.059199. Epub 2022 Jun 10. Biol Open. 2022. PMID: 35603711 Free PMC article.
-
Bone morphogenetic protein signaling: the pathway and its regulation.Genetics. 2024 Feb 7;226(2):iyad200. doi: 10.1093/genetics/iyad200. Genetics. 2024. PMID: 38124338 Free PMC article. Review.
-
EpicTope: narrating protein sequence features to identify non-disruptive epitope tagging sites.bioRxiv [Preprint]. 2024 Mar 11:2024.03.03.583232. doi: 10.1101/2024.03.03.583232. bioRxiv. 2024. PMID: 38559275 Free PMC article. Preprint.
-
Reconstitution of morphogen shuttling circuits.Sci Adv. 2023 Jul 14;9(28):eadf9336. doi: 10.1126/sciadv.adf9336. Epub 2023 Jul 12. Sci Adv. 2023. PMID: 37436981 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

