Abstract
It is known that metabolic reprogramming (MR) contributes to tumorigenesis through the activation of processes that support survival of cells, proliferation, and grow in the tumor microenvironment. In order to keep the tumor proliferating at a high rate, metabolic pathways must be upregulated, and tumor metabolism must be adapted to meet this requirement. Additionally, immune cells engage in metabolic remodeling to maintain body and self-health. With the advent of immunotherapy, the fate of individuals suffering from non-small cell lung cancer (NSCLC) has been transformed dramatically. MR may have a profound influence on their prognosis. The aim of this review is to summarize current research advancements in metabolic reprogramming and their impact on immunotherapy in NSCLC. Moreover, we talk about promising approaches targeting and manipulating metabolic pathways to improve cancer immunotherapy’s effectiveness in NSCLC.
Keywords: Metabolic reprogramming, TME, NSCLC, Immunotherapy
Introduction
As the main reason for cancer-associated deaths, universally non-small cell lung cancer (NSCLC) has seen a remarkable increase in incidence rate for the past few years [1, 2]. According to histological and pathological classification, lung cancer is mainly divided into lung adenocarcinoma (LUAD), lung squamous cell carcinoma (LUSC), large cell carcinoma and other rare types [3]. With its low 5-year survival rate and high mortality rate, advanced non-small cell lung cancer keeps being a big challenge of oncology [4]. To date, treatment options available for NSCLC incorporate surgery, adjuvant therapy, chemotherapy, radiotherapy, and immunotherapy. Nevertheless, effective therapies of NSCLC, especially for advanced stage cancers, is still lacking [5]. Consequently, to create alternative therapies for such cancer is in urgent need. Compared with traditional treatments, cancer immunotherapy represented by PD-1 blockade has led to a model transition in cancer treatment owing to better survival, less side effects and wider scope of application [6, 7]. So far, two monoclonal antibodies blocking PD-1 (nivolumab, pembrolizumab) together with two blocking PD-L1 (atezolizumab, durvalumab) gain the approval from the FDA to be applied to first-line regimens without prior platinum-containing chemotherapy and for second-line regimens after failure of platinum-containing chemotherapy [8–11].
However, responses to immune checkpoint blockade (ICB) therapy are not widespread, with many patients displaying primary resistance to ICB monotherapy [12]. In some patients, ICB treatment may even result in immune activation against specific organs immunotherapeutic-induced adverse events (irAE). In general, irAEs colocalize with barrier tissues (gut, lungs, and skin) as well as with endocrine tissues (pancreas and thyroid), but importantly, the most common irAE differ from drug to drug [13, 14]. It can be challenging to use combined ICB in clinical settings due to its higher number of side effects. Studies are underway to develop effective approaches to minimize IRAEs without compromising anti-tumor immunity.
With further understanding of immunoregulatory mechanisms in the range of the tumor microenvironment (TME),the answers to these questions will emerge. A number of elements acting in the TME restrain the curative activities of ICBs. Various findings suggest that immune cells, like Myeloid-derived suppressor cells (MDSC), Tumor-associated macrophages (TAMs), Tumor associated dendritic cells (TADCs) have different potentials in predicting response to anti-PD-(L)1 therapy in patients with NSCLC, respectively. For instance, the immune response to immunotherapy was significantly improved when PMN-MDSC levels were high or the CD8/PMN-MDSC ratio was low [15]. While, TIM-3 expression on lymphocytes and early accumulation of M-MDSC with (Lin*CD33+ CD14 + CD15* HLA-DR*) is associated with resistance to PD-1 blockade [16]. Additionally, the ratio of circulating Treg to G-MDSCs may also affect the response to nivolumab, since patients with a high proportion of circulating Tregs and low proportion of G-MDSCs show improved PFS in patients with NSCLC [17]. In NSCLC, mature DCs are located exclusively within TLSs and are associated with a good prognosis [18]. High DC-LAMP1+ mature DCs and High stromal CD561 cells were correlated with prolonged survival in patients with NSCLC [19, 20]. At the same time, High CD1a1+LCs together with CD14+CD68low interstitial DCs were both associated with longer DSS [21].
Here, we review how the metabolic pattern in lung cancer is remodeled with transformation and malignant progression and how this gives rises to immune escape and resistance to ICBs. We also conclude how these discoveries are applied to strengthen the efficacy of ICBs in patients with NSCLC.
Glucose metabolism in NSCLC
Glucose metabolism of tumor cells
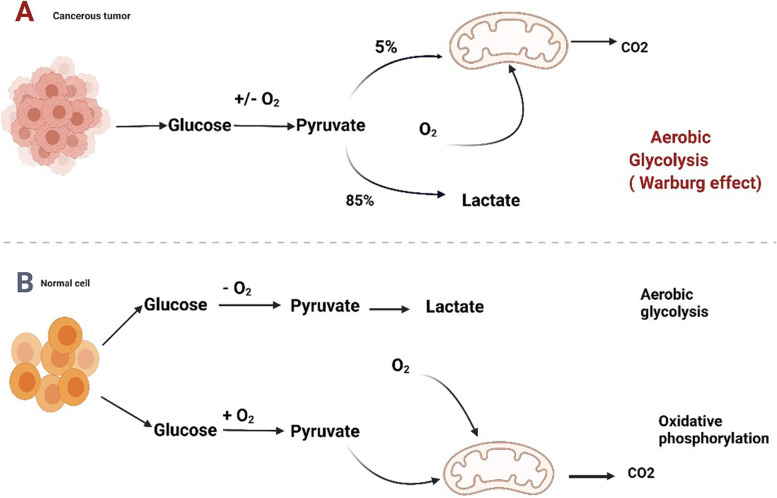
Otto Warburg’s pioneering work displayed in the 1920s that tumor cells consume more glucose than normal cells. The phenomenon went by the name of the aerobic glycolysis or Warburg effect (Fig. 1) [22]. With the physiological condition in oxygenated environment, glucose is metabolized by cells by the course of glycolysis, the tricarboxylic acid (TCA) cycle and oxidative phosphorylation (OXPHOS), and eventually molecular oxidation is completed. This process which is dependent on oxygen is known as oxidative phosphorylation, and 32–38 ATP molecules are ultimately produced from a glucose molecule. However, pyruvate, being converted to lactate, is the byproduct of glycolysis. Merely two molecules of ATP are contained in per mol of glucose. For cancer cells,this metabolic shift includes an upregulation of biosynthetic along with bioenergetic pathways in order to maintain high proliferative rate and adapt metabolism (shown in Fig. 2).
Fig. 1.
Metabolic control of glucose by cancer cells. Mitochondria are primarily involved in glycosis and the TCA cycle in glucose metabolism. The pathways in tumor cells are generally altered compared to those of normal cells. Figure created using BioRender.com
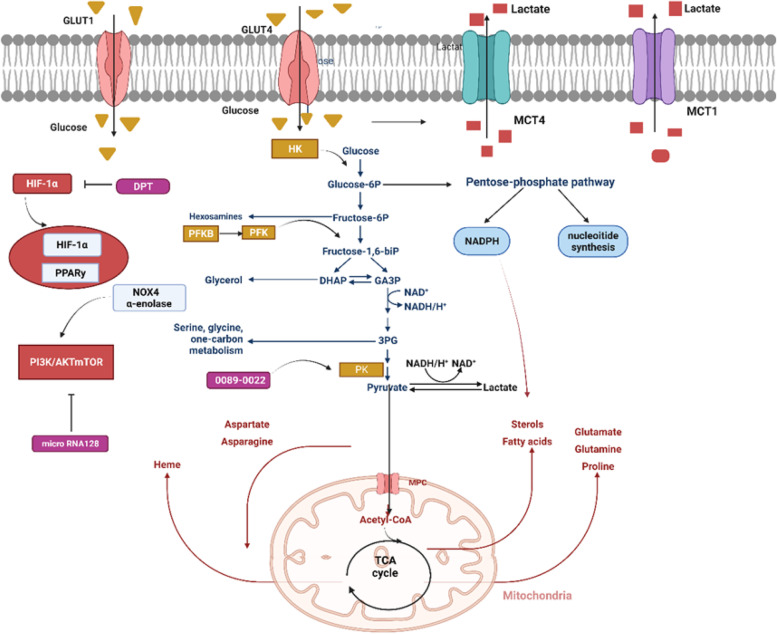
Fig. 2.
