Abstract
Purpose
Randomized trials report that single-inhaler triple therapy is more effective than dual bronchodilators at reducing exacerbations in patients with chronic obstructive pulmonary disease (COPD). However, this effect may have been influenced by the forced withdrawal of inhaled corticosteroids (ICS) at randomization. We used an adaptive selection new-user design to compare single-inhaler triple therapy with dual bronchodilators in real-world clinical practice.
Patients and Methods
We identified a cohort of COPD patients, 40 years or older, treated during 2017–2020, from the United Kingdom’s Clinical Practice Research Datalink, a real-world practice setting. ICS-naïve patients initiating single-inhaler triple therapy or dual bronchodilators were compared on the incidence of COPD exacerbation and pneumonia over one year, after adjustment by propensity score weighting.
Results
The cohort included 4106 new users of single-inhaler triple therapy and 29,702 of dual bronchodilators. Single-inhaler triple therapy was the first maintenance treatment in 44% of the users and 43% had no COPD exacerbations in the prior year. The adjusted hazard ratio (HR) of a first moderate or severe exacerbation with triple therapy relative to dual bronchodilators was 1.08 (95% confidence interval (CI): 1.00–1.16). Among patients with two or more prior exacerbations the HR was 0.83 (95% CI: 0.74–0.92), while for those with prior asthma diagnosis it was 0.86 (95% CI: 0.70–1.06) and with blood eosinophil count >300 cells/µL it was 0.89 (95% CI: 0.76–1.05). The incidence of severe pneumonia was increased with triple therapy (HR 1.50; 95% CI: 1.29–1.75).
Conclusion
In a real-world setting of COPD treatment among ICS-naïve patients, thus unaffected by ICS withdrawal, single-inhaler triple therapy was not more effective than dual bronchodilators at reducing the incidence of exacerbation, except among patients with multiple exacerbations. Single-inhaler triple therapy should be initiated mainly in patients with multiple exacerbations while, for most others, dual bronchodilators are just as effective whilst avoiding the excess risk of severe pneumonias.
Keywords: cohort studies, COPD exacerbation, pneumonia, propensity scores, real-world evidence
Introduction
The recommended pharmacological treatment of chronic obstructive pulmonary disease (COPD) includes first long-acting bronchodilators, namely long-acting muscarinic antagonists (LAMAs) and long-acting beta2-agonists (LABAs), or their combination.1 Inhaled corticosteroids (ICS) are generally added with increasing disease severity or for specific patient profiles such as high eosinophil counts or frequent exacerbations. Currently, several combinations of these three treatment classes are available, including single-inhaler triple therapy.
Two large trials compared the effectiveness and safety of single-inhaler triple therapy with single-inhaler dual bronchodilator (combining LAMA and LABA) on major outcomes in patients with COPD.2,3 The Informing the Pathway of COPD Treatment (IMPACT) and the Efficacy and Safety of Triple Therapy in Obstructive Lung Disease (ETHOS) trials reported a significant reduction in the rate of COPD exacerbation and mortality with the triple inhaler compared with the dual bronchodilator.4,5 However, these beneficial effects of triple therapy on reducing exacerbations and mortality were limited to the early period after randomization, with no benefit observed during the subsequent follow-up.6,7 Such an early benefit is compatible with an effect of abrupt ICS withdrawal at randomization among those allocated to LAMA-LABA.6–9 Indeed, the reductions in exacerbations and mortality in the IMPACT trial were observed mainly among patients previously on ICS.4,10 Adaptive selection trial designs for COPD treatments that tailor the randomisation scheme to the treatment already used by each subject at the time of trial enrolment have been proposed specifically to circumvent such effects of confounding by ICS discontinuation at randomization.11
In this paper, we report on an observational study that uses an adaptive selection design to compare the new single-inhaler triple with single-inhaler dual bronchodilator therapies in a real-world clinical practice setting.
Methods
Data Source
The study cohort was identified from the Clinical Practice Research Datalink (CPRD), a primary care database from the United Kingdom (UK) that contains primary care medical records for over 50 million people enrolled in more than 1800 general practices. Participating general practitioners in the CPRD-GOLD and Aurum networks record medical information as part of the routine care of patients, including demographic data, lifestyle factors, medical diagnoses recorded using Read and SNOMED codes, and prescriptions. Over 85% of the CPRD practices can be linked to the Hospital Episodes Statistics (HES) database. The CPRD population is representative of the overall population and these data sources have been validated.12–14 The information on medications and diagnoses has been validated and shown to be of high quality, particularly for studies of COPD.13–17
Study Design
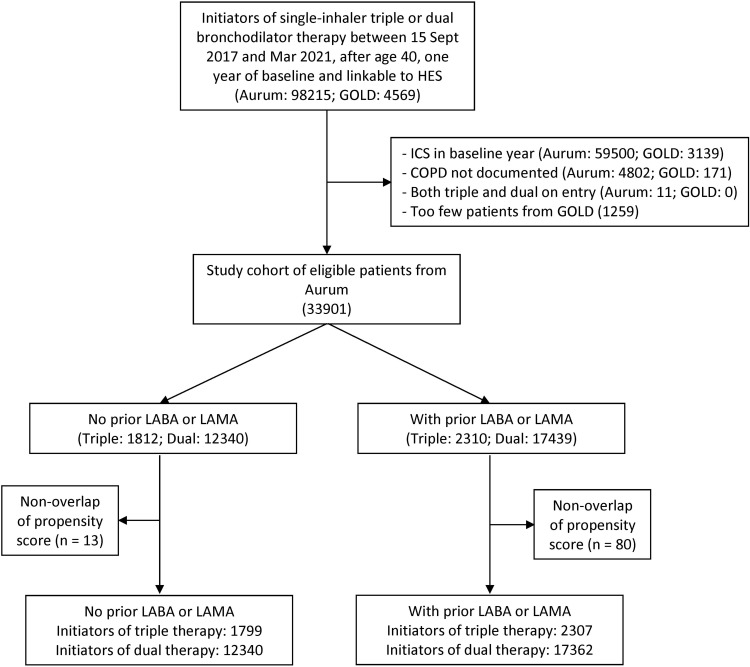
The study cohort included all patients with a physician diagnosis of COPD who received a single-inhaler triple or a single-inhaler dual bronchodilator after 15 September 2017, the date the triple inhaler became available in the UK. All subjects had to be at least 40 years of age on the date of their first COPD diagnosis. The study was restricted to the larger Aurum database, as few eligible patients were available in the GOLD database, and to patients linkable to the Hospital Episodes Statistics (HES) database.
