Abstract
Transfer RNA (tRNA) is present at tens of millions of transcripts in a human cell and is the most abundant RNA in moles among all cellular RNAs. tRNA is also the most extensively modified RNA with, on an average, 13 modifications per molecule. The primary function of tRNA as the adaptor of amino acids and the genetic code in protein synthesis is well known. tRNA modifications play multi-faceted roles in decoding and other cellular processes. The abundance, modification, and aminoacylation (charging) levels of tRNAs contribute to mRNA decoding in ways that reflect the cell type and its environment; however, how these factors work together to maximize translation efficiency remains to be understood. tRNAs also interact with many proteins not involved in translation and this may coordinate translation activity and other processes in the cell. This review focuses on the modifications and the functional genomics of human tRNA and discusses future perspectives on the explorations of human tRNA biology.
Introduction
Together with ribosomal RNA (rRNA) and messenger RNA (mRNA), tRNA is among the three types of RNA presented to students of any introductory biology course. The primary function of tRNA, as the adaptor molecule for amino acid identity and the genetic code, is well understood. The other property of tRNA sometimes briefly mentioned in introductory biology is the presence of RNA modification. Beyond this rudimentary knowledge, tRNA is generally not on the mind of most research scientists, unless they meet it through chance encounter.
Recently, however, tRNA is becoming a prominent research subject once again due to many new discoveries. The consequences of tRNA overexpression on gene expression in human diseases such as cancer have now been well documented.1–4 Human mitochondrial tRNA mutations are a huge source of a wide range of human diseases,5 including MELAS (mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes)6 and MERRF (myoclonic epilepsy with ragged-red fibers).7 Mutations in many tRNA modification enzymes have been linked to human diseases8–12 and many tRNA modifications are receiving renewed interest as increasing numbers of human tRNA modification enzymes become identified, thus enabling focused studies of specific tRNA modifications. Recent discoveries implicate tRNA fragments as small RNAs that affect many cellular processes,13–16 are involved in human diseases17–19 and even directly in epigenetic inheritance in germ-line cells.20,21 Thus tRNA molecules provide a reservoir of RNA species whose function extends beyond translation.
This review focuses on genome-wide aspects of the function of human tRNAs and consequences of their modifications on decoding. I refer to several recent, excellent reviews on the roles of tRNAs in other organisms, the functions of mitochondrial tRNAs, modification enzymes and diseases, and tRNA fragments.22–28
Human tRNA gene features
When the human genome was sequenced in 2001–2003, tRNA genes could be readily identified computationally because a canonical tRNA gene contains highly conserved residues located at predictable distances from each other. This revealed the number of tRNA genes in the reference human genome to be 610 according to the genomic tRNA database (hg19 version)29 although additional tRNA genes could be found in the human population.30 This gene copy number is not unusual among eukaryotic genomes, which generally contain from a few hundred to a few thousand tRNA genes. However, two completely unexpected features of human tRNA genes were found: a large sequence diversity of the tRNA genes and the clustering of tRNA genes at specific chromosomal locations.
Whether a gene truly encodes a tRNA may reflect the secondary structure of its gene product. tRNA folds into a cloverleaf secondary structure, which requires a sufficient number of Watson–Crick or G-U wobble pairs in the four helical stems. The propensity of a tRNA sequence to fold into this secondary structure is reflected in the tRNAScan score,31 assigned for each tRNA gene sequence in the genomic tRNA database. As a rule of thumb, a tRNA gene with a score of >50 can be considered to fold into this secondary structure. tRNA genes with a score of <50 may have too many base pair mismatches in their helical stems and therefore can fold into alternative secondary structures. Indeed, it was shown that a low-scoring tRNAAsp (score 28.6) folds into a non-tRNA structure and cannot be aminoacylated. However, this “tRNA” interacts with a specific mRNA in cells to affect the alternative polyadenylation of this mRNA.32 When only human tRNA genes with a score of >50 are taken into account, 264 different sequences were identified among 423 potential human tRNA genes,29 representing a gene/sequence ratio of 1.6. In comparison, the tRNA gene count and sequences in the Saccharomyces cerevisiae genome are 272 (score >50) and 53, respectively, representing a gene/sequence ratio of 5.1. Therefore, human tRNA genes are much more diverse in sequence than either their yeast counterparts or the tRNA genes of two other model organisms, Caenorhabditis elegans and Drosophila melanogaster (Fig. 1). This high tRNA gene sequence diversity is conserved in all mammalian genomes, suggesting that the diversity was already present upon the evolutionary emergence of the mammals.
