ECM alterations in Fndc3a (Fibronectin Domain Containing Protein 3A) deficient zebrafish cause temporal fin development and regeneration defects
- PMID: 31527654
- PMCID: PMC6746793
- DOI: 10.1038/s41598-019-50055-w
ECM alterations in Fndc3a (Fibronectin Domain Containing Protein 3A) deficient zebrafish cause temporal fin development and regeneration defects
Abstract
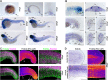
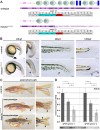
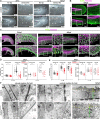
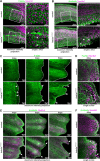
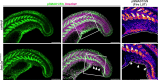
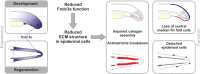
Fin development and regeneration are complex biological processes that are highly relevant in teleost fish. They share genetic factors, signaling pathways and cellular properties to coordinate formation of regularly shaped extremities. Especially correct tissue structure defined by extracellular matrix (ECM) formation is essential. Gene expression and protein localization studies demonstrated expression of fndc3a (fibronectin domain containing protein 3a) in both developing and regenerating caudal fins of zebrafish (Danio rerio). We established a hypomorphic fndc3a mutant line (fndc3awue1/wue1) via CRISPR/Cas9, exhibiting phenotypic malformations and changed gene expression patterns during early stages of median fin fold development. These developmental effects are mostly temporary, but result in a fraction of adults with permanent tail fin deformations. In addition, caudal fin regeneration in adult fndc3awue1/wue1 mutants is hampered by interference with actinotrichia formation and epidermal cell organization. Investigation of the ECM implies that loss of epidermal tissue structure is a common cause for both of the observed defects. Our results thereby provide a molecular link between these developmental processes and foreshadow Fndc3a as a novel temporal regulator of epidermal cell properties during extremity development and regeneration in zebrafish.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Dynamic remodeling of the extra cellular matrix during zebrafish fin regeneration.Gene Expr Patterns. 2015 Sep-Nov;19(1-2):21-9. doi: 10.1016/j.gep.2015.06.001. Epub 2015 Jun 21. Gene Expr Patterns. 2015. PMID: 26101996
-
Gene profile of zebrafish fin regeneration offers clues to kinetics, organization and biomechanics of basement membrane.Matrix Biol. 2019 Jan;75-76:82-101. doi: 10.1016/j.matbio.2018.07.005. Epub 2018 Jul 19. Matrix Biol. 2019. PMID: 30031067
-
Mechanical role of actinotrichia in shaping the caudal fin of zebrafish.Dev Biol. 2022 Jan;481:52-63. doi: 10.1016/j.ydbio.2021.09.003. Epub 2021 Sep 17. Dev Biol. 2022. PMID: 34537221
-
Zebrafish (Danio rerio) as a Model for Understanding the Process of Caudal Fin Regeneration.Zebrafish. 2020 Dec;17(6):359-372. doi: 10.1089/zeb.2020.1926. Epub 2020 Dec 1. Zebrafish. 2020. PMID: 33259770 Review.
-
Linking wound response and inflammation to regeneration in the zebrafish larval fin.Int J Dev Biol. 2018;62(6-7-8):473-477. doi: 10.1387/ijdb.170331hr. Int J Dev Biol. 2018. PMID: 29938759 Review.
Cited by
-
Elf3 deficiency during zebrafish development alters extracellular matrix organization and disrupts tissue morphogenesis.PLoS One. 2022 Nov 16;17(11):e0276255. doi: 10.1371/journal.pone.0276255. eCollection 2022. PLoS One. 2022. PMID: 36383615 Free PMC article.
-
S1PR1 serves as a viable drug target against pulmonary fibrosis by increasing the integrity of the endothelial barrier of the lung.Acta Pharm Sin B. 2023 Mar;13(3):1110-1127. doi: 10.1016/j.apsb.2022.10.006. Epub 2022 Oct 12. Acta Pharm Sin B. 2023. PMID: 36970190 Free PMC article.
-
Colocalization of corneal resistance factor GWAS loci with GTEx e/sQTLs highlights plausible candidate causal genes for keratoconus postnatal corneal stroma weakening.Front Genet. 2023 Aug 9;14:1171217. doi: 10.3389/fgene.2023.1171217. eCollection 2023. Front Genet. 2023. PMID: 37621707 Free PMC article.
-
Genome-wide transcriptome analysis reveals equine embryonic stem cell-derived tenocytes resemble fetal, not adult tenocytes.Stem Cell Res Ther. 2020 May 19;11(1):184. doi: 10.1186/s13287-020-01692-w. Stem Cell Res Ther. 2020. PMID: 32430075 Free PMC article.
-
UBA2 variants underlie a recognizable syndrome with variable aplasia cutis congenita and ectrodactyly.Genet Med. 2021 Sep;23(9):1624-1635. doi: 10.1038/s41436-021-01182-1. Epub 2021 May 26. Genet Med. 2021. PMID: 34040189 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

