Loss of heterozygosity at D8S262: an early genetic event of hepatocarcinogenesis
- PMID: 26076954
- PMCID: PMC4469120
- DOI: 10.1186/s13000-015-0308-y
Loss of heterozygosity at D8S262: an early genetic event of hepatocarcinogenesis
Abstract
Background: Hepatocellular carcinoma (HCC) is a multi-factor, multi-step, multi-gene and complicated process resulting from the accumulation of sequential genetic and epigenetic alterations. An important change among them is from precancerous lesions to HCC. However, only few studies have been reported about the sequential genetic changes during hepatocarcinogenesis.
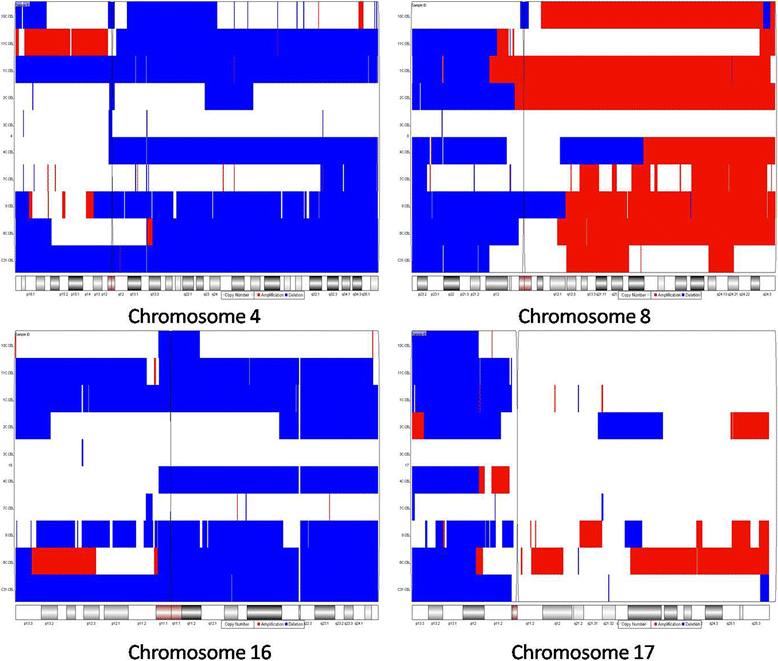
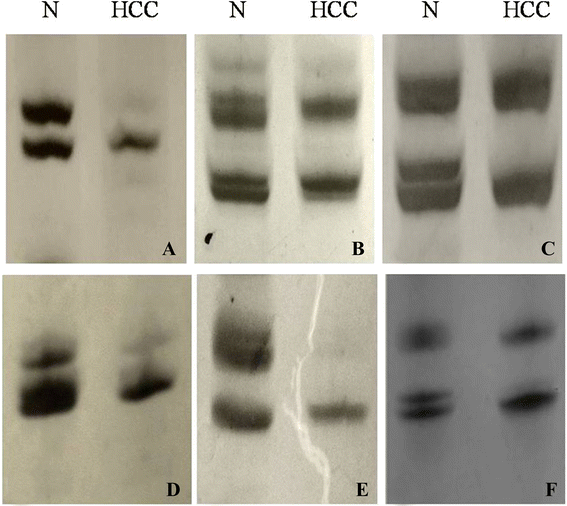
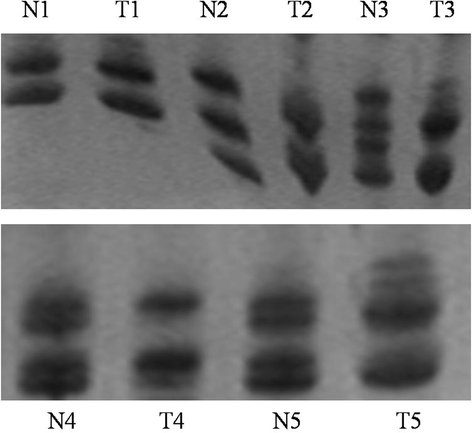
Methods: We observed firstly molecular karyotypes of 10 matched HCC using Affymetrix single-nucleotide polymorphism (SNP) 6.0 arrays, and found chromosomal fragments with high incidence (more than 70%) of loss of heterozygosity (LOH). Then, we selected 28 microsatellite markers at some gene spanning these chromosomal fragments, and examined the frequency of LOH of 128 matched HCC and 43 matched precancerous lesions-dysplastic nodules (DN) by a PCR-based analysis. Finally, we investigated the expression of proteins encoded by these genes in HCC, DN and the surrounding hepatic tissues.