Cohort entry was taken as the date of the first prescription for a triple (LAMA-LABA-ICS) or a dual bronchodilator (LAMA-LABA) single-inhaler. The study cohort included all subjects with at least one year of medical history prior to cohort entry to allow a baseline period to measure the covariates and to identify new use of these study combinations. To apply the adaptive selection design, we only included patients who did not receive ICS during the baseline year or at cohort entry.11
We used an incident new-user cohort design to compare the initiators of triple therapy with the initiators of dual bronchodilators.18,19 The subjects were stratified by their prior use of a LABA or LAMA (none, one, both) in the baseline year before initiating the study inhalers, to fit the adaptive selection trial design concept.11 The propensity score of treatment initiation with triple therapy was thus estimated by logistic regression using all covariates, separately for the three prior LABA or LAMA treatment strata. Subjects in the study cohort were followed for up to one year from cohort entry, with follow-up ending at death, 31 March 2021, or the end of the patient’s registration in the practice, whichever occurred first.
Outcome Events
The main outcome was the occurrence of a moderate or severe exacerbation of COPD, while severe exacerbation of COPD, severe pneumonia and all-cause mortality were also identified, all during the one-year follow-up. A moderate exacerbation was defined by a new prescription for prednisolone, while a severe exacerbation was defined as a hospitalization for COPD (ICD-10: J41, J42, J43, J44). Severe pneumonia was defined as a hospitalization for community-acquired pneumonia (ICD10: J10.0; J11.0; J12-J18; J22; J69; J85.0; J85.1; J86). These diagnostic codes have been shown to have good accuracy and were used in several studies of COPD using the CPRD.16,17,20–22
Covariates
The covariates used to compute the propensity score of treatment initiation included several factors identified using lifestyle, clinical diagnoses, and prescriptions from CPRD and HES data. Age, sex, body mass index (BMI), smoking status and alcohol abuse were measured at or prior to cohort entry. The severity of COPD at treatment initiation was assessed by the number of prior moderate and severe COPD exacerbations, and the use of other respiratory drugs, during the baseline period, as well as by the percent predicted FEV1. A moderate exacerbation in the baseline year was defined by a new prescription for prednisolone. Other respiratory drugs included the number of prescriptions for short-acting inhaled beta-agonists and anticholinergics, methylxanthines, and antibiotics used for respiratory conditions. Prior use of a LABA or LAMA, alone or together, was matched on to satisfy the adaptive selection design. The most recent measures of dyspnea, FEV1 and blood eosinophil count prior to cohort entry, were identified. Dyspnea was measured by mMRC, CAT score or the presence of dyspnea symptoms.23 The percent predicted FEV1 measurement, generally post-bronchodilator,24 was calculated from the absolute FEV1 value using age, sex and height, with race imputed as “Caucasian” for all.25 Baseline co-morbidity in the year prior to cohort entry was measured using clinical diagnoses, hospitalizations and prescriptions (Table 1).
Table 1.
Baseline Characteristics of the Study Cohort of 29,702 Initiators of Single-Inhaler Dual Bronchodilator Therapy and 4106 Initiators of Single-Inhaler Triple Therapy, After Fine Stratification Weighting from Probability of Treatment Propensity Scores, with Corresponding Standardized Mean Differences
| Single-Inhaler Dual Bronchodilator | Single-Inhaler Triple Therapy | Standardized Mean Difference | |
|---|---|---|---|
| Number of patients | 29,702 | 4106 | |
| Age at cohort entry, mean (sd) | 70.1 (10.6) | 70.2 (10.9) | 0.0049 |
| Female sex, n (%) | 13,399 (45.1) | 1808 (44.0) | −0.0218 |
| Current smoker, n (%) | 16,189 (54.5) | 2226 (54.2) | −0.0057 |
| Obesity Status, n (%) | |||
| Obese | 8500 (28.6) | 1171 (28.5) | −0.0021 |
| Non-Obese | 19,396 (65.3) | 2674 (65.1) | −0.0039 |
| Missing data | 1806 (6.1) | 261 (6.4) | 0.0118 |
| Alcohol Abuse, n (%) | 830 (2.8) | 125 (3.0) | 0.015 |
| Blood eosinophils cells/µL*, mean (sd) | 227.7 (179.1) | 233.0 (183.5) | 0.029 |
| Blood eosinophil count %*, mean (sd) | 3.0 (2.1) | 3.1 (2.3) | 0.0469 |
| FEV1 (% predicted)**, mean (sd) | 60.0 (17.9) | 59.8 (18.6) | -0.0101 |
| FEV1/FVC†, mean (sd) | 60.6 (13.5) | 61.4 (14.2) | 0.0579 |
| Severity of dyspnea | |||
| None-Mild | 10,501 (35.4) | 1470 (35.8) | 0.0094 |
| Moderate-Severe | 14,377 (48.4) | 1967 (47.9) | −0.0100 |
| Missing | 4824 (16.2) | 669 (16.3) | 0.0013 |
| Respiratory events and medications in year prior to cohort entry, n (%) | |||
| Hospitalization for COPD | 2350 (7.9) | 340 (8.3) | 0.0131 |
| Moderate/severe COPD exacerbation | |||
| None | 18,001 (60.6) | 2480 (60.4) | −0.004 |
| One | 6993 (23.5) | 979 (23.9) | 0.0073 |
| Two | 2541 (8.6) | 351 (8.6) | 0.0001 |
| Three or more | 2167 (7.3) | 295 (7.2) | −0.0044 |
| Asthma diagnosis | 2306 (7.8) | 343 (8.3) | 0.0215 |
| Pneumonia hospitalization | 2083 (7.0) | 314 (7.6) | 0.0241 |
| LAMA or LABA (no ICS)‡ | |||
| None | 12,340 (41.5) | 1799 (43.8) | |
| Any or both | 17,362 (58.5) | 2307 (56.2) | |
| Short-acting beta-agonists | 23,996 (80.8) | 3312 (80.7) | −0.0034 |
| Short-acting anti muscarinic | 665 (2.2) | 94 (2.3) | 0.0026 |
| Prednisolone | 10,490 (35.3) | 1447 (35.2) | −0.0015 |
| Methylxanthines | 102 (0.3) | 16 (0.4) | 0.0061 |
| Respiratory antibiotics | 17,883 (60.2) | 2483 (60.5) | 0.0057 |
