The adenosine generating enzymes CD39/CD73 control microglial processes ramification in the mouse brain
- PMID: 28376099
- PMCID: PMC5380357
- DOI: 10.1371/journal.pone.0175012
The adenosine generating enzymes CD39/CD73 control microglial processes ramification in the mouse brain
Abstract
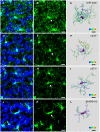
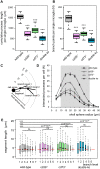
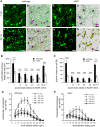
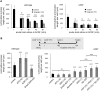
Microglial cells invade the brain as amoeboid precursors and acquire a highly ramified morphology in the postnatal brain. Microglia express all essential purinergic elements such as receptors, nucleoside transporters and ecto-enzymes, including CD39 (NTPDase1) and CD73 (5'-nucleotidase), which sequentially degrade extracellular ATP to adenosine. Here, we show that constitutive deletion of CD39 and CD73 or both caused an inhibition of the microglia ramified phenotype in the brain with a reduction in the length of processes, branching frequency and number of intersections with Sholl spheres. In vitro, unlike wild-type microglia, cd39-/- and cd73-/- microglial cells were less complex and did not respond to ATP with the transformation into a more ramified phenotype. In acute brain slices, wild-type microglia retracted approximately 50% of their processes within 15 min after slicing of the brain, and this phenomenon was augmented in cd39-/- mice; moreover, the elongation of microglial processes towards the source of ATP or towards a laser lesion was observed only in wild-type but not in cd39-/- microglia. An elevation of extracellular adenosine 1) by the inhibition of adenosine transport with dipyridamole, 2) by application of exogenous adenosine or 3) by degradation of endogenous ATP/ADP with apyrase enhanced spontaneous and ATP-induced ramification of cd39-/- microglia in acute brain slices and facilitated the transformation of cd39-/- and cd73-/- microglia into a ramified process-bearing phenotype in vitro. These data indicate that under normal physiological conditions, CD39 and CD73 nucleotidases together with equilibrative nucleoside transporter 1 (ENT1) control the fate of extracellular adenosine and thereby the ramification of microglial processes.
Conflict of interest statement
Figures
Similar articles
-
Role of the CD39/CD73 Purinergic Pathway in Modulating Arterial Thrombosis in Mice.Arterioscler Thromb Vasc Biol. 2016 Sep;36(9):1809-20. doi: 10.1161/ATVBAHA.116.307374. Epub 2016 Jul 14. Arterioscler Thromb Vasc Biol. 2016. PMID: 27417582 Free PMC article.
-
CD73 controls ocular adenosine levels and protects retina from light-induced phototoxicity.Cell Mol Life Sci. 2022 Feb 25;79(3):152. doi: 10.1007/s00018-022-04187-4. Cell Mol Life Sci. 2022. PMID: 35212809 Free PMC article.
-
Adenosine and inflammation: CD39 and CD73 are critical mediators in LPS-induced PMN trafficking into the lungs.FASEB J. 2009 Feb;23(2):473-82. doi: 10.1096/fj.08-119701. Epub 2008 Oct 6. FASEB J. 2009. PMID: 18838482
-
The role of the CD39-CD73-adenosine pathway in liver disease.J Cell Physiol. 2021 Feb;236(2):851-862. doi: 10.1002/jcp.29932. Epub 2020 Jul 10. J Cell Physiol. 2021. PMID: 32648591 Review.
-
Extracellular adenosine generation in the regulation of pro-inflammatory responses and pathogen colonization.Biomolecules. 2015 May 5;5(2):775-92. doi: 10.3390/biom5020775. Biomolecules. 2015. PMID: 25950510 Free PMC article. Review.
Cited by
-
Extracellular ATP/adenosine dynamics in the brain and its role in health and disease.Front Cell Dev Biol. 2024 Jan 18;11:1343653. doi: 10.3389/fcell.2023.1343653. eCollection 2023. Front Cell Dev Biol. 2024. PMID: 38304611 Free PMC article. Review.
-
P2Y13 receptors regulate microglial morphology, surveillance, and resting levels of interleukin 1β release.Glia. 2020 Feb;68(2):328-344. doi: 10.1002/glia.23719. Epub 2019 Sep 14. Glia. 2020. PMID: 31520551 Free PMC article.
-
Assessing Microglial Dynamics by Live Imaging.Front Immunol. 2021 Mar 8;12:617564. doi: 10.3389/fimmu.2021.617564. eCollection 2021. Front Immunol. 2021. PMID: 33763064 Free PMC article. Review.
-
Expanding the clinical relevance of the 5'-nucleotidase cN-II/NT5C2.Purinergic Signal. 2018 Dec;14(4):321-329. doi: 10.1007/s11302-018-9627-2. Epub 2018 Oct 25. Purinergic Signal. 2018. PMID: 30362044 Free PMC article. Review.
-
Spatial Distribution and Expression of Ectonucleotidases in Rat Hippocampus After Removal of Ovaries and Estradiol Replacement.Mol Neurobiol. 2019 Mar;56(3):1933-1945. doi: 10.1007/s12035-018-1217-3. Epub 2018 Jul 6. Mol Neurobiol. 2019. PMID: 29978426
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