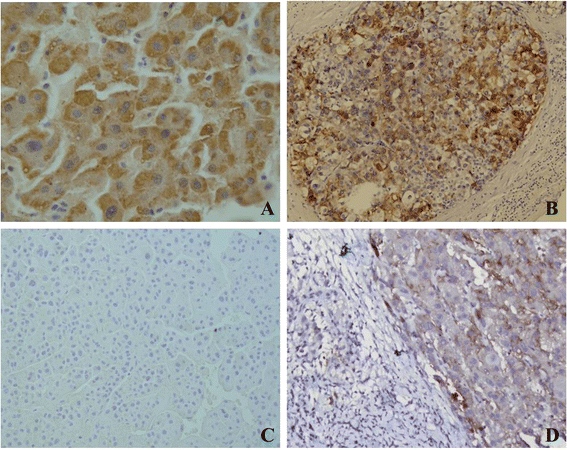
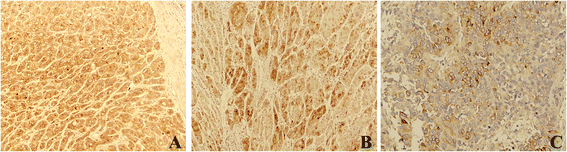
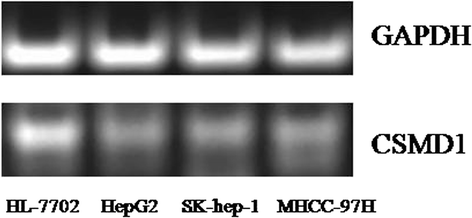
Results: The result of Affymetrix SNP6.0 arrays demonstrated that more than 70% (7/10) cases had chromosomal fragment deletion on 4q13.3-35.1, 8p23.2-21.2, 16q11.2-24.3, and 17p13.3-12. Among 28 microsatellite markers selected, LOH frequencies at D8S262 for DN and HCC were found to be the highest, 51.2% and 72.7%, respectively. Immunohistochemically, the positive rate of its adjacent gene CSMD1 in HCC, DN, and the surrounding hepatic tissues were 27.3% (35/128), 75% (33/44), and 82% (105/128), respectively.
Conclusions: LOH at D8S262 may be associated with an early genetic event of hepatocarcinogenesis, and a predictor for the monitor and prevention of HCC.
Virtual slides: The virtual slides for this article can be found here: http://www.diagnosticpathology.diagnomx.eu/vs/1557074981159099 .
Figures
Similar articles
-
Dysplastic nodules with glypican-3 positive immunostaining: a risk for early hepatocellular carcinoma.PLoS One. 2014 Jan 31;9(1):e87120. doi: 10.1371/journal.pone.0087120. eCollection 2014. PLoS One. 2014. PMID: 24498024 Free PMC article.
-
Loss of heterozygosity of chromosome 8p and 11p in the dysplastic nodule and hepatocellular carcinoma.J Gastroenterol Hepatol. 2003 Apr;18(4):430-6. doi: 10.1046/j.1440-1746.2003.02997.x. J Gastroenterol Hepatol. 2003. PMID: 12653892
-
An early lesion in hepatic carcinogenesis: loss of heterozygosity in human cirrhotic livers and dysplastic nodules at the 1p36-p34 region.Hepatology. 2001 Jun;33(6):1415-24. doi: 10.1053/jhep.2001.24751. Hepatology. 2001. PMID: 11391530
-
Latest developments in precancerous lesions of hepatocellular carcinoma.World J Gastroenterol. 2016 Mar 28;22(12):3305-14. doi: 10.3748/wjg.v22.i12.3305. World J Gastroenterol. 2016. PMID: 27022212 Free PMC article. Review.
-
Review of genetic and epigenetic alterations in hepatocarcinogenesis.J Gastroenterol Hepatol. 2006 Jan;21(1 Pt 1):15-21. doi: 10.1111/j.1440-1746.2005.04043.x. J Gastroenterol Hepatol. 2006. PMID: 16706806 Review.
Cited by
-
High expression of TOP2A gene predicted poor prognosis of hepatocellular carcinoma after radical hepatectomy.Transl Cancer Res. 2020 Feb;9(2):983-992. doi: 10.21037/tcr.2019.12.46. Transl Cancer Res. 2020. PMID: 35117443 Free PMC article.
-
Deregulation of CSMD1 targeted by microRNA-10b drives gastric cancer progression through the NF-κB pathway.Int J Biol Sci. 2019 Aug 6;15(10):2075-2086. doi: 10.7150/ijbs.23802. eCollection 2019. Int J Biol Sci. 2019. PMID: 31592231 Free PMC article.
-
Genomic and transcriptomic somatic alterations of hepatocellular carcinoma in non-cirrhotic livers.Cancer Genet. 2022 Jun;264-265:90-99. doi: 10.1016/j.cancergen.2022.04.002. Epub 2022 Apr 30. Cancer Genet. 2022. PMID: 35568002 Free PMC article.
-
Partial monosomy 8p and trisomy 16q in two children with developmental delay detected by array comparative genomic hybridization.Mol Med Rep. 2017 Dec;16(6):8808-8818. doi: 10.3892/mmr.2017.7760. Epub 2017 Oct 10. Mol Med Rep. 2017. PMID: 29039589 Free PMC article. Review.
References
-
- Bannasch P, Jahn UR, Hacker HJ, Su Q, Hoffmann W, Pichlmayr R, et al. Focal hepatic glycogenosis: a putative preneoplastic lesion associated with neoplasia and cirrhosis in explanted human livers. Int J Oncol. 1997;10(2):261–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