| Comorbidity in year prior to cohort entry, n (%) | |||
| Cancer | 2211 (7.4) | 306 (7.5) | 0.0007 |
| Heart failure | 3161 (10.6) | 456 (11.1) | 0.0146 |
| Myocardial infarction (hospitalized) | 296 (1.0) | 42 (1.0) | 0.0016 |
| Stroke (hospitalized) | 738 (2.5) | 112 (2.7) | 0.0155 |
| Renal disease | 2215 (7.5) | 302 (7.4) | −0.0040 |
| Diabetes | 7602 (25.6) | 1044 (25.4) | −0.0041 |
| Other medication use in year prior to cohort entry, n (%) | |||
| ACE-inhibitors | 7865 (26.5) | 1127 (27.4) | 0.0218 |
| ARBs | 3292 (11.1) | 468 (11.4) | 0.0102 |
| Beta-blockers | 7865 (26.5) | 1097 (26.7) | 0.0055 |
| Calcium-channel blockers | 8151 (27.4) | 1121 (27.3) | −0.0029 |
| Thiazide diuretics | 2737 (9.2) | 385 (9.4) | 0.0058 |
| Antiarrhythmics | 562 (1.9) | 82 (2.0) | 0.0075 |
| Aspirin | 6213 (20.9) | 867 (21.1) | 0.0048 |
| Oral anticoagulant | 3927 (13.2) | 581 (14.1) | 0.0268 |
| Statins | 14,924 (50.2) | 2098 (51.1) | 0.0172 |
| PPIs | 14,024 (47.2) | 1914 (46.6) | −0.0121 |
| NSAIDs | 3132 (10.5) | 422 (10.3) | −0.0085 |
| Opioids | 11,088 (37.3) | 1523 (37.1) | −0.0051 |
Notes: *Based on available data from 86% for EOS count (cells/uL) and 86% for EOS count (%). ** Based on available data from 63% of subjects. † Based on available data from 45% of subjects. ‡ Stratification factor, not weighted. Propensity scores computed separately for each stratum.
Data Analysis
The propensity score of treatment initiation was estimated by logistic regression using all covariates, separately for the prior LAMA or LABA treatment strata. Fine stratification weights were computed using 100 strata defined by the propensity score to create comparable treatment arms with the same covariate distribution as the study cohort, within each prior treatment stratum.26 Standardized mean differences of covariates were computed to assess the comparability of the two treatment groups. Rates of outcomes were computed crudely and weighted, after stratification by prior treatment. A weighted Cox proportional hazard regression with a robust variance estimator, with stratification by prior treatment strata, was used in an as-treated analysis to compare triple with dual therapy on the risks of COPD exacerbation, death, and severe pneumonia during the first year after treatment initiation. Continuing exposure was measured by successive prescriptions of the initial single-inhaler treatment with a maximal 60-day gap between the prescription dates, with current use exposure ending 60 days after the last such prescription date. The comparative analysis was stratified by the number of exacerbations during the baseline year, by prior asthma diagnosis, and by baseline blood eosinophil counts as <150, 150–300, and >300 cells/µL, using the weighted regression model with an interaction term between these factors and the treatment.
Sensitivity analyses included an intention-to-treat analysis over the one-year follow-up. The definition of continuous use for the as-treated analyses was evaluated by varying the prescription gap to 30 and 90 days. The analysis was stratified by the presence of prior LAMA or LABA treatment. The analysis was also restricted to the subgroup of users of the single-inhaler combinations used in the IMPACT trial (fluticasone furoate-vilanterol-umeclidinium versus dual vilanterol-umeclidinium) and in the TRIBUTE trial (beclomethasone-formoterol-glycopyrronium versus indacaterol-glycopyrronium).27 Finally, we used an alternative definition for a moderate or severe exacerbation that has been validated in the CPRD.28 It is defined by an outpatient or inpatient diagnosis of lower respiratory tract infection (LRTI) or acute exacerbation of COPD (AECOPD), or a prescription of COPD-specific antibiotics combined with an oral corticosteroid, or a record of two or more respiratory symptoms of acute exacerbation of COPD along with a prescription of COPD-specific antibiotics and/or an oral corticosteroid on the same day. All analyses were conducted using SAS version 9.4. The study protocol was approved by the Independent Scientific Advisory Committee of the CPRD (Protocol 21_000491) and the Ethics Committee of the Jewish General Hospital (JGH Protocol #17-137), Montreal, Quebec, Canada.
Results
The study cohort included 4122 new users of triple therapy and 29,779 new users of dual bronchodilators, after excluding patients with no COPD diagnosis, users of ICS prior to treatment initiation, and the few patients (1259) contributed by the GOLD database (Figure 1). As there were only 245 new users of triple therapy that had used both a LABA and LAMA (in separate inhalers) in the baseline year, we regrouped the three baseline long-acting bronchodilator groups into two categories, any use or no use of LABA or LAMA. Thus, propensity scores were computed separately for these two strata only. The baseline characteristics, comparing initiators of single-inhaler triple therapy with initiators of dual bronchodilators after weighting by fine stratification of propensity scores and averaging over the two strata, show that the two groups are well-balanced (Table 1). The unweighted baseline characteristics of the two treatment groups are provided in Table S1. Of note, the unweighted data show that single-inhaler triple therapy was the very first maintenance treatment in 44% of the patients who used this inhaler, while 43% of triple therapy users had no COPD exacerbation in the year prior to initiation (Table S1). The adjusted characteristics, after weighting by fine stratification of propensity scores, stratified by prior use of a LABA or LAMA, are provided in Tables S2 and S3.
Figure 1.
Flowchart of cohort selection.