Fig. 1.
tRNAAla(AGC) gene sequence diversity. Only genes with tRNAScan score >50 are shown. The four helices are connected by lines. Anticodon nucleotides are marked in bold. The number of genes with identical sequences is in parenthesis. The sequence difference compared to the tRNA with the highest number of gene copies is in red. a The human reference genome has 22 genes, 15 sequences. b C. elegans has 19 genes, 3 sequences. c D. melanogaster has 12 genes, 2 sequences. d S. cerevisiae has 11 genes and 1 sequence
tRNA genes with the same anticodon and different body sequences are termed “isodecoders”.33 The more commonly known term of tRNA “isoacceptors” refers to tRNA genes with different anticodons and charged with the same amino acid. In the example shown in Fig. 1a, all 15 human tRNA isodecoders belong to a single-isoacceptor family of Ala (AGC). It is clear that not all isodecoder genes are expressed in every human cell, and isodecoder expression is expected to vary widely in human tissues.34,35 Direct sequencing of all tRNAs by the DM-tRNA-seq approach (see below) identified ~200 different tRNA sequences (some with tRNAScan score <50) that are present within a 1000-fold molar range in HEK293T cells.36
tRNA isodecoder sequences are highly conserved in the mammalian genomes, suggesting that they serve important functions. However, a large number of tRNA isodecoders may be more harmful than useful. Each tRNA isodecoder in cells is aminoacylated (charged) by its cognate aminoacyl-tRNA synthetase with the correct amino acid according to a set of “identity” elements in the tRNA. At the same time, each tRNA isodecoder needs to avoid being mischarged by the 19 other tRNA synthetases as this could result in potentially harmful levels of mistranslation; it therefore should also carry “preventive” elements against mischarging. Changes in a tRNA sequence that produce a new isodecoder may therefore reduce or eliminate a preventive element for mischarging.
Using a green fluorescent protein reporter that contains an UAG stop codon, different tRNA isodecoders have been analyzed for their comparative stop codon suppression efficiency in HeLa cells.37 Intriguingly, the stop codon suppression efficiency of human tRNASer isodecoders varied over a 10-fold range. Furthermore, a small change in a tRNASer isodecoder sequence increased the suppression efficiency by another 3-fold, suggesting that the natural isodecoder sequences are not conserved for maximizing their utility in translation, at least in the context of stop codon suppression.
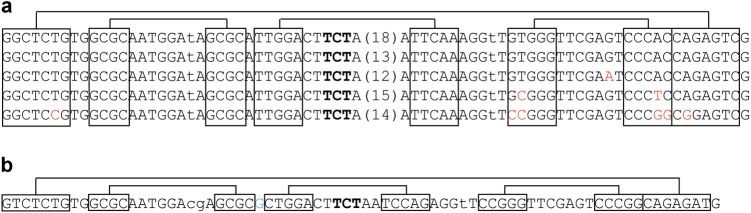
The best-characterized tRNA isodecoder so far was from the tRNAArg(UCU) family that reads the AGA codon of arginine.38 This family has five genes in mice and six genes in humans. Both human and mouse have one isodecoder (Tr-20 in mouse) that is very different from the other isodecoders in both sequences and the presence of introns (Fig. 2). The mouse Tr-20 isodecoder is primarily expressed in the central nervous system (CNS) where it accounts for ~60% of all tRNAArg(UCU) expression, whereas the other isodecoders are expressed in all tissues. A genomic mutation in mice that significantly reduced the expression of the Tr-20 isodecoder increased ribosome pausing specifically at AGA codons and resulted in neuro-degeneration, likely due to increased protein misfolding or degradation. These findings point to the differential, tissue-specific functions of isodecoder tRNAs.
Fig. 2.
