Minimization of extracellular space as a driving force in prokaryote association and the origin of eukaryotes
- PMID: 25406691
- PMCID: PMC4289276
- DOI: 10.1186/1745-6150-9-24
Minimization of extracellular space as a driving force in prokaryote association and the origin of eukaryotes
Erratum in
-
Erratum to: Minimization of extracellular space as a driving force in prokaryote association and the origin of eukaryotes.Biol Direct. 2015 Mar 28;10:11. doi: 10.1186/s13062-015-0037-x. Biol Direct. 2015. PMID: 25888113 Free PMC article.
Abstract
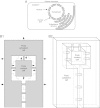
Background: Internalization-based hypotheses of eukaryotic origin require close physical association of host and symbiont. Prior hypotheses of how these associations arose include chance, specific metabolic couplings between partners, and prey-predator/parasite interactions. Since these hypotheses were proposed, it has become apparent that mixed-species, close-association assemblages (biofilms) are widespread and predominant components of prokaryotic ecology. Which forces drove prokaryotes to evolve the ability to form these assemblages are uncertain. Bacteria and archaea have also been found to form membrane-lined interconnections (nanotubes) through which proteins and RNA pass. These observations, combined with the structure of the nuclear envelope and an energetic benefit of close association (see below), lead us to propose a novel hypothesis of the driving force underlying prokaryotic close association and the origin of eukaryotes.
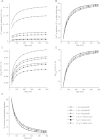
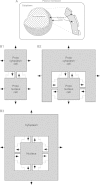
Results: Respiratory proton transport does not alter external pH when external volume is effectively infinite. Close physical association decreases external volume. For small external volumes, proton transport decreases external pH, resulting in each transported proton increasing proton motor force to a greater extent. We calculate here that in biofilms this effect could substantially decrease how many protons need to be transported to achieve a given proton motor force. Based as it is solely on geometry, this energetic benefit would occur for all prokaryotes using proton-based respiration.
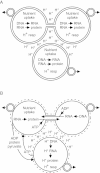
Conclusions: This benefit may be a driving force in biofilm formation. Under this hypothesis a very wide range of prokaryotic species combinations could serve as eukaryotic progenitors. We use this observation and the discovery of prokaryotic nanotubes to propose that eukaryotes arose from physically distinct, functionally specialized (energy factory, protein factory, DNA repository/RNA factory), obligatorily symbiotic prokaryotes in which the protein factory and DNA repository/RNA factory cells were coupled by nanotubes and the protein factory ultimately internalized the other two. This hypothesis naturally explains many aspects of eukaryotic physiology, including the nuclear envelope being a folded single membrane repeatedly pierced by membrane-bound tubules (the nuclear pores), suggests that species analogous or homologous to eukaryotic progenitors are likely unculturable as monocultures, and makes a large number of testable predictions.
Reviewers: This article was reviewed by Purificación López-García and Toni Gabaldón.
Figures

Similar articles
-
Division of labour in a matrix, rather than phagocytosis or endosymbiosis, as a route for the origin of eukaryotic cells.Biol Direct. 2020 Apr 28;15(1):8. doi: 10.1186/s13062-020-00260-9. Biol Direct. 2020. PMID: 32345370 Free PMC article.
-
Energetics and genetics across the prokaryote-eukaryote divide.Biol Direct. 2011 Jun 30;6:35. doi: 10.1186/1745-6150-6-35. Biol Direct. 2011. PMID: 21714941 Free PMC article.
-
Role of lipid membrane-nucleic acid interactions, DNA-membrane contacts and metal (II) cations in origination of initial cells and in evolution of prokaryotes to eukaryotes.Bioelectrochemistry. 2002 Nov;58(1):41-6. doi: 10.1016/s1567-5394(02)00134-2. Bioelectrochemistry. 2002. PMID: 12401569
-
The path from the RNA world.J Mol Evol. 1998 Jan;46(1):1-17. doi: 10.1007/pl00006275. J Mol Evol. 1998. PMID: 9419221 Review.
-
On the origin of eukaryotic cells and their endomembranes.Riv Biol. 2006 Sep-Dec;99(3):499-519. Riv Biol. 2006. PMID: 17299702 Review.
Cited by
-
Erratum to: Minimization of extracellular space as a driving force in prokaryote association and the origin of eukaryotes.Biol Direct. 2015 Mar 28;10:11. doi: 10.1186/s13062-015-0037-x. Biol Direct. 2015. PMID: 25888113 Free PMC article.
-
Membrane nanotubes are ancient machinery for cell-to-cell communication and transport. Their interference with the immune system.Biol Futur. 2021 Mar;72(1):25-36. doi: 10.1007/s42977-020-00062-0. Epub 2021 Feb 8. Biol Futur. 2021. PMID: 34554502 Free PMC article. Review.
References
-
- Atteia A, Adrait A, Brugière S, Tardif M, van Lis R, Deusch O, Degan T, Kuhn L, Gontero B, Martin W, Garin J, Joyard J, Rolland N. A proteomic survey of Chlamydomonas reinhardtii mitochondria sheds new light on the metabolic plasticity of the organelle and on the nature of the α-proteobacterial mitochondrial ancestor. Mol Biol Evol. 2009;26:1533–1548. doi: 10.1093/molbev/msp068. - DOI - PubMed
-
- Esser C, Ahmadinejad N, Wiegand C, Rotte C, Sebastiani F, Gelius-Dietrich G, Henze K, Kretschmann E, Richly E, Leister D, Bryant D, Steel MA, Lockhart PJ, Penny D, Martin W. A genome phylogeny for mitochondria among α-proteobacterial and a predominantly eubacterial ancestry of yeast nuclear genes. Mol Biol Evol. 2004;21:1643–1660. doi: 10.1093/molbev/msh160. - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