The single-inhaler triple therapy group included beclomethasone dipropionate-formoterol-glycopyrronium (57%) and fluticasone furoate-vilanterol-umeclidinium (43%), while the single-inhaler dual bronchodilators included vilanterol-umeclidinium (51%), olodaterol-tiotropium (19%), indacaterol-glycopyrronium (17%) and formoterol-aclidinium (13%). The mean duration of continuous treatment was 6 months in each of the two arms, with 4.8% of the dual bronchodilator patients switching to a triple inhaler during the one-year follow-up and 1.8% doing the reverse.
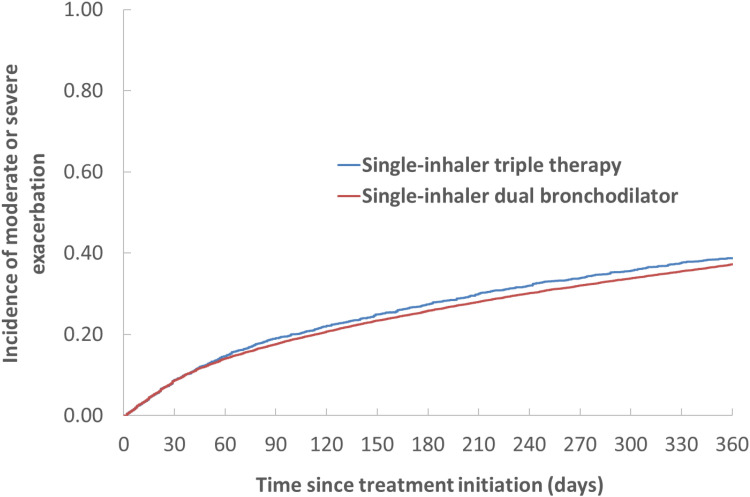
The cumulative incidence of a first moderate or severe exacerbation over 1 year was approximately 40% for the two treatment groups (Figure 2). The corresponding adjusted hazard ratio (HR) of a first moderate or severe exacerbation associated with triple therapy relative to dual bronchodilators was 1.08 (95% confidence interval (CI): 1.00–1.16) (Table 2). The HR of a severe exacerbation with triple therapy was 1.32 (95% CI: 1.13–1.55), while for all-cause mortality it was 1.53 (95% CI: 1.30–1.79) and for severe pneumonia 1.50 (95% CI: 1.29–1.75), relative to dual bronchodilators (Table 2).
Figure 2.
Kaplan–Meier curves for the one-year cumulative incidence of the first moderate or severe COPD exacerbation comparing single-inhaler triple therapy with single-inhaler dual bronchodilators in patients with COPD in the first year after treatment initiation, from the as-treated analysis, after adjustment by inverse probability of treatment weights.
Table 2.
Adjusted Hazard Ratios of COPD Exacerbation, Mortality and Severe Pneumonia Comparing Single-Inhaler Triple Therapy with Single-Inhaler Dual Bronchodilator Therapy in Patients with COPD in the First Year After Treatment Initiation, from the as-Treated Analysis
| Number of Patients | Number with Events | Person-Years | Rate* Per 100 Per Year | Rate† Per 100 Per Year | Adjusted† HR (95% CI) | |
|---|---|---|---|---|---|---|
| Moderate or severe exacerbation | ||||||
| Triple therapy | 4106 | 1304 | 1494 | 86.6 | 64.8 | 1.08 (1.00–1.16) |
| Dual bronchodilator | 29,702 | 7030 | 12,344 | 56.1 | 59.1 | 1.00 (Reference) |
| Severe exacerbation | ||||||
| Triple therapy | 4106 | 283 | 1961 | 14.4 | 10.1 | 1.32 (1.13–1.55) |
| Dual bronchodilator | 29,702 | 1029 | 14,739 | 7.0 | 7.6 | 1.00 (Reference) |
| All-cause mortality | ||||||
| Triple therapy | 4106 | 275 | 2034 | 13.3 | 9.6 | 1.53 (1.30–1.79) |
| Dual bronchodilator | 29,702 | 900 | 14,970 | 6.0 | 6.3 | 1.00 (Reference) |
| Severe pneumonia | ||||||
| Triple therapy | 4106 | 317 | 1966 | 15.9 | 11.7 | 1.50 (1.29–1.75) |
| Dual bronchodilator | 29,702 | 1079 | 14,740 | 7.3 | 7.8 | 1.00 (Reference) |
Notes: *Crude, computed before weighing by fine stratification of propensity scores. † After weighting by fine stratification weights from the probability of treatment propensity scores, stratified by prior use of LAMA or LABA.
Stratification by patient phenotypes shows that single-inhaler triple therapy is associated with a 17% lower incidence of moderate or severe exacerbation among patients with two or more exacerbations in the year prior to cohort entry (HR 0.83; 95% CI: 0.74–0.92), but a higher incidence among patients with one or no prior exacerbations (Table 3). A similar trend is observed for patients with a prior diagnosis of asthma (HR 0.86; 95% CI: 0.70–1.06) and those with a blood eosinophil count >300 cells/µL (HR 0.89; 95% CI: 0.76–1.05), albeit not reaching statistical significance. In contrast, the incidence is elevated with triple therapy in patients with no prior asthma diagnosis or those with blood eosinophil count <=300 cells/µL (Table 3).
Table 3.
