Supplemental Digital Content is available in the text.
Keywords: coronavirus, COVID-19, pandemics, pyroptosis, thrombosis
Abstract
Objective:
Alveolar-capillary endothelial cells can be activated by severe acute respiratory syndrome coronavirus 2 infection leading to cytokine release. This could trigger endothelial dysfunction, pyroptosis, and thrombosis, which are the vascular changes, commonly referred to as coronavirus disease 2019 (COVID-19) endotheliopathy. Thus, this study aimed to identify tissue biomarkers associated with endothelial activation/dysfunction and the pyroptosis pathway in the lung samples of patients with COVID-19 and to compare them to pandemic influenza A virus H1N1 subtype 2009 and control cases.
Approach and Results:
Postmortem lung samples (COVID-19 group =6 cases; H1N1 group =10 cases, and control group =11 cases) were analyzed using immunohistochemistry and the following monoclonal primary antibodies: anti-IL (interleukin)-6, anti-TNF (tumor necrosis factor)-α, anti-ICAM-1 (intercellular adhesion molecule 1), and anticaspase-1. From the result, IL-6, TNF-α, ICAM-1, and caspase-1 showed higher tissue expression in the COVID-19 group than in the H1N1 and control groups.
Conclusions:
Our results demonstrated endothelial dysfunction and suggested the participation of the pyroptosis pathway in the pulmonary samples. These conditions might lead to systemic thrombotic events that could impair the clinical staff’s efforts to avoid fatal outcomes. One of the health professionals’ goals should be to identify the high risk of thrombosis patients early to block endotheliopathy and its consequences.
Highlights.
Coronavirus disease 2019 (COVID-19) postmortem lung samples showed higher endothelial expression of IL (interleukin)-6, TNF (tumor necrosis factor)-α, ICAM-1 (intercellular adhesion molecule 1), and caspase-1 than the H1N1 and control samples.
There is some evidence of pyroptosis and endothelial dysfunction in the COVID-19 lung samples.
Endothelial dysfunction and pyroptosis can lead to systemic thrombosis that could impair the clinical staff’s efforts to prevent fatal outcomes.
Patients with COVID-19 could benefit if the high risk of thrombosis is identified early to block the endotheliopathy and its consequences.
Pneumocyte injury caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection contributes to the release of inflammatory cytokines that subsequently injure the endothelium. In addition, endothelial cells express ACE (angiotensin-converting enzyme)-2 and allowing infection by SARS-CoV-2.1
The pulmonary capillary endothelium may switch to a responsive inflammatory phenotype after its activation, expressing cytokines and vascular adhesion molecules that could aggravate the cytokine storm, and contribute to thrombosis.1,2
Evidence suggests that endothelial activation/dysfunction induced by SARS-CoV-2 infection differs from the pandemic influenza A virus H1N1 subtype (disease 2009), especially concerning the significant involvement of the pyroptosis in the former, which may drive endothelial cell death thereby contributing to the increasing of the proinflammatory stimuli and thrombogenic events.2,3
This study aimed to analyze endothelial activation/dysfunction and pyroptosis pathway tissue biomarkers in patients who died from coronavirus disease 2019 (COVID-19) compared with cases of pandemic H1N1 deaths and controls.
Methods
The authors declare that all supporting data are available within the article.
The COVID-19 group (n=6) comprises the postmortem lung biopsies (Research Ethics Committee: 944.734/2020) of patients who died of SARS-CoV-2 (positive nasopharyngeal swab real-time reverse transcription–polymerase chain reaction) severe infections.
The H1N1 group (n=10) comprise the postmortem lung biopsies (Research Ethics Committee: 2.550.445/2018) of patients who died of the pandemic influenza A virus H1N1 subtype (positive nasopharyngeal swab real-time reverse transcription–polymerase chain reaction) severe infections.
Lung samples (Research Ethics Committee: 2.550.445/2018) from necropsies of patients without lung lesions were used as a control group (n=11).
Immunohistochemical assays of paraffin-embedded samples were performed using primary antibodies anti-IL (interleukin)-6; TNF (tumor necrosis factor)-α; ICAM-1 (intercellular adhesion molecule 1); and caspase-1 (online Table I).
