Plasmodium berghei MAPK1 displays differential and dynamic subcellular localizations during liver stage development
- PMID: 23544094
- PMCID: PMC3609774
- DOI: 10.1371/journal.pone.0059755
Plasmodium berghei MAPK1 displays differential and dynamic subcellular localizations during liver stage development
Abstract
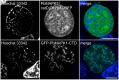
Mitogen-activated protein kinases (MAPKs) regulate key signaling events in eukaryotic cells. In the genomes of protozoan Plasmodium parasites, the causative agents of malaria, two genes encoding kinases with significant homology to other eukaryotic MAPKs have been identified (mapk1, mapk2). In this work, we show that both genes are transcribed during Plasmodium berghei liver stage development, and analyze expression and subcellular localization of the PbMAPK1 protein in liver stage parasites. Live cell imaging of transgenic parasites expressing GFP-tagged PbMAPK1 revealed a nuclear localization of PbMAPK1 in the early schizont stage mediated by nuclear localization signals in the C-terminal domain. In contrast, a distinct localization of PbMAPK1 in comma/ring-shaped structures in proximity to the parasite's nuclei and the invaginating parasite membrane was observed during the cytomere stage of parasite development as well as in immature blood stage schizonts. The PbMAPK1 localization was found to be independent of integrity of a motif putatively involved in ATP binding, integrity of the putative activation motif and the presence of a predicted coiled-coil domain in the C-terminal domain. Although PbMAPK1 knock out parasites showed normal liver stage development, the kinase may still fulfill a dual function in both schizogony and merogony of liver stage parasites regulated by its dynamic and stage-dependent subcellular localization.
Conflict of interest statement
Figures

Similar articles
-
Features of autophagic cell death in Plasmodium liver-stage parasites.Autophagy. 2013 Apr;9(4):568-80. doi: 10.4161/auto.23689. Epub 2013 Feb 6. Autophagy. 2013. PMID: 23388496 Free PMC article.
-
Identification of an AP2-family protein that is critical for malaria liver stage development.PLoS One. 2012;7(11):e47557. doi: 10.1371/journal.pone.0047557. Epub 2012 Nov 7. PLoS One. 2012. PMID: 23144823 Free PMC article.
-
P. berghei telomerase subunit TERT is essential for parasite survival.PLoS One. 2014 Oct 2;9(9):e108930. doi: 10.1371/journal.pone.0108930. eCollection 2014. PLoS One. 2014. PMID: 25275500 Free PMC article.
-
RNA-Seq Analysis Illuminates the Early Stages of Plasmodium Liver Infection.mBio. 2020 Feb 4;11(1):e03234-19. doi: 10.1128/mBio.03234-19. mBio. 2020. PMID: 32019802 Free PMC article.
-
Effect of thioredoxin peroxidase-1 gene disruption on the liver stages of the rodent malaria parasite Plasmodium berghei.Parasitol Int. 2015 Jun;64(3):290-4. doi: 10.1016/j.parint.2014.09.013. Epub 2014 Oct 5. Parasitol Int. 2015. PMID: 25284813
Cited by
-
Revisiting the Role of Toxoplasma gondii ERK7 in the Maintenance and Stability of the Apical Complex.mBio. 2021 Oct 26;12(5):e0205721. doi: 10.1128/mBio.02057-21. Epub 2021 Oct 5. mBio. 2021. PMID: 34607461 Free PMC article.
-
PfMAP-2 is essential for male gametogenesis in the malaria parasite Plasmodium falciparum.Sci Rep. 2020 Jul 17;10(1):11930. doi: 10.1038/s41598-020-68717-5. Sci Rep. 2020. PMID: 32681115 Free PMC article.
-
A Plasmodium berghei putative serine-threonine kinase 2 (PBANKA_0311400) is required for late liver stage development and timely initiation of blood stage infection.Biol Open. 2019 Aug 23;8(8):bio042028. doi: 10.1242/bio.042028. Biol Open. 2019. PMID: 31444161 Free PMC article.
-
Not your Mother's MAPKs: Apicomplexan MAPK function in daughter cell budding.PLoS Pathog. 2022 Oct 13;18(10):e1010849. doi: 10.1371/journal.ppat.1010849. eCollection 2022 Oct. PLoS Pathog. 2022. PMID: 36227859 Free PMC article. Review.
References
-
- Sturm A, Amino R, van de Sand C, Regen T, Retzlaff S, et al. (2006) Manipulation of host hepatocytes by the malaria parasite for delivery into liver sinusoids. Science 313: 1287–1290. - PubMed
-
- Stanway RR, Mueller N, Zobiak B, Graewe S, Froehlke U, et al. (2011) Organelle segregation into Plasmodium liver stage merozoites. Cell Microbiol 13: 1768–1782. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