Adjusted Hazard Ratios of a Moderate or Severe COPD Exacerbation Comparing Single-Inhaler Triple Therapy with Single-Inhaler Dual Bronchodilator Therapy in Patients with COPD in the First Year After Treatment Initiation, from the as-Treated Analyses Over One-Year Follow-Up, Stratified by Number of Exacerbations in the Prior Year, Prior Asthma, and Baseline Peripheral Blood Eosinophil Count
| Number of Patients | Number with Events | Person-Years | Rate* Per 100 Per Year | Rate† Per 100 Per Year | Adjusted† HR (95% CI) | |
|---|---|---|---|---|---|---|
| Overall | ||||||
| Triple therapy | 4106 | 1304 | 1494 | 86.6 | 64.8 | 1.08 (1.00–1.16) |
| Dual bronchodilator | 29,702 | 7030 | 12,344 | 56.1 | 59.1 | 1.00 (Reference) |
| COPD exacerbations in prior year | ||||||
| None | ||||||
| Triple therapy | 1754 | 271 | 731 | 37.2 | 34.8 | 1.19 (1.02–1.39) |
| Dual bronchodilator | 18,738 | 2435 | 8551 | 28.5 | 28.7 | 1.00 (Reference) |
| One | ||||||
| Triple therapy | 1222 | 430 | 434 | 98.7 | 98.4 | 1.17 (1.04–1.32) |
| Dual bronchodilator | 6731 | 2174 | 2620 | 82.1 | 82.4 | 1.00 (Reference) |
| Two or more | ||||||
| Triple therapy | 1130 | 603 | 330 | 182.9 | 169.1 | 0.83 (0.74–0.92) |
| Dual bronchodilator | 4233 | 2421 | 1174 | 206.2 | 209.4 | 1.00 (Reference) |
| Prior asthma diagnosis | ||||||
| None | ||||||
| Triple therapy | 3553 | 1129 | 1296 | 86.4 | 65.2 | 1.10 (1.01–1.19) |
| Dual bronchodilator | 27,630 | 6493 | 11,534 | 55.5 | 58.1 | 1.00 (Reference) |
| Yes | ||||||
| Triple therapy | 553 | 175 | 198 | 88.1 | 62.3 | 0.86 (0.70–1.06) |
| Dual bronchodilator | 2072 | 537 | 810 | 66.1 | 72.9 | 1.00 (Reference) |
| Baseline blood eosinophil count (cells/µL) | ||||||
| < 150 | ||||||
| Triple therapy | 1191 | 382 | 412 | 92.2 | 67.6 | 1.12 (0.98–1.29) |
| Dual bronchodilator | 9445 | 2201 | 3921 | 55.6 | 58.6 | 1.00 (Reference) |
| 150–300 | ||||||
| Triple therapy | 1501 | 472 | 539 | 86.6 | 67.2 | 1.13 (0.99–1.28) |
| Dual bronchodilator | 11,285 | 2644 | 4722 | 54.9 | 57.5 | 1.00 (Reference) |
| >300 | ||||||
| Triple therapy | 799 | 274 | 305 | 88.4 | 62.5 | 0.89 (0.76–1.05) |
| Dual bronchodilator | 4764 | 1314 | 1900 | 68.1 | 71.9 | 1.00 (Reference) |
Notes: *Crude, computed before weighing by fine stratification of propensity scores. †After weighting by fine stratification weights from the probability of treatment propensity scores, stratified by prior use of LAMA or LABA.
Sensitivity analyses mainly confirmed the robustness of the results (Table 4). As expected, the HR was slightly attenuated in the intention-to-treat analysis over the one-year follow-up (HR 1.05; 95% CI: 0.98–1.12). Using the alternative definition of exacerbation and varying the gap between prescriptions to define continuous treatment did not significantly alter the main result. The stratified analysis by whether the single inhalers were used de novo or stepped up from a LAMA or LABA did not affect the findings. Finally, the analysis restricted to the subgroup of users of the single-inhaler combinations used in the IMPACT trial (fluticasone furoate-vilanterol-umeclidinium versus vilanterol-umeclidinium) and in the TRIBUTE trial (beclomethasone-formoterol-glycopyrronium versus indacaterol-glycopyrronium) produced consistent findings.
Table 4.
Adjusted Hazard Ratios of a Moderate or Severe COPD Exacerbation Comparing Single-Inhaler Triple Therapy with Single-Inhaler Dual Bronchodilator Therapy in Patients with COPD in the First Year After Treatment Initiation, from the as-Treated Sensitivity Analyses
| Number of Patients | Number with Events | Person-Years | Rate* Per 100 Per Year | Rate† Per 100 per Year | Adjusted† HR (95% CI) | |
|---|---|---|---|---|---|---|
| Primary as-treated analysis | ||||||
| Triple therapy | 4106 | 1304 | 1494 | 86.6 | 64.8 | 1.08 (1.00–1.16) |
| Dual broncho | 29,702 | 7030 | 12,344 | 56.1 | 59.1 | 1.00 (Reference) |
| Intent-to-treat analysis (one year) | ||||||
| Triple therapy | 4106 | 1684 | 2279 | 73.8 | 53.6 | 1.05 (0.98–1.12) |
| Dual bronchodilator | 29,702 | 9566 | 19,577 | 48.4 | 50.8 | 1.00 (Reference) |
| Using validated definition of COPD exacerbation‡ | ||||||
| Triple therapy | 4106 | 1339 | 1499 | 88.7 | 69.8 | 1.16 (1.07–1.25) |
| Dual bronchodilator | 29,702 | 7110 | 12,356 | 56.9 | 59.5 | 1.00 (Reference) |
| Continuous use defined by 30-day gap | ||||||
| Triple therapy | 4106 | 727 | 607 | 119.1 | 87.4 | 1.05 (0.95–1.16) |
| Dual bronchodilator | 29,702 | 3658 | 4633 | 78.1 | 83.0 | 1.00 (Reference) |
| Continuous use defined by 90-day gap | ||||||
| Triple therapy | 4106 | 1487 | 1832 | 80.7 | 59.1 | 1.06 (0.99–1.14) |
| Dual bronchodilator | 29,702 | 8116 | 15,333 | 52.3 | 55.0 | 1.00 (Reference) |
| Initial treatment, no prior LABA or LAMA | ||||||
| Triple therapy | 1799 | 478 | 635 | 75.2 | 58.0 | 1.12 (0.99–1.28) |
| Dual bronchodilator | 12,340 | 2356 | 4921 | 47.8 | 50.3 | 1.00 (Reference) |
| Treatment stepped up from LABA or LAMA | ||||||
| Triple therapy | 2307 | 826 | 859 | 96.1 | 70.7 | 1.05 (0.96–1.15) |
| Dual bronchodilator | 17,362 | 4674 | 7423 | 62.9 | 66.4 | 1.00 (Reference) |
| Fluticasone furoate-vilanterol-umeclidinium versus vilanterol-umeclidinium | ||||||
| Triple therapy | 1757 | 525 | 690 | 74.8 | 59.5 | 1.08 (0.96–1.21) |
| Dual bronchodilator | 15,224 | 3303 | 6408 | 51.7 | 54.5 | 1.00 (Reference) |
| Beclomethasone-formoterol-glycopyrronium versus indacaterol-glycopyrronium | ||||||
| Triple therapy | 2349 | 779 | 805 | 97.0 | 69.9 | 1.08 (0.97–1.21) |
| Dual bronchodilator | 5059 | 1297 | 2130 | 58.9 | 62.4 | 1.00 (Reference) |
Notes: *Crude, computed before weighing by fine stratification of propensity scores. †After weighting by fine stratification weights from the probability of treatment propensity scores, stratified by prior use of LAMA or LABA. ‡COPD exacerbation defined as an outpatient or inpatient diagnosis of LRTI or AECOPD, or a prescription of COPD-specific antibiotic combined with OCS, or a record of two or more respiratory symptoms of AECOPD along with a prescription of COPD-specific antibiotics and/or OCS on the same day, as per Rothnie et al.28
Discussion
This first real-world observational study of single-inhaler triple therapy in COPD suggests that patients prescribed triple therapy had a similar incidence of moderate or severe exacerbation over the first year of use as those treated with a single-inhaler dual bronchodilator. However, the incidence was significantly lower with triple therapy for the subgroup of patients with two or more exacerbations in the year prior to treatment initiation and numerically lower for patients with a prior diagnosis of asthma and those with an elevated blood eosinophil count (>300 cells/µL). On the other hand, the incidence of a COPD exacerbation was slightly higher with triple therapy for those patients who did not have these three phenotypes. The incidence of severe exacerbation and severe pneumonia requiring hospitalization, and all-cause mortality, was higher with the single-inhaler triple combination compared with a dual bronchodilator.