Reprogramming the metabolic process of glucose in lung cancer. In addition to HIF-1α pathway,PPARy and PI3K/AKT/mTOR signaling impair the activity of enzymes and transporters limiting metabolic reprogramming in NSCLC. In the cell, glucose is transported by glucose transporters (GLUT)1 and 4. Glycolysis begins with the phosphorylation of glucose by hexokinase (HK). In the cycle of pentose phosphate (PPP), glucose-6-phosphate is converted into nucleosides and NADPH by glucose-6-phosphate dehydrogenase (G6PD). Dephosphorylating PEP is accomplished by pyruvate kinase (PK) at the end of the glycolytic pathway to synthesize pyruvate and ATP. After that, pyruvate is turned into lactate by-lactate dehydrogenase (LDH). Lactate is delivered to the outside of the cell by monocarboxylate transporters (MCT) 1 and 4. During metabolism, pyruvate can be converted into acetyl coenzyme A (acetyl-CoA), which is following used by the tricarboxylic acid cycle in mitochondria to give rise to ATP and intermediate molecules that are essential to the biosynthesis of both lipids and amino acids. Figure created using BioRender.com
Effect of glucose metabolism in NSCLC
There is more and more evidence suggesting metabolic remodeling is activated deeply in carcinogenesis and malignant progression in lung cancer (LC) [23]. High glucose uptake is discovered in NSCLC via positron emission tomography/computed tomography (PET/CT). Patients who has NSCLC with high glucose uptake can be identified with glucose-analog fluorodeoxyglucose (18F-FDG) PET/CT scans, which is on the rise as a potential instrument to choose patients for metabolically targeted anti-tumor treatment [24]. fLC tissue illustrates growing glucose contribution to tricarboxylic acid cycle (TCA) cycle in comparison with normal lung tissue, and lung cancer cells embody diverse glycolysis rates and mitochondrial abilities [25]. In addition, in LC cells, pyruvate carboxylase (PC) and pyruvate were overexpressed compared to normal lung tissues. Even when tumor cells are cultured without glucose, activation of alternative pathways for phosphopentose (PPP) still acts a vital part in tumorigenesis [26]. Moreover, PPP activation can trigger a good deal of glutathione and nicotinamide adenine dinucleotide phosphate (NADPH), whose oxidase activity and expression are associated with malignant biological behavior of LC. The function of inhibiting NADPH oxidase downregulates the proliferation and invasion of LC [27, 28]. Thus, Lung cancer cells alter their metabolism by taking away glucose for energy production through glycolysis, generating biomass through PPP and deprotonation, and counteracting oxidative stress through PPP.
Regulation of glucose transport and metabolism in NSCLC
There are two pathways of cellular glycolysis: oxygen-dependent and oxygen-independent pathway. These two pathways both depend on some familiar glucose transporters and glycolytic enzymes (Fig. 2). The oxygen-dependent mechanisms are mediated by transcription factor hypoxia-inducible factor 1-alpha (HIF-1α) pathway [29]. It has been shown that hypoxia promotes glycogen accumulation in cells through HIF-1α stabilization [30]. The recent study showed DPT (Deoxypodophyllotoxin) serves as an anticancer agent in NSCLC by suppressing HIF-1α activation at the protein level in NSCLC cells to reduce glycolysis [31]. Another study demonstrates that AC020978’s part in advancing cell growth and metabolic reprogramming in NSCLC, which uncovered that AC020978 could regulate PKM2-enhanced HIF-1α transcription activity [32].
Activation of the PI3K-AKT-mTOR signaling pathway mainly mediates oxygen non-dependent mechanisms of glucose utilization in LC [33, 34]. From a previous report,50–73% of NSCLC patients with poor prognosis exhibit high expression of AKT [35], while only minor patients with NSCLC embody mutations of PI3K and AKT.
Aside from activation of mutations, other molecules could also foster PI3K/AKT/MTOR signaling in LC. For instance, microRNA128 (Mir-128) plays an inhibitory role in LC progression by way of inhibition of AKT expression,thereby down-regulating glycolysis [36]. However, some other molecules have been reported activate glycolysis metabolism of LC by targeting PI3K/AKT pathway, such as oxidase 4 (NOX4) and α-enolase [37, 38].
Glucose transporters (GLUTs) belong to a protein family which is beneficial to the transferring of glucose into the blood. Several studies have shown that higher level of GLUT-1 protein was spotted to be remarkably connected with resistance to radiotherapy and poor disease particular overall survival of lung cancer [39, 40]. However, another study performed by Osugi et al. indicated that NSCLC patients with GLUT-1 expression failed to independently display poorer overall survival in comparison to GLUT1-negative patients [41]. Furthermore, GLUT-4 was also identified to be an appropriate potential target for epigenetic treatment or metabolic targeting in the management and NSCLC therapy [42].
Hexokinase (HK) is the first rate-limiting enzyme when cells begin glycolysis. HK is composed of four isoforms featured with diverse functions along with cellular positions. HK-II serves as an enzyme which catalyzes the phosphorylation of glucose, and it is the first step of glycolytic rate [43]. The expression levels of the HK-II protein in aggressive cancer cells greatly exceed those in normal cells [44]. Recently,supramolecular assemblies of new-type amphiphilic cell-penetrating peptides in order to target cancer cell mitochondria stand for an emerging instrument for suppressing tumor growth. The adopted strategy is designed to amplify the apoptotic stimuli by impairing the mitochondrial VDAC1 (voltage-dependent anion channel-1)-hexokinase-II (HK-II) interaction [45].
NSCLC cells have overexpressed phosphofructokinase (PFK) (Fig. 1) as well, driving glycolytic flux to grow. Among the members of PFKFB, PFKFB3 appears higher level of (740-fold) kinase activity and lesser level of bisphosphatase activity. Based on previous studies, PFKFB3 was widely expressed in diverse organs and tumor cells, including lung, gastric, breast, ovarian, and thyroid carcinomas. It causes shifts in metabolism, giving rise to the proliferation and survival of tumor cells [46, 47]. A recent study also demonstrates that targeting PFKFB3 restrained cell viability and glycolytic activity, which may be a promising therapeutic strategy in treating lung adenocarcinoma [48]. Another PFK-1 family member, PFKP, was found to be up-regulated in both NSCLC tissues and cell lines and is correlated with lung cancer cell proliferation and patient prognosis [49].
The M2 isoform of pyruvate kinase (PKM2), as a glycolytic terminal enzyme, catalyzes the last step of glycolysis, shifting the phosphate from phosphoenolpyruvate (PEP) to adenosine diphosphate (ADP) [43]. It has become a vital element that adjusts aerobic glycolysis in cancer cells [50]. The strategies of PKM2 inhibition or silencing [51] as well as activation [52, 53] have shared equal debates in literature owing to potential of treatment in limiting tumor growth.
Gopinath Prakasam found that knockdown of AMPK in cells silenced for PKM2 or PKM1 took on inhibiting growth and contributed to apoptosis [54]. While another study carried out by Li.et al. showed that a potential PKM2 activator, 0089–0022, serves as a promising anti-cancer therapy candidate in NSCLC [55].
Due to the Warburg effect, a large amount of lactic acid is synthesized from pyruvate [22]. Lactate generated by LDH is shifted by monocarboxylate anion transporters (MCT) so that an alkaline internal environment is maintained. This will bring benefits to metabolism [56]. As is well-known to all that MCT4 transfers lactate out of the cell and MCT1 moves the entry of lactate to tumor cells [57]. A previous study had shown cellular expression levels of MCT1 and MCT4 were in relation to invasion activity. This suggests inhibitors of the MCT maybe offer a new-model strategy in order to hinder cancer metastasis [58].
In short, there is evidence demonstrates that LC employs HIF-1α to improve glycolytic flux and to neutralize ROS via the upregulated activity of glucose importers (GLUT1, GLUT4), glycolytic enzymes (PFK, PK), and lactate transporters (MCT1, MCT4). Suppressing metabolic enzymes activity involved in glycolysis and lactate production may become promising new alternative therapeutic targets for lung cancer. Up to date, the majority of prior available evidence was concentrated on small single studies, which only involve partly LC metabolism in a limited number of primary human LC cell lines. Before transforming into clinical trials, more comprehensive preclinical investigations on LC metabolism in divergent phases of the disease are in demanded to enhance the effectiveness of these findings.
Impact of metabolism reprogramming on cells of the tumor microenvironment and immunotherapy in NSCLC
Glucose and glycolysis
The tumor microenvironment (TME) plays a vital role in tumor behavior and therapeutic effect [59]. Meanwhile, cancer cells can regulate a well-characterized metabolic phenotype that can deeply affect TME [60]. In addition, an emerging theme is that metabolic phenotypes are tissue specific and cancer subtype specific. As a highly heterogeneous disease covering a heterogeneous population, NSCLC is blessed with a complicated system to identify the disease state and progression [61]. Tumor-induced TME metabolic reprogramming as shown in Fig. 3 appears to be more common.
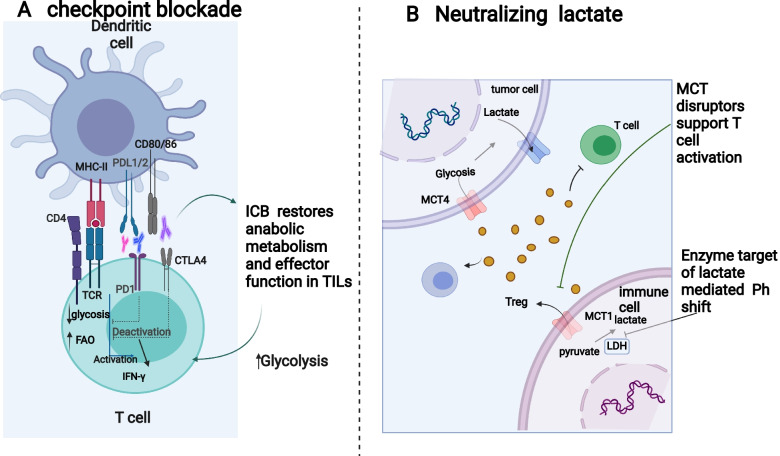
Fig. 3.