Slides were scanned on the Axio Scan Scanner Z1 (Carl Zeiss, Germany). Afterward, 10 high-power fields per case were selected, and the measurements of biomarkers in the vascular endothelium.
The Image-Pro Plus software version 4.5 (Media Cybernetics) was used to quantify the tissue expression of IL-6, TNF-α, and ICAM-1, in percentages per high-power fields. Semiquantitative analysis was performed for caspase-1 using the Allred score.
Quantitative variables were described by means and SD and medians with minimum and maximum values. Normality condition was evaluated using Shapiro-Wilks test. Kruskal-Wallis test was used for comparison to quantitative variables (IBM SPSS Statistics v.20.0 software).
Results
Levels of IL-6, TNF-α, ICAM-1, and caspase-1 were significantly highly expressed in the lung tissues in the COVID-19 group than in the H1N1 and control groups (Figure).
Figure.
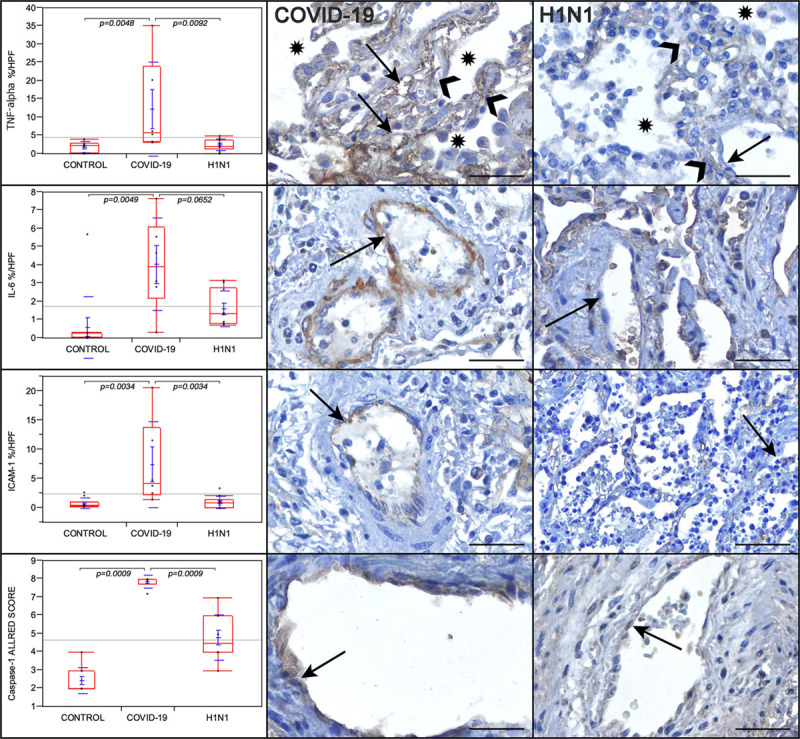
Graphs showing the comparison between the coronavirus disease 2019 (COVID-19), H1N1, and control groups regarding TNF (tumor necrosis factor)-α, IL (interleukin)-6, ICAM-1 (intercellular adhesion molecule 1); in percentage per high-power fields (HPF), and caspase-1 tissue expression (in Allred score). TNF-α is remarkably high in the alveolar septal cells (black arrowheads) and the alveolar-capillary cells (black arrows) of the COVID-19 case than in the H1N1 case. Alveolar lumens are identified with asterisks. IL-6 tissue expression is remarkably high in the endothelial cells (black arrows) of the COVID-19 case than in the H1N1 case. ICAM-1 tissue expression is remarkably higher in the endothelial cells (black arrows) of the COVID-19 case than in the H1N1 case. Caspase-1 is higher in the endothelial cells of the COVID-19 case than in the H1N1 case. Kruskal-Wallis test was used for P values. Scale bars=50 µm.
Discussion
Endothelial cell expresses numerous anticoagulant and antiplatelet molecules and is also covered by a glycocalyx that serves as a barrier against the platelets and blood cells. The impairment of glycocalyx would lead to endothelial activation, endotheliitis, and thrombotic events (endotheliopathy or endothelial dysfunction).