The findings of our observational study differ from those reported in the large IMPACT and ETHOS randomized trials that found significant reductions in the incidence of moderate or severe exacerbation with single-inhaler triple therapy compared with dual bronchodilators.2,3 Besides the observational nature of our study, several reasons can explain these differences. First, the patients enrolled in these trials included 40% who were already treated with triple therapy and 70–80% whose treatment involved ICS. The abrupt withdrawal of prior ICS treatment at randomization imposed by the trials’ design can bias the results.29 Second, the trials included COPD patients with a history of asthma and with multiple COPD exacerbations, patients for whom ICS are indicated and should not be withdrawn. Together, these two issues will distort the results.6–8 On the other hand, our real-world study population was restricted to patients not previously treated with an ICS, thus avoiding the confounding effects of abruptly withdrawing prior ICS use. Thus, while the effectiveness with triple therapy compared with dual bronchodilators reported in the trials can be in great part attributed to the effect of abrupt ICS withdrawal, our observational study avoids this issue by its use of an adaptive selection design.
While the IMPACT and ETHOS trials reported significant overall reductions in all-cause mortality comparing single-inhaler triple therapy with dual bronchodilators, with HRs of 0.72 (95% CI: 0.53–0.99) and 0.51 (95% CI: 0.33–0.80), respectively, this effect was observed exclusively in patients who had to discontinue ICS at randomization. Indeed, among non-users of ICS prior to randomization, the HRs of all-cause mortality comparing triple therapy with dual bronchodilators were 1.25 (95% CI: 0.60–2.59) in IMPACT and 1.49 (95% CI: 0.49–4.55) in ETHOS.4,5 These estimates, unimpacted by the effect of abrupt ICS withdrawal at randomization, are more in line with the corresponding HR of all-cause mortality of 1.53 (95% CI: 1.30–1.79) found in our observational study that excluded patients already treated with ICS.
Our study identified several noteworthy and non-recommended patterns of triple therapy prescribing in this real-world clinical practice setting. First, single-inhaler triple therapy was the very first maintenance treatment in 44% of the patients who used this inhaler, with no LABA, LAMA or ICS in the year prior to initiation. This is rather discordant with the GOLD evidence-based recommendations for the initial treatment of COPD, which advocate mainly a LAMA, a LABA, their combination, or a LABA-ICS for patients with eosinophilia, but not triple therapy.1 Second, 43% of triple therapy users had no COPD exacerbation in the year prior to initiation and would not have been eligible for the IMPACT and ETHOS trials. In addition, 31% had no or mild dyspnea, and 81% had a blood eosinophil count <300 cells/µL. While triple therapy is not generally recommended for these patient profiles, there are no randomized controlled data on the impact of this treatment in the many such patients prescribed triple therapy in our real-world clinical setting. Indeed, the trials of triple therapy included only patients with a history of exacerbations, with moderate or severe dyspnea, and with long-standing disease.
Our stratified analyses are quite revealing with respect to the treatment for these non-recommended patient profiles. Indeed, single-inhaler triple therapy was associated with a significantly lower incidence of moderate or severe exacerbation among patients with two or more exacerbations in the year prior to starting treatment, as per recommendations, but a higher incidence among patients with less than two prior exacerbations. Similarly, for patients with a prior diagnosis of asthma and those with a blood eosinophil count >300 cells/µL, the incidence of exacerbation was also lower with triple therapy, though not reaching statistical significance.
We found a 50% increased incidence of severe pneumonia requiring hospitalization with triple therapy compared with dual bronchodilators. This is consistent with the 65% and 78% increases in serious pneumonias with triple therapy reported in the IMPACT and ETHOS trials, respectively.2,3
Our study has several strengths and limitations. First, the new-user design identified study subjects at the time of initiation of the single-inhaler triple and dual bronchodilator therapies, thus emulating the randomized trial approach in an observational setting, albeit without randomization. Second, our adaptive selection study design excluded patients treated with ICS that made up 70-80% of the patients in the IMPACT and ETHOS trials, including 40% already using triple therapy. Consequently, our design avoided the confounding effect of prior ICS use that affected the two randomized trials. Third, the observational setting of general clinical practice provides real-world evidence on the effects of these treatments. Fourth, several of the outcome events used validated definitions, including hospitalization and death records, and thus less subject to misclassification errors.15,16 The study has limitations. First, the treatment exposure measures are based on written prescriptions, not necessarily dispensed, which could introduce some misclassification, potentially compounded by the less-than-optimal inhaler technique in the real-world setting. Second, the continuous treatment duration was relatively short (mean 6 months), which reflects the real-world clinical situation, though the intent-to-treat analysis which used the entire one-year follow-up supported the findings of the as-treated analysis. Third, with the 40 years of age cut-off, the use of physician diagnoses to identify the cohort of patients with COPD may have misclassified some patients with asthma as COPD, which would anyhow tend to favor the triple inhaler. However, the physician diagnosis of COPD was validated and found to be accurate, with spirometry only marginally improving accuracy.15 Fourth, while large, the study size is limited by the number of new users of single-inhaler triple therapy (4106) who did not use ICS previously, thus limiting the power of stratified analyses, particularly for the less common outcomes of severe exacerbation and all-cause mortality. Fifth, the study involved two different triple inhalers and four dual bronchodilators, which could have different effects on the outcome. However, the analysis restricted to the single-inhaler combinations studied in the IMPACT and TRIBUTE trials produced consistent findings. Finally, residual confounding cannot be ruled out in any observational study, despite the use of propensity score weighting which created groups highly comparable on all available measures of patient characteristics, including COPD severity.