Aspects of glucose metabolism that may serve as therapeutic targets. A Cancer patients have successfully used immune-based interventions, including immune checkpoint inhibitors, to treat a variety of cancers. Low glucose, acidity and lactic acid in tumor microenvironment lead to enhanced expression of checkpoint receptors. In turn, this results in reduced glycosis and increased FAO, therefore causing immunosuppression. Immunotherapy targeted at these checkpoint receptors has been successful in restoring glycolysis to immune cells, which promotes anti-tumor immunity against tumors. B In the TME, increasing lactate promotes the survival of immune suppressive T cells and restraing the function of T effector. Lactate accumulation can be reduced by inhibiting lactate-producing enzymes, inhibiting lactate transporters or neutralizing acid-induced by lactic acid. These strategies have been proven effective in improving anti-tumor immunity. Figure created using BioRender.com
With the increase of metabolic activity of tumor cells, glucose and amino acids in tumor microenvironment were significantly deficient. Glucose utilization by tumors metabolically limits T cells, which paralyzes their ability to maintain aerobic glycolysis and anti-tumor activity [62–64]. This is partly attributed to T cells, like tumor cells, being metabolically dependent on glycolysis. Further, T cell metabolism is regulated by HIF-1α, PPARy signaling and the transcription factor C-MYC-associated pathway and nuclear receptor family pathway [65, 66].
However, tumor-restricted glucose utilization would not totally make T cells disrupted, presumably because T cells seek for available metabolic sources. For example,mitochondrial activation of CD8 + T cells by PPAR-γ agonists strengthens the anti-tumor immunity in T cells during PD-1 blockade [66]. Another experiment has demonstrated that reactivation of depleted T cells relies on reserve of lipids by fatty acid oxidation in T lymphocytes receiving PD-1 signals [67]. Thus, blocking these pathways with checkpoint inhibitors can partially rescue glycolysis and biosynthesis, thereby reversing the effector function of CD8 + T cells (Fig. 4).
Fig. 4.
The effects of lung cancer metabolism on immune cells within the tumor microenvironment (TME). Cancerous TME develop in lungs, lacking glucose and tryptophan, while immune-suppressive molecules such as lactate and kynurenine accumulate. In the tumor microenvironment, the high levels of lactate inhibit the tumor immune responses by (1) polarizing macrophages toward the suppressive M2 phenotypes, as well as (2) preventing monocyte migration and dendritic cell differentiation. (3) The presence of excessive lactate also inhibits the function of CD8+ T cell directly. Besides kynurenine, tumors are also known to repress T-cell activity, as is tumor-induced tryptophan deficiency and glucose deprivation. Figure created using BioRender.com
On the basis of the previous studies, declining metabolic burden on effector T cells, that lies in tumor microenvironment, might cause durable and steady anti-tumor immune responses. For instance, inhibition of GLUT1 receptors may contribute to more useful T cells and strengthen anti-tumor immune responses, or under the circumstance of glycolysis inhibitors like 2-deoxyglucose (2-DG), antitumor function can be enhanced in primed T cells [68, 69].However,several other studies have demonstrated that inhibiting nutrient transporters and enzymes got involved in glucose metabolism of CD8 + T cells could regulate T cell differentiation and inhibit CD8 + T cell function under low-glucose conditions [70].
The Treg cells accumulate in the TME and play a critical role in dampening antitumor effect. According to growing evidence, Tregs are able to differentitate and survive due to low glucose availability imposed by tumor condition. This extreme environment requires Tregs to utilize oxidative phosphorylation (OXPHOS) as a source of energy. In fact, lactate and kynurenin, metabolic waste products of glycolysis pathways, inhibit conventional T cell activation and cytotoxicity in Treg cells [71–73]. Reprogramming tumor cells, for example, inhibits the infiltration of effector T cells (Teffs) or induces apoptosis, enhances the differentiation of regulatory T cell (Tregs). As a result of reprogramming tumor cells, lactic acid accumulates and carbon dioxide is released, thereby suppressing the immune system [74].
Contrary to CD8+ and CD4+ T cells, Treg metabolism dependes on external factors which include nutrient availability as well as TCR triggering and cytokine milieu, [75, 76]. It is likely that Treg cells are not affected by glucose competition in the tumor site because they are able to utilize alternative to glucose for energy [77]. Rather than glycolysis, the metabolism of Tregs is primarily based on the oxidation of fatty acids [78, 79]. Tregs rely primarily on FAO for self-maintenance, and they exhibit low mTOR activity, so these fatty acids provide the perfect soil for Treg maintenance [80].
It has been demonstrated that fatty acids, combined with in vitro inhibition of glucose uptake and glucose oxidation, lead to Treg differentiation [81]. Moreover, lipid uptake and oxidation are mandatory for the expression of Foxp3, as demonstrated in murine models [81]. In hypoxic tumors, hypoxia-inducible factor (HIF)-1* elicits pyruvate to exit mitochondria with OXPHOS, causing Tregs reliant upon fatty acids for mitochondrial metabolism. As a result, FAO plays an important role in cancer metabolism of Tregs [82].
Effects of acidic extracellular microenvironment on immune cells
One main driving force of the metabolic remodeling appearing in the TME is without doubt rendered by hypoxia and accumulation of lactate. Lactate production is able to be higher level (40-fold) in tumor cells, and lactate dehydrogenase (LDH) display a positive relationship with tumor volume and clinical severity, as well as prognosis [83, 84].
Table 1 summarizes the effects of tumor-derived lactic acid on tumor-infiltrating lymphocytes in TME.
Table 1.
The impacts of lactate and acidification on immune cells in the TME
| Immune cells | Effects |
|---|---|
| TADCs | Inhibition monocyte activation [64] |
| Inhibition antigen presentation [65] | |
| MDSCs | Immunosuppressive microenvironment [66] |
| Cancer aggressiveness [66] | |
| NK cells | Inhibition of effector functions [66] |
| TAMs | Induction of M2 polarization [72, 73] |
| Treg cells | Increase in Treg proliferation [76] |
| Enhancement of immunosuppressive effect [76] | |
| CD8 + T cells | Inhibition of effector functions [74] |
As antigen presentation cells, tumor associated dendritic cells (TADCs) initiate and enhance antitumor immune responses. TADCs promote antitumor immune surveillance, as they are able to exhibit neoantigens to T cells, thereby initiating a T-cell-mediated immune response [85]. Studies have shown that lactic acid accumulation has a direct immunosuppressive effect on immune cells. Lactic acid inhibits monocyte activation and dendritic cell antigen presentation [85, 86].
Myeloid-derived suppressor cells (MDSC) are key components of protumor immune responses and their accumulation in immune organs relate to immunosuppression and cancer aggressiveness. Tumoral lactate production was also found to increases MDSCs and suppresses natural killer (NK) cell function, further accelerating the immunosuppressive microenvironment [87]. A recent study conducted by Baumann found that MDSC blocks T cells by transferring the glycolytic metabolite methylglyoxal, which acts as an immunosuppressant by consuming L-arginine in CD8+ T cells [88].
Tumor-associated macrophages (TAMs) serve as main components in TME [89].TAM, being highly plastic, is able to polarize to two primary phenotypes: the antitumor M1 (TAM1) together with the protumor M2 (TAM2). Several experiments and clinical trials demonstrate that TAMs are primarily of the M2 phenotype, which drive tumor progression and metastasis [90, 91] as well as suppression of antitumor immune responses [92].
Tumor-cell-derived lactic acid facilitate M2-polarization of TAMs through improved arginase and HIF-1α stabilization [93, 94]. After we treated TAMs from patients either lactate or conditioned medium from two tumor cell lines: the Lewis lung carcinoma (LLC) and the melanoma cell lines, TAMs show enhanced expression of HIF-1α and M2-polarization [93].
Therefore, lactic acid produced by cancer cells has a direct immunosuppressive effect, further initiating MDSC-mediated immunosuppression and driving M2 polarization by inhibiting the differentiation of monocyte derived dendritic cells. Tumor-derived lactate can also decrease T cell function by reducing lactate export through MCT1, which inhibits their capability to retain aerobic glycolysis [95]. Additionally, inhibition of effector T cell and boost of Treg in TME by lactate lead to enhanced immunosuppressive microenvironment [96, 97]. Hence, neutralization of an acidic environment is probably to have a significant implication on further the efficacy of ICB (Fig. 3).