The capillary-alveolar endothelial cells receive a shower of cytokines from the infected SARS-CoV-2 pneumocytes and under these proinflammatory conditions, the breakdown of the glycocalyx would promote endothelial dysfunction and thrombosis.4
The higher levels of IL-6 and TNF-α in the COVID-19 group may be responsible, in part, for the endothelial dysfunction on patients with COVID-19.
The endothelial expression of ICAM-1 was significantly higher in the COVID-19 group than H1N1 and control groups. The activated endothelial cells can express the ICAM-1 molecules that could attract leukocytes (endotheliitis) and transmit intracellular signals leading the sustained proinflammatory status. The proinflammatory condition would result in a systemic endothelial dysfunction and lead to the loss of its integrity via endothelial cell death.5 The persistent inflammatory signaling of these adhesion molecules would also contribute to later thrombotic events.
The relationship between endothelial dysfunction and subsequent thrombotic events is already well known in cardiovascular diseases and diabetes. Five of our cases had comorbidities, such as systemic arterial hypertension, dyslipidemia, diabetes mellitus, chronic kidney disease, and signs of chronic arterial disease. Thus, the previous status of endothelial activation promoted by these comorbidities would aggravate endothelial dysfunction caused by a SARS-CoV-2 infection and its inflammatory response and could be responsible for the most prevalent fatal outcome described in these patients.
Pyroptosis is an inflammatory form of programmed cell death and occurs by activating a protein complex known as the inflammasome. The consequent activation of caspase-1 leads to the proinflammatory cytokines release and cell fragmentation. It has been proposed that pyroptosis could contribute to the endothelial cell death after SARS-CoV-2 infection and could increase proinflammatory cytokines releases, such as IL-1 beta and IL-18.3 Our results showed higher endothelial tissue expression of caspase-1 in the COVID-19 group compared with H1N1 and control groups, and this aspect might suggest the presence of the pyroptosis in capillary-alveolar endothelial cells.
This study’s main limitations are the small number of cases (n=6) and data based on paraffin-embedded postmortem samples that cannot reconstruct the evolving disease process.
In conclusion, our findings suggest the involvement of endothelial dysfunction and thrombosis in the COVID-19. These thrombotic events may be responsible for the fatal outcomes, and early diagnosis could guide health professionals in implementing therapeutic anticoagulation and endothelial stabilization in high-risk patients, mitigating thrombogenic events, systemic inflammatory response syndrome, and multiple organ failure.
Acknowledgments
We are grateful to the Faculdades Pequeno Príncipe, Universidade Federal do Paraná, and Pontificia Universidade Católica do Paraná for their support.
Disclosures
None.
Supplementary Material
Footnotes
Nonstandard Abbreviations and Acronyms
- COVID-19
- coronavirus disease 2019
- ICAM-1
- intercellular adhesion molecule 1
- IL
- interleukin
- SARS-CoV-2
- severe acute respiratory syndrome coronavirus 2
- TNF
- tumor necrosis factor
For Disclosures, see page 2407.
The Data Supplement is available with this article at https://www.ahajournals.org/doi/suppl/10.1161/ATVBAHA.120.314860.
References
- 1.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020;383:120–128. doi: 10.1056/NEJMoa2015432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Henry BM, Vikse J, Benoit S, Favaloro EJ, Lippi G. Hyperinflammation and derangement of renin-angiotensin-aldosterone system in COVID-19: a novel hypothesis for clinically suspected hypercoagulopathy and microvascular immunothrombosis. Clin Chim Acta. 2020;507:167–173. doi: 10.1016/j.cca.2020.04.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yang M; SSRN Electronic Journal. Cell Pyroptosis, a Potential Pathogenic Mechanism of 2019-nCoV Infection. 2020. https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3527420 Accessed April 10, 2020
- 4.Xu J, Zou MH. Molecular insights and therapeutic targets for diabetic endothelial dysfunction. Circulation. 2009;120:1266–1286. doi: 10.1161/CIRCULATIONAHA.108.835223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Woywodt A, Bahlmann FH, De Groot K, Haller H, Haubitz M. Circulating endothelial cells: life, death, detachment and repair of the endothelial cell layer. Nephrol Dial Transplant. 2002;17:1728–1730. doi: 10.1093/ndt/17.10.1728 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