Conclusion
While randomized controlled trials provide evidence for the effectiveness of treatments for COPD, observational studies are now receiving special attention in providing complementary real-world evidence (RWE) for regulatory decision-making.30–32 This observational study, conducted in a real-world clinical practice setting of COPD treatment among ICS-naïve patients, using an adaptive selection design, found that single-inhaler triple therapy was not more effective than dual bronchodilators at reducing the incidence of exacerbation, except among patients with multiple exacerbations. Thus, single-inhaler triple therapy should be mainly reserved for patients with multiple exacerbations and likely those with asthma and eosonophilia while, for most others, dual bronchodilators are just as effective whilst circumventing the excess risk of severe pneumonias with triple therapy. These latter patients, for whom the GOLD reports do not recommend triple therapy, made up a large proportion of the patients prescribed single-inhaler triple therapy in this real-world clinical practice setting.
Funding Statement
This study is not funded. Pr. Suissa is the recipient of the Distinguished James McGill Professorship award.
Abbreviation
AECOPD, acute exacerbation of COPD; CI, confidence interval; COPD, chronic obstructive pulmonary disease; CPRD, Clinical Practice Research Datalink; ETHOS, Efficacy and Safety of Triple Therapy in Obstructive Lung Disease; HES, Hospital Episodes Statistics; HR, hazard ratio; ICS, inhaled corticosteroids; LAMA, long-acting muscarinic antagonists; LABA, long-acting beta2-agonists; LRTI, lower respiratory tract infection; IMPACT, Informing the Pathway of COPD Treatment; UK, United Kingdom; sd, standard deviation.
Data Sharing Statement
This study is based in part on data from the Clinical Practice Research Datalink obtained under license from the UK Medicines and Healthcare products Regulatory Agency. The data are provided by patients and collected by the UK National Health Service as part of their care and support. The interpretation and conclusions contained in this study are those of the author/s alone. Because electronic health records are classified as “sensitive data” by the UK Data Protection Act, information governance restrictions (to protect patient confidentiality) prevent data sharing via public deposition. Data are available with approval through the individual constituent entities controlling access to the data. Specifically, the primary care data can be requested via application to the Clinical Practice Research Datalink (https://www.cprd.com).
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
SS attended scientific advisory committee meetings or consulted for AstraZeneca, Atara, Bristol-Myers-Squibb, Merck, Novartis, Panalgo, Pfizer and Seqirus, and received speaking fees from AstraZeneca, Boehringer-Ingelheim and Novartis. Dr. Ernst and Ms Dell’Aniello have no conflicts of interest.
References
- 1.Vogelmeier CF, Criner GJ, Martinez FJ., et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 Report: GOLD Executive Summary. Eur Respir J. 2017;49(3):548. doi: 10.1183/13993003.00214-2017 [DOI] [PubMed] [Google Scholar]
- 2.Lipson DA, Barnhart F, Brealey N, et al. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378(18):1671–1680. doi: 10.1056/NEJMoa1713901 [DOI] [PubMed] [Google Scholar]
- 3.Rabe KF, Martinez FJ, Ferguson GT, et al. Triple inhaled therapy at two glucocorticoid doses in moderate-to-very-severe COPD. N Engl J Med. 2020;383(1):35–48. doi: 10.1056/NEJMoa1916046 [DOI] [PubMed] [Google Scholar]
- 4.Lipson DA, Crim C, Criner GJ, et al. Reduction in all-cause mortality with fluticasone furoate/umeclidinium/vilanterol in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2020;201(12):1508–1516. doi: 10.1164/rccm.201911-2207OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Martinez FJ, Rabe KF, Ferguson GT, et al. Reduced all-cause mortality in the ethos trial of budesonide/glycopyrrolate/formoterol for chronic obstructive pulmonary disease. a randomized, double-blind, multicenter, parallel-group study. Am J Respir Crit Care Med. 2021;203(5):553–564. doi: 10.1164/rccm.202006-2618OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Suissa S, Drazen JM. Making sense of triple inhaled therapy for COPD. N Engl J Med. 2018;378(18):1723–1724. doi: 10.1056/NEJMe1716802 [DOI] [PubMed] [Google Scholar]
- 7.Suissa S, Ariel A. Triple therapy trials in COPD: a precision medicine opportunity. Eur Respir J. 2018;52(6):1801848. doi: 10.1183/13993003.01848-2018 [DOI] [PubMed] [Google Scholar]
- 8.Wedzicha JA, Banerji D, Kostikas K. Single-inhaler triple versus dual therapy in patients with COPD. N Eng J Med. 2018;379(6):590–593. [DOI] [PubMed] [Google Scholar]
- 9.Suissa S. Perplexing mortality data from triple therapy trials in COPD. Lancet Respir Med. 2021;9(7):684–685. doi: 10.1016/S2213-2600(21)00238-1 [DOI] [PubMed] [Google Scholar]
- 10.Han MK, Dransfield MT, Dransfield MT, et al. The effect of ICS withdrawal and baseline inhaled treatment on exacerbations in the IMPACT study. Am J Respir Crit Care Med. 2020;202(9):1237–1243. doi: 10.1164/rccm.201912-2478OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Suissa S. Triple Therapy in COPD: time for adaptive selection trials. COPD. 2021;18(6):597–601. doi: 10.1080/15412555.2021.1982886 [DOI] [PubMed] [Google Scholar]
