Human adipose tissue-derived stromal/stem cells promote migration and early metastasis of triple negative breast cancer xenografts
- PMID: 24586900
- PMCID: PMC3938488
- DOI: 10.1371/journal.pone.0089595
Human adipose tissue-derived stromal/stem cells promote migration and early metastasis of triple negative breast cancer xenografts
Abstract
Background: Fat grafting is used to restore breast defects after surgical resection of breast tumors. Supplementing fat grafts with adipose tissue-derived stromal/stem cells (ASCs) is proposed to improve the regenerative/restorative ability of the graft and retention. However, long term safety for ASC grafting in proximity of residual breast cancer cells is unknown. The objective of this study was to determine the impact of human ASCs derived from abdominal lipoaspirates of three donors, on a human breast cancer model that exhibits early metastasis.
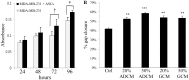
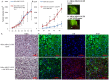
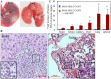
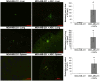
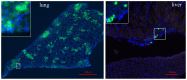
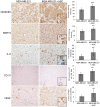
Methodology/principal findings: Human MDA-MB-231 breast cancer cells represents "triple negative" breast cancer that exhibits early micrometastasis to multiple mouse organs [1]. Human ASCs were derived from abdominal adipose tissue from three healthy female donors. Indirect co-culture of MDA-MB-231 cells with ASCs, as well as direct co-culture demonstrated that ASCs had no effect on MDA-MB-231 growth. Indirect co-culture, and ASC conditioned medium (CM) stimulated migration of MDA-MB-231 cells. ASC/RFP cells from two donors co-injected with MDA-MB-231/GFP cells exhibited a donor effect for stimulation of primary tumor xenografts. Both ASC donors stimulated metastasis. ASC/RFP cells were viable, and integrated with MDA-MB-231/GFP cells in the tumor. Tumors from the co-injection group of one ASC donor exhibited elevated vimentin, matrix metalloproteinase-9 (MMP-9), IL-8, VEGF and microvessel density. The co-injection group exhibited visible metastases to the lung/liver and enlarged spleen not evident in mice injected with MDA-MB-231/GFP alone. Quantitation of the total area of GFP fluorescence and human chromosome 17 DNA in mouse organs, H&E stained paraffin sections and fluorescent microscopy confirmed multi-focal metastases to lung/liver/spleen in the co-injection group without evidence of ASC/RFP cells.
Conclusions: Human ASCs derived from abdominal lipoaspirates of two donors stimulated metastasis of MDA-MB-231 breast tumor xenografts to multiple mouse organs. MDA-MB-231 tumors co-injected with ASCs from one donor exhibited partial EMT, expression of MMP-9, and increased angiogenesis.
Conflict of interest statement
Figures

Similar articles
-
The Crosstalk Between Adipose-Derived Stem or Stromal Cells (ASC) and Cancer Cells and ASC-Mediated Effects on Cancer Formation and Progression-ASCs: Safety Hazard or Harmless Source of Tropism?Stem Cells Transl Med. 2022 Apr 29;11(4):394-406. doi: 10.1093/stcltm/szac002. Stem Cells Transl Med. 2022. PMID: 35274703 Free PMC article. Review.
-
Human Adipose Tissue-Derived Stromal/Stem Cells Promote Migration and Early Metastasis of Head and Neck Cancer Xenografts.Aesthet Surg J. 2016 Jan;36(1):93-104. doi: 10.1093/asj/sjv090. Epub 2015 Jun 10. Aesthet Surg J. 2016. PMID: 26063833
-
Differential Response of Non-cancerous and Malignant Breast Cancer Cells to Conditioned Medium of Adipose tissue-derived Stromal Cells (ASCs).Int J Med Sci. 2019 Jun 2;16(6):893-901. doi: 10.7150/ijms.27125. eCollection 2019. Int J Med Sci. 2019. PMID: 31337963 Free PMC article.
-
Breast Cancer-Stromal Interactions: Adipose-Derived Stromal/Stem Cell Age and Cancer Subtype Mediated Remodeling.Stem Cells Dev. 2022 Oct;31(19-20):604-620. doi: 10.1089/scd.2021.0279. Epub 2022 Jul 12. Stem Cells Dev. 2022. PMID: 35579936 Free PMC article.
-
Adult adipose-derived stem cells and breast cancer: a controversial relationship.Springerplus. 2014 Jul 8;3:345. doi: 10.1186/2193-1801-3-345. eCollection 2014. Springerplus. 2014. PMID: 25089245 Free PMC article. Review.
Cited by
-
The role of adipose-derived stem cells in breast cancer progression and metastasis.Stem Cells Int. 2015;2015:120949. doi: 10.1155/2015/120949. Epub 2015 Apr 27. Stem Cells Int. 2015. PMID: 26000019 Free PMC article. Review.
-
Visfatin Mediates Malignant Behaviors through Adipose-Derived Stem Cells Intermediary in Breast Cancer.Cancers (Basel). 2019 Dec 20;12(1):29. doi: 10.3390/cancers12010029. Cancers (Basel). 2019. PMID: 31861872 Free PMC article.
-
Tumor-Stroma-Inflammation Networks Promote Pro-metastatic Chemokines and Aggressiveness Characteristics in Triple-Negative Breast Cancer.Front Immunol. 2019 Apr 12;10:757. doi: 10.3389/fimmu.2019.00757. eCollection 2019. Front Immunol. 2019. PMID: 31031757 Free PMC article.
-
Breast fat grafting and cancer: a systematic review of the science behind enhancements and concerns.Transl Breast Cancer Res. 2024 Apr 29;5:14. doi: 10.21037/tbcr-23-54. eCollection 2024. Transl Breast Cancer Res. 2024. PMID: 38751673 Free PMC article. Review.
-
The Crosstalk Between Adipose-Derived Stem or Stromal Cells (ASC) and Cancer Cells and ASC-Mediated Effects on Cancer Formation and Progression-ASCs: Safety Hazard or Harmless Source of Tropism?Stem Cells Transl Med. 2022 Apr 29;11(4):394-406. doi: 10.1093/stcltm/szac002. Stem Cells Transl Med. 2022. PMID: 35274703 Free PMC article. Review.
References
-
- Anbalagan M, Ali A, Jones RK, Marsden CG, Sheng M, et al. (2012) Peptidomimetic Src/pretubulin inhibitor KX-01 alone and in combination with paclitaxel suppresses growth, metastasis in human ER/PR/HER2-negative tumor xenografts. Mol Cancer Ther 1535-7163.MCT-12-0146 [pii];10.1158/1535-7163.MCT-12-0146 [doi] - DOI - PMC - PubMed
-
- Coleman SR (1997) Facial recontouring with lipostructure. Clin Plast Surg 24: 347–367. - PubMed
-
- Fournier PF (1990) Facial recontouring with fat grafting. Dermatol Clin 8: 523–537. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

