TGFβ1 inhibition increases the radiosensitivity of breast cancer cells in vitro and promotes tumor control by radiation in vivo
- PMID: 22028490
- PMCID: PMC3724539
- DOI: 10.1158/1078-0432.CCR-11-0544
TGFβ1 inhibition increases the radiosensitivity of breast cancer cells in vitro and promotes tumor control by radiation in vivo
Abstract
Purpose: To determine whether inhibition of TGFβ signaling prior to irradiation sensitizes human and murine cancer cells in vitro and in vivo.
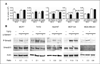
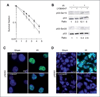
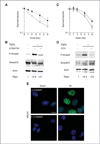
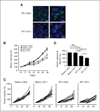
Experimental design: TGFβ-mediated growth and Smad phosphorylation of MCF7, Hs578T, MDA-MB-231, and T47D human breast cancer cell lines were examined and correlated with clonogenic survival following graded radiation doses with and without pretreatment with LY364947, a small molecule inhibitor of the TGFβ type I receptor kinase. The DNA damage response was assessed in irradiated MDA-MB-231 cells pretreated with LY364947 in vitro and LY2109761, a pharmacokinetically stable inhibitor of TGFβ signaling, in vivo. The in vitro response of a syngeneic murine tumor, 4T1, was tested using a TGFβ neutralizing antibody, 1D11, with single or fractionated radiation doses in vivo.
Results: Human breast cancer cell lines pretreated with TGFβ small molecule inhibitor were radiosensitized, irrespective of sensitivity to TGFβ growth inhibition. Consistent with increased clonogenic cell death, radiation-induced phosphorylation of H2AX and p53 was significantly reduced in MDA-MB-231 triple-negative breast cancer cells when pretreated in vitro or in vivo with a TGFβ type I receptor kinase inhibitor. Moreover, TGFβ neutralizing antibodies increased radiation sensitivity, blocked γH2AX foci formation, and significantly increased tumor growth delay in 4T1 murine mammary tumors in response to single and fractionated radiation exposures.
Conclusion: These results show that TGFβ inhibition prior to radiation attenuated DNA damage responses, increased clonogenic cell death, and promoted tumor growth delay, and thus may be an effective adjunct in cancer radiotherapy.
©2011 AACR
Conflict of interest statement
Figures


Similar articles
-
Attenuation of the DNA damage response by transforming growth factor-beta inhibitors enhances radiation sensitivity of non-small-cell lung cancer cells in vitro and in vivo.Int J Radiat Oncol Biol Phys. 2015 Jan 1;91(1):91-9. doi: 10.1016/j.ijrobp.2014.09.026. Int J Radiat Oncol Biol Phys. 2015. PMID: 25835621
-
Blockade of TGF-β signaling by the TGFβR-I kinase inhibitor LY2109761 enhances radiation response and prolongs survival in glioblastoma.Cancer Res. 2011 Dec 1;71(23):7155-67. doi: 10.1158/0008-5472.CAN-11-1212. Epub 2011 Oct 17. Cancer Res. 2011. PMID: 22006998
-
Effect of transforming growth factor beta (TGF-β) receptor I kinase inhibitor on prostate cancer bone growth.Bone. 2012 Mar;50(3):695-703. doi: 10.1016/j.bone.2011.11.022. Epub 2011 Dec 7. Bone. 2012. PMID: 22173053 Free PMC article.
-
Inhibiting breast cancer progression by exploiting TGFbeta signaling.Curr Drug Targets. 2010 Sep;11(9):1089-102. doi: 10.2174/138945010792006771. Curr Drug Targets. 2010. PMID: 20545612 Review.
-
Tumors as organs: biologically augmenting radiation therapy by inhibiting transforming growth factor β activity in carcinomas.Semin Radiat Oncol. 2013 Oct;23(4):242-51. doi: 10.1016/j.semradonc.2013.05.001. Semin Radiat Oncol. 2013. PMID: 24012338 Free PMC article. Review.
Cited by
-
RAC1B Induces SMAD7 via USP26 to Suppress TGFβ1-Dependent Cell Migration in Mesenchymal-Subtype Carcinoma Cells.Cancers (Basel). 2020 Jun 11;12(6):1545. doi: 10.3390/cancers12061545. Cancers (Basel). 2020. PMID: 32545415 Free PMC article.
-
TGFβ Is a Master Regulator of Radiation Therapy-Induced Antitumor Immunity.Cancer Res. 2015 Jun 1;75(11):2232-42. doi: 10.1158/0008-5472.CAN-14-3511. Epub 2015 Apr 9. Cancer Res. 2015. PMID: 25858148 Free PMC article.
-
Immunomodulatory effects of carbon ion radiotherapy in patients with localized prostate cancer.J Cancer Res Clin Oncol. 2023 Jul;149(8):4533-4545. doi: 10.1007/s00432-022-04194-9. Epub 2022 Sep 23. J Cancer Res Clin Oncol. 2023. PMID: 36138265 Free PMC article.
-
Tumor in the Crossfire: Inhibiting TGF-β to Enhance Cancer Immunotherapy.BioDrugs. 2022 Mar;36(2):153-180. doi: 10.1007/s40259-022-00521-1. Epub 2022 Mar 30. BioDrugs. 2022. PMID: 35353346 Free PMC article. Review.
-
Radiation-Induced Fibrotic Tumor Microenvironment Regulates Anti-Tumor Immune Response.Cancers (Basel). 2021 Oct 19;13(20):5232. doi: 10.3390/cancers13205232. Cancers (Basel). 2021. PMID: 34680381 Free PMC article.
References
-
- Moran JM, Elshaikh MA, Lawrence TS. Radiotherapy: what can be achieved by technical improvements in dose delivery? Lancet Oncol. 2005;6:51–58. - PubMed
-
- Andarawewa KL, Kirshner J, Mott JD, Barcellos-Hoff MH. TGFβ: Roles in DNA damage responses. In: Jakowlew s., editor. Transforming growth factor-beta in cancer therapy, volume II cancer treatment and therapy. Totowa: Humana Press; 2007. pp. 321–334.
-
- Jobling MF, Mott JD, Finnegan MT, Jurukovski V, Erickson AC, Walian PJ, et al. Isoform specificity of redox-mediated TGF-β activation. Radiat Res. 2006;166:839–848. - PubMed
-
- Andarawewa KL, Costes SV, Fernandez-Garcia I, Chou WS, Ravani SA, Park H, et al. Radiation Dose and Quality Dependence of Epithelial to Mesenchymal Transition (EMT) Mediated by Transforming Growth Factor β. Int J Rad Onc Biol Phys. 2011;79:1523–1531. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