- 12.Wolf A, Dedman D, Campbell J, et al. Data resource profile: Clinical Practice Research Datalink (CPRD) Aurum. Int J Epidemiol. 2019;48(6):1740–1740g. doi: 10.1093/ije/dyz034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Herrett E, Thomas SL, Schoonen WM, Smeeth L, Hall AJ. Validation and validity of diagnoses in the general practice research database: a systematic review. Br J Clin Pharmacol. 2010;69(1):4–14. doi: 10.1111/j.1365-2125.2009.03537.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Herrett E, Gallagher AM, Bhaskaran K, et al. Data Resource Profile: Clinical Practice Research Datalink (CPRD). Int J Epidemiol. 2015;44(3):827–836. doi: 10.1093/ije/dyv098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Quint JK, Mullerova H, DiSantostefano RL, et al. Validation of chronic obstructive pulmonary disease recording in the Clinical Practice Research Datalink (CPRD-GOLD). BMJ Open. 2014;4(7):e005540. doi: 10.1136/bmjopen-2014-005540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rothnie KJ, Müllerová H, Thomas SL, et al. Recording of hospitalizations for acute exacerbations of COPD in UK electronic health care records. Clin Epidemiol. 2016;8:771–782. doi: 10.2147/CLEP.S117867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Suissa S, Dell’Aniello S, Ernst P. Comparative effectiveness of LABA-ICS versus LAMA as initial treatment in COPD targeted by blood eosinophils: a population-based cohort study. Lancet Respir Med. 2018;6(11):855–862. doi: 10.1016/S2213-2600(18)30368-0 [DOI] [PubMed] [Google Scholar]
- 18.Johnson ES, Bartman BA, Briesacher BA, et al. The incident user design in comparative effectiveness research. Pharmacoepidemiol Drug Saf. 2013;22(1):1–6. doi: 10.1002/pds.3334 [DOI] [PubMed] [Google Scholar]
- 19.Suissa S, Moodie EE, Dell’Aniello S. Prevalent new-user cohort designs for comparative drug effect studies by time-conditional propensity scores. Pharmacoepidemiol Drug Saf. 2017;26(4):459–468. doi: 10.1002/pds.4107 [DOI] [PubMed] [Google Scholar]
- 20.Meropol SB, Metlay JP. Accuracy of pneumonia hospital admissions in a primary care electronic medical record database. Pharmacoepidemiol Drug Saf. 2012;21(6):659–665. doi: 10.1002/pds.3207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Suissa S, Dell’Aniello S, Ernst P. Comparative effectiveness and safety of LABA-LAMA vs LABA-ICS treatment of COPD in Real-world clinical practice. CHEST. 2019;155(6):1158–1165. doi: 10.1016/j.chest.2019.03.005 [DOI] [PubMed] [Google Scholar]
- 22.Suissa S, Dell’Aniello S, Ernst P. Comparative effectiveness of initial LAMA versus LABA in COPD: real-world cohort study. J Chronic Obstructive Pulmonary Dis. 2021;18(1):1–8. doi: 10.1080/15412555.2021.1877649 [DOI] [PubMed] [Google Scholar]
- 23.Rebordosa C, Plana E, Aguado J, et al. GOLD assessment of COPD severity in the Clinical Practice Research Datalink (CPRD). Pharmacoepidemiol Drug Saf. 2019;28(2):126–133. doi: 10.1002/pds.4448 [DOI] [PubMed] [Google Scholar]
- 24.Whittaker HR, Pimenta JM, Jarvis D, Kiddle SJ, Quint JK. Characteristics associated with accelerated lung function decline in a primary care population with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2020;15:3079–3091. doi: 10.2147/COPD.S278981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Quanjer PH, Stanojevic S, Cole TJ, et al. Multi-ethnic reference values for spirometry for the 3–95-yr age range: the global lung function 2012 equations. Eur Respir J. 2012;40(6):1324–1343. doi: 10.1183/09031936.00080312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Desai RJ, Franklin JM. Alternative approaches for confounding adjustment in observational studies using weighting based on the propensity score: a primer for practitioners. BMJ. 2019;367:l5657. doi: 10.1136/bmj.l5657 [DOI] [PubMed] [Google Scholar]
- 27.Papi A, Vestbo J, Fabbri L, et al. Extrafine inhaled triple therapy versus dual bronchodilator therapy in chronic obstructive pulmonary disease (TRIBUTE): a double-blind, parallel group, randomised controlled trial. Lancet. 2018;391(10125):1076–1084. doi: 10.1016/S0140-6736(18)30206-X [DOI] [PubMed] [Google Scholar]
- 28.Rothnie KJ, Müllerová H, Hurst JR, et al. Validation of the recording of acute exacerbations of COPD in UK primary care electronic healthcare records. PLoS One. 2016;11(3):e0151357. doi: 10.1371/journal.pone.0151357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Suissa S. Run-in bias in randomised trials: the case of COPD medications. Eur Respir J. 2017;49(6):1700361. doi: 10.1183/13993003.00361-2017 [DOI] [PubMed] [Google Scholar]
- 30.Sherman RE, Anderson SA, Dal Pan GJ, et al. Real-world evidence — what is it and what can it tell us? N Engl J Med. 2016;375(23):2293–2297. doi: 10.1056/NEJMsb1609216 [DOI] [PubMed] [Google Scholar]
- 31.Frieden TR, Drazen JM, Harrington DP, McMurray JJV, Ware JH, Woodcock J. Evidence for health decision making — beyond randomized, controlled trials. N Engl J Med. 2017;377(5):465–475. doi: 10.1056/NEJMra1614394 [DOI] [PubMed] [Google Scholar]
- 32.Eichler HG, Koenig F, Arlett P, et al. Are novel, nonrandomized analytic methods fit for decision making? The need for prospective, controlled, and transparent validation. Clin Pharmacol Ther. 2020;107(4):773–779. doi: 10.1002/cpt.1638 [DOI] [PMC free article] [PubMed] [Google Scholar]