Meanwhile, in a recent preclinical study, buffering the TME with bicarbonate administration able to limit tumor growth and improve antitumor responses in animals when merged with either anti-PD-1antibodies (Abs), anti-CTLA or adoptive T cell transfer in a melanoma model [98] .Although molecular mechanism underlying this combination were not clear,these results imply that reversing the acidic TME may increase the selectivity of treatments and meanwhile improve ICB therapy by increasing tumor infiltration by activated T lymphocytes. Fig. 4.
Amino acids metabolism
NSCLC has metabolically increased dependence on glucose and cystine or glutamine. Some amino acids have been studied as anti-cancer targets for drug discovery, involving glutamine and L-tryptophan.
Smoking induces the expression of cystine-glutamate antiporter xCT (SLC7A11) in NSCLC cells. Hence, one approach is to deprive cancer cells of up taking cystine by regulating glutaminolysis. Sulfasalazine (SASP), a FDA-approved medication owns suppressive influence on the function of xCT, which inhibits cystine uptake and contribute to cystine depletion [99, 100]. By reinforcing the correlation of xCT, the combination of tumor cell xCT deficiency with anti-CTLA, accelerated the frequencies and anti-tumor activity and persistence [101, 102].
An available strategy is to induce cancer cells glutamine deprivation by reducing glutamine uptake. CB-839 (Telaglenestat) is one of the most effective and selective GSL1(glutaminase1) inhibitors. It has been shown to suppress growth in non-small-lung-cancer, synergize with immune checkpoint blockade Nivolumab or pembrolizumab [103].
Indoleamine-2,3-dioxygenase 1 (IDO1) catalyzes the conversion of tryptophan into kynurenine which lead to an immune-suppressive TME and thus contributing to tumor growth [104]. Growing IDO1 activity prevents T effector activation,dampens NK cell function, facilitates Treg activation, and drives the expansion and activation of DCs and MDSCs [105, 106]. Accordingly, IDO is an appealing therapeutic target for anti-tumor drug discovery. So, can we potentiate the efficacy of ICB for NSCLC patients by restoring TME tryptophan levels and inhibiting IDO1-dependent tryptophan metabolism? Several clinical trials have been investigating it and more comprehensive investigations are in needed. Epacadostat (Keynote-037) is one of the clinical-stage IDO1 inhibitors (Table 1). Its combination with immune checkpoint inhibitor pembrolizumab yielded promising data in solid tumors (including NSCLC). However, in the phase III trial (KEYNOTE-252), Epacadostat failed to meet their endpoint in unresectable or metastatic melanoma [107, 108].
Meanwhile, several clinical trials are under way to test the combination of IDO1 inhibitor with antitumor vaccines. A phase I IDO-derived peptide vaccination study was performed in patients with stage III/IV NSCLC based on long-term follow up (NCT01219348). Here, we present the long-term clinical and immunological outcomes of the vaccine-based strategy [109, 110]. All these has prompted researchers to make breakthrough about IDO1 inhibition and inhibitory metabolites tryptophan, thereby identifying patients candidates most susceptible to benefit from cancer immunotherapy.
Finally, Metformin, the old anti-diabetic drug, has also found its new place when combining with immunotherapy. According to studies in vivo and in vitro, targeting AMPK could be an meaningful strategy to augment immune checkpoint blockade efficiency. A phase II trial is evaluating the initial efficacy of metformin combined with nivolumab to treat metastatic lung cancer patients (NCT03048500).
Considering the metabolic interplay between cancer and immunes cells, the immune checkpoint blockade can synergize with metabolic intervention therapies. Table 2 lists the tentative therapeutic targets that integrate metabolism with immunotherapy and will be further discussed below. Table 3 lists novel metabolic reprogramming immunotherapies that are in the preclinic.
Table 2.
Metabolic reprogramming in immunotherapy clinical trials
| Medications | Target | Combination Therapy | Context | identifier/reference |
|---|---|---|---|---|
| Metformin | AMPK | Nivolumab (anti-PD1) | NSCLC | NCT03048500 |
| epacadostat | IDO | Pembrolizumab | NSCLC | Keynote-037 |
| epacadostat | IDO | Pembrolizumab + Platinum doublet | NSCLC | NCT03322566 |
| epacadostat | IDO | Pembrolizumab | NSCLC | NCT03322540 |
| IO102 | IDO | Pembrolizumab ± Platinum doublet | NSCLC | NCT T03562871 |
| ID05-peptide IDO | IDO | vaccine-based | NSCLC | NCT01219348 |
| Telaglenestat (CB-839) | Glutaminase1 inhibitor | Nivolumab | NSCLC | NCT02771626 |
| (CB-839) | Glutaminase | pembrolizumab | NSCLC | NCT04265534 |
Table 3.
Metabolic reprogramming immunotherapies in the preclinic
| Inhibitors | Targets | Cancer types | References |
|---|---|---|---|
| Dichloroacetic acid | PDH | breast cancer | [111] |
| Dimethyl fumarate | G6PD | breast cancer | [112] |
| L-Arg bacteria | L-arginine | Colon Cancer | [113] |
| oxamate | LDH | lung cancer cells | [114] |
| CB-839 | Glutaminase | Melanoma mouse | [115] |
| Protein phosphatase 2A | PPP | SCLC | [116] |
| AZD3965 | MCT1 | TC-1 cancer cells | [117] |
Conclusion
TME consists of a variety of cell populations and matrices, in which the bioenergetic requirements of quickly dividing cancer cells and immune cells are pitted against one another for essential nutrients. The metabolic reprogramming of lung cells is magnified by hypoxia, PPARy and PI3K/mTOR/AKT signaling-mediated overexpression of PPP, as well as glycolysis and lactate production as well.
Several immunotherapeutic options have been explored in recent decades in order to block or reprogram TAMs’ or MDSCs’ immunosuppressive activities. Many therapeutic strategies have utilized TAMs and M-MDSCs or their functional mediators as direct targets, including inhibiting CSF1 (M-CSF) interactions with its receptor CSF1R,by small molecules or neutralizing anti-CSF1R (e.g., PLX3397, GW2580, IMC-CS4, AMG820) or anti-CSF1 (e.g., emactuzumab, cabiralizumab) monoclonal antibodies (mAbs), by blocking the production of anti-CSF1R or anti-CSF1 monoclonal antibodies, inhibiting the M2-like phenotypes of TAMs, increasing infiltrating CD8+ T cells, and improving the immune response to ICB. The further investigation of synergistic effects of checkpoint blockade-based immunotherapies on TAMs or MDSCs will improve ongoing immunotherapeutics.
In addition to TAMs and MDSCs, DCs,as the most powerful antigen-presenting cells (APCs) of the immune system, also suffer from metabolic disorders. When DCs transition from an immature state to a mature state, they demonstrate metabolic plasticity. FAO drives ATP production in immature DCs via OXPHOS. Tumor-associated DCs accumulate lipids, which are negative regulators of their ability to process antigens via MHC class II and to stimulate allogenic T cells. There is now a need for new studies to evaluate how anticancer therapies affect myelopoiesis and immunomodulation, as well as their interactions with metabolism in the host.
Energy and nutrients deprivation in the TME synergy with immune-suppressive molecules like lactic acid jointly promoting the suppressive TME. This further exacerbate immune escape. Strategies for combing ICB with LC metabolism meet the metabolic requirements of immune cells like M1 macrophages or CD8+ T cells, boosting anticancer immunity. Recently,clinical studies in relation to the integration of ICB with drugs tumor-targeting are under way. IDO-inhibition could overcome the detrimental effects of tryptophan depletion and kynurenine accumulation and balance tryptophan-kynurenine pathway.
Simultaneous inhibition of glycolysis and lactic acid production in LC cells seems to be an effective method to improve the efficacy of immunotherapy. But so far, tumor targeting and localization targeting to protect non-cancer cells from glucose starvation still need to be further explored.
In a word, disrupting LC metabolism perhaps be a promising option to promote cancer immunotherapies. Novel therapeutic targets are under development in preclinical models, at the same time, retooling existing drugs for optimized ICB should be investigated as well.
Acknowledgements
Not applicable.
Abbreviations
- MR
Metabolic reprogramming
- NSCLC
Non-small cell lung cancer
- LUAD
Lung adenocarcinoma
- LUSC
Lung squamous cell carcinoma
- ICB
Immune checkpoint blockade
- irAE
Immunotherapeutic-induced adverse events
- TME
Tumor microenvironment
- TCA
Tricarboxylic acid
- OXPHOS
Oxidative phosphorylation
- LC
Lung cancer
- PET/CT
Positron emission tomography/computed tomography
- 18F-FDG
Glucose-analog fluorodeoxyglucose
- PC
Pyruvate carboxylase
- PPP
Phosphopentose
- NADPH
nicotinamide adenine dinucleotide phosphate
- HIF-1α
transcription factor hypoxia-inducible factor 1-alpha
- DPT
Deoxypodophyllotoxin
- Mir-128
microRNA128
- NOX4
Oxidase 4
- GLUTS
Glucose transporters
- HK
Hexokinase
- VDAC1
Voltage-dependent anion channel-1
- PEK
Phosphofructokinase
- PKM2
M2 isoform of pyruvate kinase
- PEP
Phosphoenolpyruvate
- ADP
Adenosine diphosphate
- MCT
Monocarboxylate anion transporters
- 2-DG
2-deoxyglucose
- LDH
Lactate dehydrogenase
- TADCs
Tumor associated dendritic cells
- MDSC
Myeloid-derived suppressor cells
- NK
natural killer
- TAMs
Tumor-associated macrophages
- Teffs
Effector T cells (Teffs)
- LLC
The Lewis lung carcinoma
- SAPS
Sulfasalazine
- GSL1
Glutaminase1
- IDO1
Indoleamine-2,3-dioxygenase 1
Authors’ contributions
WG and JX designed the study. LL and RH drafted the manuscript. JL revised the manuscript. All authors have read and approved the final manuscript.
