Induction of autophagy by cystatin C: a mechanism that protects murine primary cortical neurons and neuronal cell lines
- PMID: 20352108
- PMCID: PMC2843718
- DOI: 10.1371/journal.pone.0009819
Induction of autophagy by cystatin C: a mechanism that protects murine primary cortical neurons and neuronal cell lines
Abstract
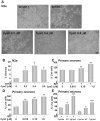
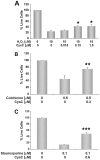
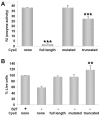
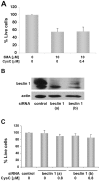
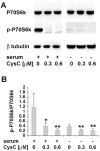
Cystatin C (CysC) expression in the brain is elevated in human patients with epilepsy, in animal models of neurodegenerative conditions, and in response to injury, but whether up-regulated CysC expression is a manifestation of neurodegeneration or a cellular repair response is not understood. This study demonstrates that human CysC is neuroprotective in cultures exposed to cytotoxic challenges, including nutritional-deprivation, colchicine, staurosporine, and oxidative stress. While CysC is a cysteine protease inhibitor, cathepsin B inhibition was not required for the neuroprotective action of CysC. Cells responded to CysC by inducing fully functional autophagy via the mTOR pathway, leading to enhanced proteolytic clearance of autophagy substrates by lysosomes. Neuroprotective effects of CysC were prevented by inhibiting autophagy with beclin 1 siRNA or 3-methyladenine. Our findings show that CysC plays a protective role under conditions of neuronal challenge by inducing autophagy via mTOR inhibition and are consistent with CysC being neuroprotective in neurodegenerative diseases. Thus, modulation of CysC expression has therapeutic implications for stroke, Alzheimer's disease, and other neurodegenerative disorders.
Conflict of interest statement
Figures


Similar articles
-
Cystatin C protects neuronal cells against mutant copper-zinc superoxide dismutase-mediated toxicity.Cell Death Dis. 2014 Oct 30;5(10):e1497. doi: 10.1038/cddis.2014.459. Cell Death Dis. 2014. PMID: 25356866 Free PMC article.
-
Neuroprotective Autophagic Flux Induced by Hyperbaric Oxygen Preconditioning is Mediated by Cystatin C.Neurosci Bull. 2019 Apr;35(2):336-346. doi: 10.1007/s12264-018-0313-8. Epub 2018 Dec 5. Neurosci Bull. 2019. PMID: 30519802 Free PMC article.
-
Cystatin C in aging and in Alzheimer's disease.Ageing Res Rev. 2016 Dec;32:38-50. doi: 10.1016/j.arr.2016.06.003. Epub 2016 Jun 19. Ageing Res Rev. 2016. PMID: 27333827 Free PMC article. Review.
-
Protective mechanisms by cystatin C in neurodegenerative diseases.Front Biosci (Schol Ed). 2011 Jan 1;3(2):541-54. doi: 10.2741/s170. Front Biosci (Schol Ed). 2011. PMID: 21196395 Free PMC article. Review.
-
Cystatin C rescues degenerating neurons in a cystatin B-knockout mouse model of progressive myoclonus epilepsy.Am J Pathol. 2010 Nov;177(5):2256-67. doi: 10.2353/ajpath.2010.100461. Epub 2010 Oct 1. Am J Pathol. 2010. PMID: 20889561 Free PMC article.
Cited by
-
Cystatins: unravelling the biological implications for neuroprotection.Arch Med Sci. 2023 Sep 19;20(1):157-166. doi: 10.5114/aoms/171706. eCollection 2024. Arch Med Sci. 2023. PMID: 38414464 Free PMC article.
-
Multiomic Analyses of Dopaminergic Neurons Isolated from Human Substantia Nigra in Parkinson's Disease: A Descriptive and Exploratory Study.Cell Mol Neurobiol. 2022 Nov;42(8):2805-2818. doi: 10.1007/s10571-021-01146-8. Epub 2021 Sep 15. Cell Mol Neurobiol. 2022. PMID: 34528139 Free PMC article.
-
Arctigenin improves neuropathy via ameliorating apoptosis and modulating autophagy in streptozotocin-induced diabetic mice.CNS Neurosci Ther. 2023 Oct;29(10):3068-3080. doi: 10.1111/cns.14249. Epub 2023 May 11. CNS Neurosci Ther. 2023. PMID: 37170684 Free PMC article.
-
Serum cystatin C and mild cognitive impairment: The mediating role of glucose homeostasis.Front Aging Neurosci. 2023 Mar 28;15:1102762. doi: 10.3389/fnagi.2023.1102762. eCollection 2023. Front Aging Neurosci. 2023. PMID: 37056689 Free PMC article.
-
Do hypoxia/normoxia culturing conditions change the neuroregulatory profile of Wharton Jelly mesenchymal stem cell secretome?Stem Cell Res Ther. 2015 Jul 24;6(1):133. doi: 10.1186/s13287-015-0124-z. Stem Cell Res Ther. 2015. PMID: 26204925 Free PMC article.
References
-
- Abrahamson M, Alvarez-Fernandez M, Nathanson CM. Cystatins. Biochem Soc Symp. 2003;70:179–199. - PubMed
-
- Bernstein HG, Kirschke H, Wiederanders B, Pollak KH, Zipress A, et al. The possible place of cathepsins and cystatins in the puzzle of Alzheimer disease: a review. Mol Chem Neuropathol. 1996;27:225–247. - PubMed
-
- Pawlik M, Sastre M, Calero M, Mathews PM, Schmidt SD, et al. Overexpression of human cystatin C in transgenic mice does not affect levels of endogenous brain amyloid β peptide. J Mol Neurosci. 2004;22:13–18. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