Human tRNAArg(TCT) gene sequences. The four helices are connected by lines. Anticodon nucleotides are marked in bold. The number of genes with identical sequences is in parenthesis. a Five “house-keeping” isodecoder genes. All have introns shown in parenthesis with the number of intronic nucleotides indicated. Sequence differences are in red. b The central nervous system isodecoder gene. It has no introns. Also shown in blue is the nucleotide between the D stem and anticodon stem, and it is a G in this isodecoder but A in all others
The second unexpected feature of human tRNA genes is the clustering of 158 genes (144 with a tRNAScan score >50) within just 2.67 million base pairs immediately next to the class I major histocompatibility complex (MHC, aka HLA) genes on chromosome 6.39 Class I MHC proteins present peptide antigens in nucleated cells for surveillance by immune cells and the class I MHC gene sequences are the most polymorphic in the human population.40 The chromosomal association of approximately one third of all human tRNA genes with the MHC therefore may suggest the somewhat unlikely possibility that tRNAs contribute to antigen presentation and are part of the human immune system. The chromosome 6 region where these tRNA genes reside is also the home for the genes that encode many histone proteins and zinc-finger transcription factors. An alternative model for the function of this tRNA cluster is to keep the chromatin structure in this region distinct through high-level RNA polymerase III transcription, to facilitate the high-level transcription of these protein-coding genes. To my knowledge, there is currently no evidence for direct, functional association of tRNA with antigen presentation, and the hypothesis of linking tRNA gene transcription to facilitate RNA polymerase II transcription in this region remains to be tested.
Human tRNA modifications
Like all mammals, humans have chromosomal or nuclear-encoded tRNA genes that produce tRNAs for cytosolic translation and mitochondrial-encoded tRNAs genes that produce tRNAs for mitochondrial translation. Some mitochondrial tRNA sequences are also present in the human chromosome, and these nuclear-derived tRNA transcripts may be imported into mitochondria.29,41 Not all human tRNA genes are transcribed in all cells,34,35 and tRNA expression is tissue specific.2,42 The nuclear-encoded tRNAs are very extensively modified, containing an average of 13 modifications per molecule. Individual tRNAs are, however, modified unevenly; for example, tRNATyr from placenta has 17 modifications43 (Fig. 3), whereas tRNASec from HeLa cells has only 3.44 Mitochondrial tRNAs are generally modified to a lesser extent, containing on average of five modifications per molecule. Since one function of tRNA modification is to increase the stability and rigidity of the tRNA, the presence of fewer modifications in mitochondrial tRNAs contributes to their lower stability and their reliance on specific modifications for structural integrity.45
Fig. 3.
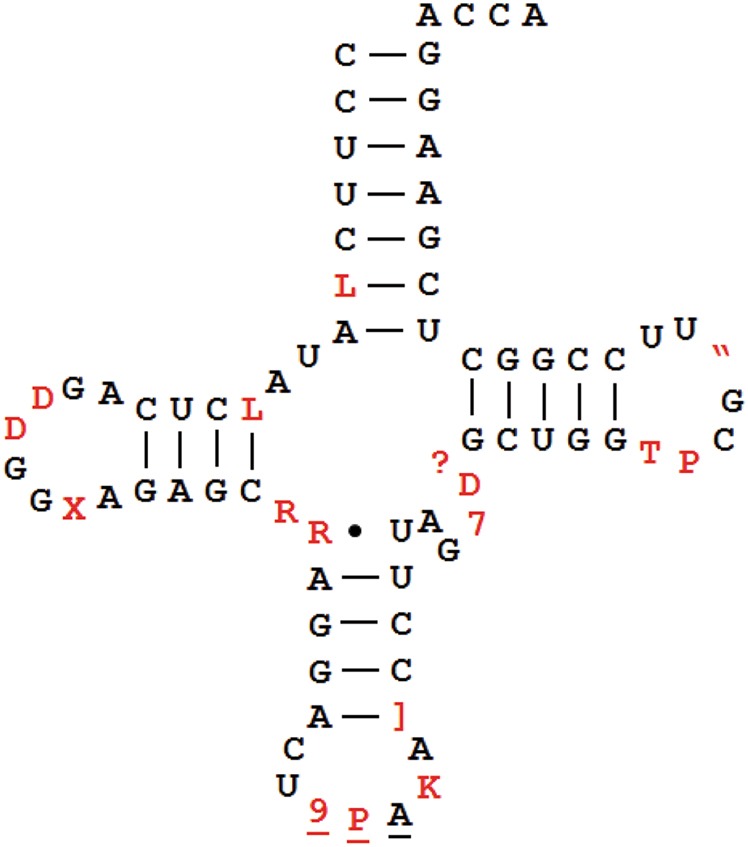
Human cytosolic tRNATyr has the most known modifications. Modified nucleotides are in red; anticodon nucleotides are underlined. Single letter and symbol designations are the nomenclatures used by the RNA modification databases such as Modomics.108–110 L N2-methyl-G (m2G), D dihydro-U, X 3-(3-amino-3-carboxypropyl)-U (acp3U), R N2,N2-dimethyl-G (m22G), 9 galactosyl-queuosine (gal-Q), P pseudoU (Ψ), K N1-methyl-G (m1G);], N1-methylpseudo-U (m1Ψ), 7 N7-methyl-G (m7G), ? 5-methyl-C (m5C), T 5-methyl-U (m5U), “ N1-methyl-A (m1A)