Funding
This work was supported by grants from National Natural Science Foundation of China (82141121), Natural Science Foundation of Jiangsu Province (BK20211380), Jiangsu Province’s Key provincial Talents Program (ZDRCA2016028),
And The Open Project of Jiangsu Biobank of Clinical Resources(SBK202004003).
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Lulu Lv and Ruo han Huang contributed equally to this work.
Contributor Information
Jing Xu, Email: xujing83@foxmail.com.
Wen Gao, Email: gaowen@jsph.org.cn.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71(1):7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 3.Gridelli C, Rossi A, Carbone DP, Guarize J, Karachaliou N, Mok T, et al. Non-small-cell lung cancer. Nat Rev Dis Primers. 2015;21(1):15009. doi: 10.1038/nrdp.2015.9. [DOI] [PubMed] [Google Scholar]
- 4.Thai AA, Solomon BJ, Sequist LV, Gainor JF, Heist RS. Lung cancer. Lancet. 2021;398(10299):535–554. doi: 10.1016/S0140-6736(21)00312-3. [DOI] [PubMed] [Google Scholar]
- 5.Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ, Jr, Wu YL, et al. Lung cancer: current therapies and new targeted treatments. Lancet. 2017;389(10066):299–311. doi: 10.1016/S0140-6736(16)30958-8. [DOI] [PubMed] [Google Scholar]
- 6.Saw SPL, Ong BH, Chua KLM, Takano A, Tan DSW. Revisiting neoadjuvant therapy in non-small-cell lung cancer. Lancet Oncol. 2021;22(11):e501–e516. doi: 10.1016/S1470-2045(21)00383-1. [DOI] [PubMed] [Google Scholar]
- 7.Reck M, Remon J, Hellmann MD. First-line immunotherapy for non-small-cell lung cancer. J Clin Oncol. 2022;40(6):586–597. doi: 10.1200/JCO.21.01497. [DOI] [PubMed] [Google Scholar]
- 8.Jotte R, et al. Atezolizumab in combination with carboplatin and nab-paclitaxel in advanced squamous NSCLC (IMpower131): results from a randomized phase III trial. J Thorac Oncol. 2020;15(8):1351–1360. doi: 10.1016/j.jtho.2020.03.028. [DOI] [PubMed] [Google Scholar]
- 9.Faivre-Finn C, et al. Four-year survival with Durvalumab after Chemoradiotherapy in stage III NSCLC—an update from the PACIFIC trial. J Thorac Oncol. 2021;16(5):860–867. doi: 10.1016/j.jtho.2020.12.015. [DOI] [PubMed] [Google Scholar]
- 10.Goldberg SB, et al. Pembrolizumab for management of patients with NSCLC and brain metastases: long-term results and biomarker analysis from a non-randomised, open-label, phase 2 trial. Lancet Oncol. 2020;21(5):655–663. doi: 10.1016/S1470-2045(20)30111-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Paz-Ares L, et al. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): an international, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(2):198–211. doi: 10.1016/S1470-2045(20)30641-0. [DOI] [PubMed] [Google Scholar]
- 12.Horvath L, Thienpont B, Zhao L, Wolf D, Pircher A. Overcoming immunotherapy resistance in non-small cell lung cancer (NSCLC) - novel approaches and future outlook. Mol Cancer. 2020;19(1):141. doi: 10.1186/s12943-020-01260-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23(5):540–547. doi: 10.1038/nm.4321. [DOI] [PubMed] [Google Scholar]
- 14.Pauken KE, Dougan M, Rose NR, Lichtman AH, Sharpe AH. Adverse events following cancer immunotherapy: obstacles and opportunities. Trends Immunol. 2019;40(6):511–523. doi: 10.1016/j.it.2019.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Passaro A, Mancuso P, Gandini S, Spitaleri G, Labanca V, Guerini-Rocco E, et al. Gr-MDSC-linked asset as a potential immune biomarker in pretreated NSCLC receiving nivolumab as second-line therapy. Clin Transl Oncol. 2020;22(4):603–611. doi: 10.1007/s12094-019-02166-z. [DOI] [PubMed] [Google Scholar]
- 16.Limagne E, Richard C, Thibaudin M, Fumet JD, Truntzer C, Lagrange A, et al. Tim-3/galectin-9 pathway and mMDSC control primary and secondary resistances to PD-1 blockade in lung cancer patients. Oncoimmunology. 2019;8(4):e1564505. doi: 10.1080/2162402X.2018.1564505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim HR, Park SM, Seo SU, Jung I, Yoon HI, Gabrilovich DI, et al. The ratio of peripheral regulatory T cells to Lox-1+ polymorphonuclear myeloid-derived suppressor cells predicts the early response to anti-PD-1 therapy in patients with non-small cell lung cancer. Am J Respir Crit Care Med. 2019;199:243–246. doi: 10.1164/rccm.201808-1502LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dieu-Nosjean MC, Antoine M, Danel C, Heudes D, Wislez M, Poulot V, et al. Long-term survival for patients with non-small-cell lung cancer with intratumoral lymphoid structures. J Clin Oncol. 2008;26:4410–4417. doi: 10.1200/JCO.2007.15.0284. [DOI] [PubMed] [Google Scholar]
- 19.Germain C, Gnjatic S, Tamzalit F, Knockaert S, Remark R, Goc J, et al. Presence of B cells in tertiary lymphoid structures is associated with a protective immunity in patients with lung cancer. Am J Respir Crit Care Med. 2014;189:832–844. doi: 10.1164/rccm.201309-1611OC. [DOI] [PubMed] [Google Scholar]
- 20.Hughes CE, Benson RA, Bedaj M, Maffia P. Antigen-presenting cells and antigen presentation in tertiary lymphoid organs. Front Immunol. 2016;7:481. doi: 10.3389/fimmu.2016.00481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Al-Shibli K, Al-Saad S, Donnem T, Persson M, Bremnes RM, Busund LT. The prognostic value of intraepithelial and stromal innate immune system cells in non-small cell lung carcinoma. Histopathology. 2009;55:301–312. doi: 10.1111/j.1365-2559.2009.03379.x. [DOI] [PubMed] [Google Scholar]
- 22.Koppenol WH, Bounds PL, Dang CV. Otto Warburg's contributions to current concepts of cancer metabolism. Nat Rev Cancer. 2011;11(5):325–337. doi: 10.1038/nrc3038. [DOI] [PubMed] [Google Scholar]
- 23.Kerr EM, Martins CP. Metabolic rewiring in mutant Kras lung cancer. FEBS J. 2018;285(1):28–41. [DOI] [PMC free article] [PubMed]
- 24.Dall'Olio FG, Calabrò D, Conci N, Argalia G, Marchese PV, Fabbri F, et al. Baseline total metabolic tumour volume on 2-deoxy-2-[18F] fluoro-d-glucose positron emission tomography-computed tomography as a promising biomarker in patients with advanced non-small cell lung cancer treated with first-line pembrolizumab. Eur J Cancer. 2021;150:99–107. doi: 10.1016/j.ejca.2021.03.020. [DOI] [PubMed] [Google Scholar]
- 25.Fan TW, Lane AN, Higashi RM, Farag MA, Gao H, Bousamra M, et al. Altered regulation of metabolic pathways in human lung cancer discerned by 13 C stable isotope-resolved metabolomics (SIRM) Mol Cancer. 2009;8:839–850. doi: 10.1186/1476-4598-8-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sellers K, Fox MP, Bousamra M, Slone SP, Higashi RM, Miller DM, et al. Pyruvate carboxylase is critical for non–small-cell lung cancer proliferation. J Clin Investig. 2015;125:687–698. doi: 10.1172/JCI72873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang J, Li H, Wu Q, Chen Y, Deng Y, Yang Z, et al. Tumoral NOX4 recruits M2 tumor-associated macrophages via ROS/PI3K signaling-dependent various cytokine production to promote NSCLC growth. Redox Biol. 2019;22:101116. doi: 10.1016/j.redox.2019.101116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xiang H, Ramil CP, Hai J, Zhang C, Wang H, Watkins AA, et al. Cancer-associated fibroblasts promote immunosuppression by inducing ROS-generating Monocytic MDSCs in lung squamous cell carcinoma. Cancer Immunol Res. 2020;8(4):436–450. doi: 10.1158/2326-6066.CIR-19-0507. [DOI] [PubMed] [Google Scholar]
- 29.Kierans SJ, Taylor CT. Regulation of glycolysis by the hypoxia-inducible factor (HIF): implications for cellular physiology. J Physiol. 2021;599(1):23–37. doi: 10.1113/JP280572. [DOI] [PubMed] [Google Scholar]
- 30.Xie H, Song J, Godfrey J, Riscal R, Skuli N, Nissim I, et al. Glycogen metabolism is dispensable for tumour progression in clear cell renal cell carcinoma. Nat Metab. 2021;3(3):327–336. doi: 10.1038/s42255-021-00367-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang Y, Liu L, Sun J, Wang S, Yang Z, Li H, Huang N, Zhao W. Deoxypodophyllotoxin Inhibits Non-Small Cell Lung Cancer Cell Growth by Reducing HIF-1α-Mediated Glycolysis. Front Oncol. 2021;11:629543. 10.3389/fonc.2021.629543. [DOI] [PMC free article] [PubMed]
- 32.Hua Q, et al. Hypoxia-induced lncRNA-AC020978 promotes proliferation and glycolytic metabolism of non-small cell lung cancer by regulating PKM2/HIF-1α axis. Theranostics. 2020;10(11):4762–4778. doi: 10.7150/thno.43839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gong T, Cui L, Wang H, Wang H, Han N. Knockdown of KLF5 suppresses hypoxia-induced resistance to cisplatin in NSCLC cells by regulating HIF-1α-dependent glycolysis through inactivation of the PI3K/Akt/mTOR pathway. J Transl Med. 2018;16(1):164. doi: 10.1186/s12967-018-1543-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hong SY, Yu FX, Luo Y, Hagen T. Oncogenic activation of the PI3K/Akt pathway promotes cellular glucose uptake by downregulating the expression of thioredoxin-interacting protein. Cell Signal. 2016;28(5):377–383. doi: 10.1016/j.cellsig.2016.01.011. [DOI] [PubMed] [Google Scholar]
- 35.Tang JM, He QY, Guo RX, Chang XJ. Phosphorylated Akt overexpression and loss of PTEN expression in non-small cell lung cancer confers poor prognosis. Lung Cancer. 2006;51(2):181–91. 10.1016/j.lungcan.2005.10.003. [DOI] [PubMed]
- 36.Yang J, Li J, Le Y, Zhou C, Zhang S, Gong Z. PFKL/miR-128 axis regulates glycolysis by inhibiting AKT phosphorylation and predicts poor survival in lung cancer. Am J Cancer Res. 2015;6:473–485. [PMC free article] [PubMed] [Google Scholar]
- 37.Zeng C, Wu Q, Wang J, Yao B, Ma L, Yang Z, et al. NOX4 supports glycolysis and promotes glutamine metabolism in non-small cell lung cancer cells. Free Radic Biol Med. 2016;101:236–248. doi: 10.1016/j.freeradbiomed.2016.10.500. [DOI] [PubMed] [Google Scholar]
- 38.Fu QF, Liu Y, Fan Y, Hua SN, Qu HY, Dong SW, et al. Alpha-enolase promotes cell glycolysis, growth, migration, and invasion in non-small cell lung cancer through FAK-mediated PI3K/AKT pathway. J Hematol Oncol. 2015;8:22. doi: 10.1186/s13045-015-0117-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ancey PB, Contat C, Boivin G, Sabatino S, Pascual J, Zangger N, et al. GLUT1 expression in tumor-associated neutrophils promotes lung Cancer growth and resistance to radiotherapy. Cancer Res. 2021;81(9):2345–2357. doi: 10.1158/0008-5472.CAN-20-2870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.van Baardwijk A, Dooms C, van Suylen RJ, Verbeken E, Hochstenbag M, Dehing-Oberije C, et al. The maximum uptake of (18) F-deoxyglucose on positron emission tomography scan correlates with survival, hypoxia inducible factor-1alpha and GLUT-1 in non-small cell lung cancer. Eur J Cancer. 2007;43(9):1392–1398. doi: 10.1016/j.ejca.2007.03.027. [DOI] [PubMed] [Google Scholar]
- 41.Osugi J, Yamaura T, Muto S, Okabe N, Matsumura Y, Hoshino M, et al. Prognostic impact of the combination of glucose transporter 1 and ATP citrate lyase in node-negative patients with non-small lung cancer. Lung Cancer. 2015;88(3):310–318. doi: 10.1016/j.lungcan.2015.03.004. [DOI] [PubMed] [Google Scholar]
- 42.O'Byrne KJ, Baird AM, Kilmartin L, Leonard J, Sacevich C, Gray SG. Epigenetic regulation of glucose transporters in non-small cell lung cancer. Cancers (Basel) 2011;3(2):1550–1565. doi: 10.3390/cancers3021550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tanner LB, Goglia AG, Wei MH, Sehgal T, Parsons LR, Park JO, et al. Four key steps control glycolytic flux in mammalian cells. Cell Syst. 2018;7(1):49–62 e8. doi: 10.1016/j.cels.2018.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Patra KC, Wang Q, Bhaskar PT, Miller L, Wang Z, Wheaton W, et al. Hexokinase 2 is required for tumor initiation and maintenance and its systemic deletion is therapeutic in mouse models of cancer. Cancer Cell. 2013;24(2):213–228. doi: 10.1016/j.ccr.2013.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Liu D, Angelova A, Liu J, Garamus VM, Angelov B, Zhang X, et al. Self-assembly of mitochondria-specific peptide amphiphiles amplifying lung cancer cell death through targeting the VDAC1-hexokinase-II complex. J Mater Chem B. 2019;7(30):4706–4716. doi: 10.1039/C9TB00629J. [DOI] [PubMed] [Google Scholar]
- 46.Li HM, Yang JG, Liu ZJ, Wang WM, Yu ZL, Ren JG, et al. Blockage of glycolysis by targeting PFKFB3 suppresses tumor growth and metastasis in head and neck squamous cell carcinoma. J Exp Clin Cancer Res. 2017;36(1):7. doi: 10.1186/s13046-016-0481-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Minchenko OH, Tsuchihara K, Minchenko DO, Bikfalvi A, Esumi H. Mechanisms of regulation of PFKFB expression in pancreatic and gastric cancer cells. World J Gastroenterol. 2014;20(38):13705–13717. doi: 10.3748/wjg.v20.i38.13705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li X, Liu J, Qian L, Ke H, Yao C, Tian W, et al. Expression of PFKFB3 and Ki67 in lung adenocarcinomas and targeting PFKFB3 as a therapeutic strategy. Mol Cell Biochem. 2018;445(1–2):123–134. doi: 10.1007/s11010-017-3258-8. [DOI] [PubMed] [Google Scholar]
- 49.Shen J, Jin Z, Lv H, Jin K, Jonas K, Zhu C, et al. PFKP is highly expressed in lung cancer and regulates glucose metabolism. Cell Oncol (Dordr) 2020;43(4):617–629. doi: 10.1007/s13402-020-00508-6. [DOI] [PubMed] [Google Scholar]
- 50.Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R, et al. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature. 2008;452(7184):230–233. doi: 10.1038/nature06734. [DOI] [PubMed] [Google Scholar]
- 51.Mazurek S. Pyruvate kinase type M2: a key regulator of the metabolic budget system in tumor cells. Int J Biochem Cell Biol. 2011;43(7):969–980. doi: 10.1016/j.biocel.2010.02.005. [DOI] [PubMed] [Google Scholar]
- 52.Sun H, Zhu A, Zhang L, Zhang J, Zhong Z, Wang F. Knockdown of PKM2 suppresses tumor growth and invasion in lung adenocarcinoma. Int J Mol Sci. 2015;16(10):24574–24587. doi: 10.3390/ijms161024574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Anastasiou D, Yu Y, Israelsen WJ, Jiang JK, Boxer MB, Hong BS, et al. Pyruvate kinase M2 activators promote tetramer formation and suppress tumorigenesis. Nat Chem Biol. 2012;8(10):839–847. doi: 10.1038/nchembio.1060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Parnell KM, Foulks JM, Nix RN, Clifford A, Bullough J, Luo B, et al. Pharmacologic activation of PKM2 slows lung tumor xenograft growth. Mol Cancer Ther. 2013;12(8):1453–1460. doi: 10.1158/1535-7163.MCT-13-0026. [DOI] [PubMed] [Google Scholar]
- 55.Prakasam G, Singh RK, Iqbal MA, Saini SK, Tiku AB, Bamezai RNK. Pyruvate kinase M knockdown-induced signaling via AMP-activated protein kinase promotes mitochondrial biogenesis, autophagy, and cancer cell survival. J Biol Chem. 2017;292(37):15561–15576. doi: 10.1074/jbc.M117.791343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Parks SK, Chiche J, Pouyssegur J. pH control mechanisms of tumor survival and growth. J Cell Physiol. 2011;226(2):299–308. doi: 10.1002/jcp.22400. [DOI] [PubMed] [Google Scholar]
- 57.Payen VL, Mina E, Van Hée VF, Porporato PE, Sonveaux P. Monocarboxylate transporters in cancer. Mol Metab. 2020;33:48–66. doi: 10.1016/j.molmet.2019.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Izumi H, Takahashi M, Uramoto H, Nakayama Y, Oyama T, Wang KY, et al. Monocarboxylate transporters 1 and 4 are involved in the invasion activity of human lung cancer cells. Cancer Sci. 2011;102(5):1007–1013. doi: 10.1111/j.1349-7006.2011.01908.x. [DOI] [PubMed] [Google Scholar]