Comprehensive mapping of tRNA modifications is laborious, as one first needs to purify a specific tRNA at appreciable amounts. The current method for tRNA purification uses affinity chromatography with a complementary DNA oligonucleotide linked to a solid resin or bead. Purified tRNAs are digested with ribonucleases (e.g., T1 that cuts 3′ of guanosine) to generate oligonucleotides whose modifications can be precisely identified by mass spectrometry.46–49 This approach works very well and has, for example, led to the complete determination of the modifications of all 22 mitochondrial tRNAs in a mammalian cell.50 Owing to the large sequence diversity of the nuclear-encoded human tRNA isodecoders, the full mapping of cytosolic tRNA modifications has not yet been accomplished because the oligonucleotide affinity columns generally do not have sufficient resolution to allow purification of all individual isodecoders. However, the total modification content of the total cellular or compartmental tRNAs can be quantified upon isolating total tRNA from whole cells or specific cellular compartments followed by nuclease digestion to mononucleosides and mass spectrometry. Importantly, such analyses of modification content have identified condition-dependent tRNA modifications in response to stresses.51,52 In this example, the functional significance of such differential modification was demonstrated by the finding that tRNALeu(CAA)-specific 5-methyl-cytosine (m5C) modification in the wobble anticodon position enhanced translation of stress response genes.53
Analysis of total tRNA modifications indicates that many tRNA modifications are incomplete under physiological conditions. This variation in the levels of tRNA modification should enable cellular adaptation to environmental changes. Therefore, dynamic tRNA modifications seem likely to play similar types of roles in cellular regulation in an analogous manner to dynamic mRNA modifications such as N6-methyladenosine (m6A).54 It is therefore of high biological interest to measure the extents of tRNA modification at individual sites. For human mitochondrial tRNAs that can be purified individually, the fractional extent of modification can be determined by high-resolution and quantitative mass spectrometric analysis of oligonucleotides generated by nucleases.53,55
Certain types of tRNA modifications and their extent can be semiquantitatively determined by high-throughput tRNA sequencing. Methylations at the Watson–Crick face (Fig. 4) do not always lead to full reverse transcriptase (RT) stops in cDNA synthesis; rather, different RTs can read through these modifications at varying efficiencies, leaving behind mutation signatures.56–58 The mutation signatures can be highly dependent upon the modification and the sequence context, as with N1-methyladenosine (m1A).59 The mutation signatures in cDNA synthesis from known tRNA modifications have been used for machine-learning algorithms to identify new modifications in tRNAs and even in mRNAs.57,60
Fig. 4.
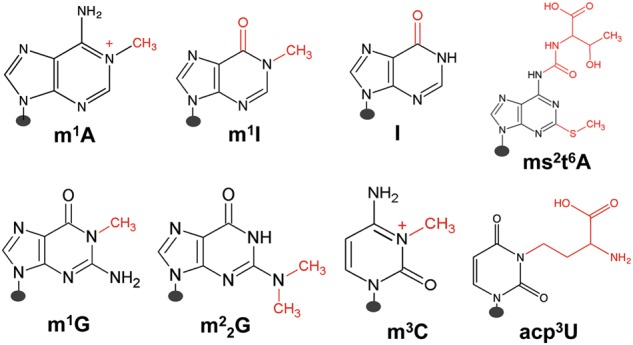
Human nuclear-encoded tRNA modifications investigated by mutation and stop signatures in demethylase-tRNA-seq (DM-tRNA-seq). m1A N1-methyl-A, m1I N1-methyl-inosine, I inosine, ms2t6A 2-methylthio- N6-threonylcarbamoyl-A, m1G N1-methyl-G, m22G N2,N2-dimethyl-G, m3C 3-methyl-C, acp3U 3-(3-amino-3-carboxypropyl)-U
Using a highly processive, thermophilic RT (TGIRT)61 for tRNA sequencing, we found that the mutation and stop fractions at several Watson–Crick face methylations can be used to semiquantitatively determine the modification fraction at specific sites58,62 (Fig. 4). The commonly used Superscript RT in Illumina sequencing stopped at high levels at modifications such as the m1A present in almost all human cytosolic tRNAs. The high level of stops made it more difficult to obtain sequencing reads that pass the m1A modification. TGIRT read through these modifications much more efficiently leaving behind mutations at the modified nucleotide; the efficient read-through also enabled the assessment of multiple modifications in the same tRNA molecule. Our sequencing library avoided 5′ adapter ligation before cDNA synthesis, so that the fraction of RT that stops at each tRNA position could also be measured. We used the term “modification index” (MI) to describe the sum of the mutation and stop fraction at each site.58 We found that the MI value approximated the modification fraction very well as validated by a gel-based method that measured stops in cDNA synthesis using an RT with high-stop fractions (AMV RT). In addition, we were able to quantify the modification from more elaborate Watson–Crick face modifications such as 2-methylthio-N6-threonylcarbamoyl-A (ms2t6A)63 by measuring the stop fraction alone.