- 59.Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19(11):1423–1437. doi: 10.1038/nm.3394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Vander Heiden MG, DeBerardinis RJ. Understanding the intersections between metabolism and Cancer biology. Cell. 2017;168(4):657–669. doi: 10.1016/j.cell.2016.12.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chen Z, Fillmore CM, Hammerman PS, Kim CF, Wong KK. Non-small-cell lung cancers: a heterogeneous set of diseases. Nat Rev Cancer. 2014;14(8):535–546. doi: 10.1038/nrc3775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Chang CH, Qiu J, O'Sullivan D, Buck MD, Noguchi T, Curtis JD, et al. Metabolic competition in the tumor microenvironment is a driver of Cancer progression. Cell. 2015;162(6):1229–1241. doi: 10.1016/j.cell.2015.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hope HC, Salmond RJ. Targeting the tumor microenvironment and T cell metabolism for effective cancer immunotherapy. Eur J Immunol. 2019;49(8):1147–1152. doi: 10.1002/eji.201848058. [DOI] [PubMed] [Google Scholar]
- 64.Kouidhi S, Elgaaied AB, Chouaib S. Impact of metabolism on T-cell differentiation and function and cross talk with tumor microenvironment. Front Immunol. 2017;8:270. doi: 10.3389/fimmu.2017.00270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cho SH, Raybuck AL, Stengel K, Wei M, Beck TC, Volanakis E, et al. Germinal Centre hypoxia and regulation of antibody qualities by a hypoxia response system. Nature. 2016;537(7619):234–238. doi: 10.1038/nature19334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chowdhury PS, Chamoto K, Kumar A, Honjo T. PPAR-induced fatty acid oxidation in T cells increases the number of tumor-reactive CD8+ T cells and facilitates anti-PD-1 therapy. Cancer Immunol Res. 2018;6(11):1375–1387. doi: 10.1158/2326-6066.CIR-18-0095. [DOI] [PubMed] [Google Scholar]
- 67.Patsoukis N, Bardhan K, Chatterjee P, Sari D, Liu B, Bell LN, Karoly ED, Freeman GJ, Petkova V, Seth P, Li L, Boussiotis VA. PD-1 alters T-cell metabolic reprogramming by inhibiting glycolysis and promoting lipolysis and fatty acid oxidation. Nat Commun. 2015;6:6692. doi: 10.1038/ncomms7692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sukumar M, Liu J, Ji Y, Subramanian M, Crompton JG, Yu Z, Roychoudhuri R, Palmer DC, Muranski P, Karoly ED, Mohney RP, Klebanoff CA, Lal A, Finkel T, Restifo NP, Gattinoni L. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J Clin Invest. 2013;123(10):4479–4488. doi: 10.1172/JCI69589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.DeVorkin L, Pavey N, Carleton G, Comber A, Ho C, Lim J, et al. Autophagy regulation of metabolism is required for CD8+ T cell anti-tumor immunity. Cell Rep. 2019;27(2):502–513.e5. doi: 10.1016/j.celrep.2019.03.037. [DOI] [PubMed] [Google Scholar]
- 70.Gemta LF, Siska PJ, Nelson ME, Gao X, Liu X, Locasale JW, Yagita H, Slingluff CL Jr, Hoehn KL, Rathmell JC, Bullock TNJ. Impaired enolase 1 glycolytic activity restrains effector functions of tumor-infiltrating CD8+ T cells. Sci Immunol. 2019;4(31):eaap9520. 10.1126/sciimmunol.aap9520. [DOI] [PMC free article] [PubMed]
- 71.Pearce EL, Pearce EJ. Metabolic pathways in immune cell activation and quiescence. Immunity. 2013;38(4):633–643. doi: 10.1016/j.immuni.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang R, Green DR. Metabolic reprogramming and metabolic dependency in T cells. Immunol Rev. 2012;249:14–26. doi: 10.1111/j.1600-065X.2012.01155.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Franco F, Jaccard A, Romero P, Yu YR, Ho PC. Metabolic and epigenetic regulation of T-cell exhaustion. Nat Metab. 2020;2(10):1001–1012. doi: 10.1038/s42255-020-00280-9. [DOI] [PubMed] [Google Scholar]
- 74.Kishton RJ, Sukumar M, Restifo NP. Metabolic regulation of T cell longevity and function in tumor immunotherapy. Cell Metab. 2017;26(1):94–109. doi: 10.1016/j.cmet.2017.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Savage PA, Klawon DEJ, Miller CH. Regulatory T cell development. Annu Rev Immunol. 2020;38:421–453. doi: 10.1146/annurev-immunol-100219-020937. [DOI] [PubMed] [Google Scholar]
- 76.Geltink RIK, Kyle RL, Pearce EL. Unraveling the complex interplay between T cell metabolism and function. Annu Rev Immunol. 2018;36:461–488. doi: 10.1146/annurev-immunol-042617-053019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Sukumar M, Roychoudhuri R, Restifo NP. Nutrient competition: a new Axis of tumor immunosuppression. Cell. 2015;162(6):1206–1208. doi: 10.1016/j.cell.2015.08.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ho PC, Liu PS. Metabolic communication in tumors: a new layer of immunoregulation for immune evasion. J Immunother Cancer. 2016;4:4. doi: 10.1186/s40425-016-0109-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Li MO, Rudensky AY. T cell receptor signalling in the control of regulatory T cell differentiation and function. Nat Rev Immunol. 2016;16(4):220–233. doi: 10.1038/nri.2016.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Li C, Jiang P, Wei S, Xu X, Wang J. Regulatory T cells in tumor microenvironment: new mechanisms, potential therapeutic strategies and future prospects. Mol Cancer. 2020;19(1):116. doi: 10.1186/s12943-020-01234-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Michalek RD, Gerriets VA, Jacobs SR, Macintyre AN, MacIver NJ, Mason EF, et al. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J Immunol. 2011;186:3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Miska J, Lee-Chang C, Rashidi A, Muroski ME, Chang AL, Lopez-Rosas A, et al. HIF-1α is a metabolic switch between glycolytic-driven migration and oxidative phosphorylation-driven immunosuppression of Tregs in glioblastoma. Cell Rep. 2019;27(1):226–237. doi: 10.1016/j.celrep.2019.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Luo HS, Xu HY, Du ZS, Li XY, Wu SX, Huang HC, et al. Prognostic significance of baseline neutrophil count and lactate dehydrogenase level in patients with esophageal squamous cell cancer treated with radiotherapy. Front Oncol. 2020;10:430. doi: 10.3389/fonc.2020.00430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Girgis H, Masui O, White NM, Scorilas A, Rotondo F, Seivwright A, et al. Lactate dehydrogenase A is a potential prognostic marker in clear cell renal cell carcinoma. Mol Cancer. 2014;13:101. doi: 10.1186/1476-4598-13-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Gottfried E, Kunz-Schughart LA, Ebner S, Mueller-Klieser W, Hoves S, Andreesen R, et al. Tumor-derived lactic acid modulates dendritic cell activation and antigen expression. Blood. 2006;107(5):2013–2021. doi: 10.1182/blood-2005-05-1795. [DOI] [PubMed] [Google Scholar]
- 86.Nasi A, Fekete T, Krishnamurthy A, Snowden S, Rajnavölgyi E, Catrina AI, et al. Dendritic cell reprogramming by endogenously produced lactic acid. J Immunol. 2013;191(6):3090–3099. doi: 10.4049/jimmunol.1300772. [DOI] [PubMed] [Google Scholar]
- 87.Husain Z, Huang Y, Seth P, Sukhatme VP. Tumor-derived lactate modifies antitumor immune response: effect on myeloid-derived suppressor cells and NK cells. J Immunol. 2013;191(3):1486–1495. doi: 10.4049/jimmunol.1202702. [DOI] [PubMed] [Google Scholar]
- 88.Baumann T, Dunkel A, Schmid C, Schmitt S, Hiltensperger M, Lohr K, et al. Regulatory myeloid cells paralyze T cells through cell-cell transfer of the metabolite methylglyoxal. Nat Immunol. 2020;21(5):555–566. doi: 10.1038/s41590-020-0666-9. [DOI] [PubMed] [Google Scholar]
- 89.Komohara Y, Fujiwara Y, Ohnishi K, Takeya M. Tumor-associated macrophages: potential therapeutic targets for anti-cancer therapy. Adv Drug Deliv Rev. 2016;99(Pt B):180–185. doi: 10.1016/j.addr.2015.11.009. [DOI] [PubMed] [Google Scholar]
- 90.DeNardo DG, Barreto JB, Andreu P, Vasquez L, Tawfik D, Kolhatkar N, et al. CD4(+) T cells regulate pulmonary metastasis of mammary carcinomas by enhancing protumor properties of macrophages. Cancer Cell. 2009;16(2):91–102. doi: 10.1016/j.ccr.2009.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lin Y, Xu J, Lan H. Tumor-associated macrophages in tumor metastasis: biological roles and clinical therapeutic applications. J Hematol Oncol. 2019;12(1):76. doi: 10.1186/s13045-019-0760-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Pan Y, Yu Y, Wang X, Zhang T. Tumor-associated macrophages in tumor immunity. Front Immunol. 2020;11:583084. doi: 10.3389/fimmu.2020.583084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Colegio OR, Chu NQ, Szabo AL, Chu T, et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014;513(7519):559–563. doi: 10.1038/nature13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jiang H, Wei H, Wang H, Wang Z, Li J, Ou Y, et al. Zeb1-induced metabolic reprogramming of glycolysis is essential for macrophage polarization in breast cancer. Cell Death Dis. 2022;13(3):206. doi: 10.1038/s41419-022-04632-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Fischer K, Hoffmann P, Voelkl S, Meidenbauer N, Ammer J, Edinger M, et al. Inhibitory effect of tumor cell-derived lactic acid on human T cells. Blood. 2007;109(9):3812–3819. doi: 10.1182/blood-2006-07-035972. [DOI] [PubMed] [Google Scholar]
- 96.Quinn WJ, 3rd, Jiao J, TeSlaa T, Stadanlick J, Wang Z, Wang L, et al. Lactate limits T cell proliferation via the NAD(H) redox state. Cell Rep. 2020;33(11):108500. doi: 10.1016/j.celrep.2020.108500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Angelin A, Gil-de-Gómez L, Dahiya S, Jiao J, Guo L, Levine MH, Beier UH, et al. Foxp3 reprograms T cell metabolism to function in low-glucose, high-lactate environments. Cell Metab. 2017;25(6):1282–1293.e7. doi: 10.1016/j.cmet.2016.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kouidhi S, Ben Ayed F, Benammar EA. Targeting tumor metabolism: a new challenge to improve immunotherapy. Front Immunol. 2018;9:353. doi: 10.3389/fimmu.2018.00353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Briggs KJ, Koivunen P, Cao S, Backus KM, Olenchock BA, Patel H, et al. Paracrine induction of HIF by glutamate in breast Cancer: EglN1 senses cysteine. Cell. 2016;166(1):126–139. doi: 10.1016/j.cell.2016.05.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Saito Y, Takahashi T, Obata Y, Nishida T, Ohkubo S, Nakagawa F, et al. TAS-116 inhibits oncogenic KIT signaling on the Golgi in both imatinib-naïve and imatinib-resistant gastrointestinal stromal tumours. Br J Cancer. 2020;122(5):658–667. doi: 10.1038/s41416-019-0688-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ji X, Qian J, Rahman SMJ, Siska PJ, Zou Y, Harris BK, et al. xCT (SLC7A11)-mediated metabolic reprogramming promotes non-small cell lung cancer progression. Oncogene. 2018;37(36):5007–5019. doi: 10.1038/s41388-018-0307-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Arensman MD, Yang XS, Leahy DM, Toral-Barza L, Mileski M, Rosfjord EC, Eng CH, et al. Cystine-glutamate antiporter xCT deficiency suppresses tumor growth while preserving antitumor immunity. Proc Natl Acad Sci U S A. 2019;116(19):9533–9542. doi: 10.1073/pnas.1814932116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Momcilovic M, Bailey ST, Lee JT, Fishbein MC, Braas D, Go J, Shackelford DB, et al. The GSK3 signaling Axis regulates adaptive glutamine metabolism in lung squamous cell carcinoma. Cancer Cell. 2018;33(5):905–921.e5. doi: 10.1016/j.ccell.2018.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Munn DH, Mellor AL. IDO in the tumor microenvironment: inflammation, counter-regulation, and tolerance. Trends Immunol. 2016;37(3):193–207. doi: 10.1016/j.it.2016.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Frumento G, Rotondo R, Tonetti M, Damonte G, Benatti U, Ferrara GB. Tryptophan-derived catabolites are responsible for inhibition of T and natural killer cell proliferation induced by indoleamine 2,3-dioxygenase. J Exp Med. 2002;196(4):459–468. doi: 10.1084/jem.20020121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Munn DH, Sharma MD, Baban B, Harding HP, Zhang Y, Ron D, Mellor AL. GCN2 kinase in T cells mediates proliferative arrest and anergy induction in response to indoleamine 2,3-dioxygenase. Immunity. 2005;22(5):633–642. doi: 10.1016/j.immuni.2005.03.013. [DOI] [PubMed] [Google Scholar]
- 107.Mitchell TC, Hamid O, Smith DC, Bauer TM, Wasser JS, Olszanski AJ, et al. Epacadostat plus Pembrolizumab in patients with advanced solid tumors: phase I results from a multicenter, open-label phase I/II trial (ECHO-202/KEYNOTE-037) J Clin Oncol. 2018;36(32):3223–3230. doi: 10.1200/JCO.2018.78.9602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Long GV, Dummer R, Hamid O, Gajewski TF, Caglevic C, Dalle S, et al. Epacadostat plus pembrolizumab versus placebo plus pembrolizumab in patients with unresectable or metastatic melanoma (ECHO-301/KEYNOTE-252): a phase 3, randomised, double-blind study. Lancet Oncol. 2019;20(8):1083–1097. doi: 10.1016/S1470-2045(19)30274-8. [DOI] [PubMed] [Google Scholar]
- 109.Iversen TZ, Engell-Noerregaard L, Ellebaek E, Andersen R, Larsen SK, Bjoern J, et al. Long-lasting disease stabilization in the absence of toxicity in metastatic lung cancer patients vaccinated with an epitope derived from indoleamine 2,3 dioxygenase. Clin Cancer Res. 2014;20(1):221–232. doi: 10.1158/1078-0432.CCR-13-1560. [DOI] [PubMed] [Google Scholar]
- 110.Kjeldsen JW, Iversen TZ, Engell-Noerregaard L, Mellemgaard A, Andersen MH, Svane IM. Durable clinical responses and Long-term follow-up of stage III-IV non-small-cell lung Cancer (NSCLC) patients treated with IDO peptide vaccine in a phase I study-a brief research report. Front Immunol. 2018;9:2145. doi: 10.3389/fimmu.2018.02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Dai Z, Wang Q, Tang J, Qu R, Wu M, Li H, et al. A Sub-6 nm MnFe2O4-dichloroacetic acid nanocomposite modulates tumor metabolism and catabolism for reversing tumor immunosuppressive microenvironment and boosting immunotherapy. Biomaterials. 2022;284:121533. doi: 10.1016/j.biomaterials.2022.121533. [DOI] [PubMed] [Google Scholar]
- 112.Lei J, Yang Y, Lu Z, Pan H, Fang J, Jing B, et al. Taming metabolic competition via glycolysis inhibition for safe and potent tumor immunotherapy. Biochem Pharmacol. 2022;202:115153. doi: 10.1016/j.bcp.2022.115153. [DOI] [PubMed] [Google Scholar]
- 113.Canale FP, Basso C, Antonini G, Perotti M, Li N, Sokolovska A, West KA, Leventhal DS, Lora JM, Sallusto F, Geiger R, et al. Metabolic modulation of tumours with engineered bacteria for immunotherapy. Nature. 2021;598(7882):662–666. doi: 10.1038/s41586-021-04003-2. [DOI] [PubMed] [Google Scholar]
- 114.Qiao T, Xiong Y, Feng Y, Guo W, Zhou Y, Zhao J, et al. Inhibition of LDH-A by Oxamate enhances the efficacy of anti-PD-1 treatment in an NSCLC humanized mouse model. Front Oncol. 2021;11:632364. doi: 10.3389/fonc.2021.632364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Varghese S, Pramanik S, Williams LJ, Hodges HR, Hudgens CW, Fischer GM, et al. The Glutaminase inhibitor CB-839 (Telaglenastat) enhances the Antimelanoma activity of T-cell-mediated immunotherapies. Mol Cancer Ther. 2021;20(3):500–511. doi: 10.1158/1535-7163.MCT-20-0430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Mirzapoiazova T, Xiao G, Mambetsariev B, Nasser MW, Miaou E, Singhal SS, et al. Protein Phosphatase 2A as a therapeutic target in small cell lung cancer. Mol Cancer Ther. 2021;20(10):1820–1835. doi: 10.1158/1535-7163.MCT-21-0013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Huang T, Feng Q, Wang Z, Li W, Sun Z, Wilhelm J, et al. Tumor-targeted inhibition of Monocarboxylate transporter 1 improves T-cell immunotherapy of solid tumors. Adv Healthc Mater. 2021;10(4):e2000549. doi: 10.1002/adhm.202000549. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.