The main advantage of sequencing-based methods is that the assessable modifications can be determined simultaneously at single-base resolution potentially for most, if not all, isodecoders. The main disadvantage is that only some modification types can be analyzed in this way. We estimate that our tRNA-seq-based method currently can assess about a quarter of all human tRNA modification sites.58 Modifications that cannot yet be assessed currently include some of the most abundant tRNA modifications such as pseudouridine (Ψ) and 5-methylcytosine (m5C) or some bulky modifications such as N6-threonylcarbamoyladenosine (t6A) and 5-methoxycarbonylmethyl-2-thiouridine (mcm5s2U).
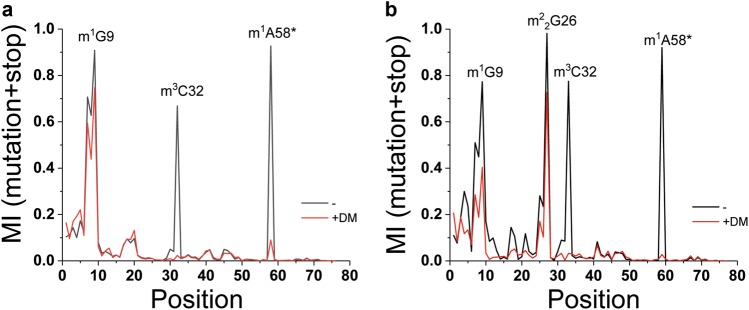
An example of applying TGIRT-based tRNA sequencing to discover new modification in a specific isodecoder is shown in Fig. 5. As discussed above, the tRNAArg(UCU) genes are expressed in a “house-keeping” group of 4–5 genes present in all cells and in a “CNS” group of a single gene only expressed in the CNS in the mouse. The “CNS” tRNAArg(UCU) isodecoder can also be found in minute amounts in the HEK293T cells by tRNA sequencing.62 To identify new modifications in an isodecoder, the MI value for each tRNA residue from sequencing reactions with or without demethylase treatment was calculated. In this case, without demethylase treatment, three high MI peaks were readily seen for a “house-keeping” isodecoder (Fig. 5a). The MI values for all three peaks decreased upon demethylase treatment, indicating that these corresponded to Watson–Crick face methylations that could be removed by the demethylases; in this case, they corresponded to the predicted m1A58, m3C32, and m1G9 modifications. For the “CNS” isodecoder, one additional high peak was present, and the MI for this peak also decreased upon demethylase treatment (Fig. 5b). This new peak is interpreted to correspond to an N2, N2-dimethyl-G (m22G) modification present in many other human tRNAs at this same location. This approach shows that the “CNS” tRNA isodecoder not only has distinct sequences compared to the “housekeeping” isodecoders but also contains an additional modification, which may contribute to its unique role in neuronal translation.
Fig. 5.
Watson–Crick face methylations in tRNAArg(UCU) isodecoders. Data from ref. 58. +DM demethylase-treated, m1A58* only mutation signature was measured for this modification due to the challenge of mapping stops derived from short reads. a An isodecoder from the “house-keeping” group of five intron-containing genes. b The single, central nervous system-specific isodecoder
The function of tRNA modifications is generally direct in mRNA decoding in the case of modifications located in the anticodon loop, but other properties can also be affected including stability, localization, ribosome binding, and translational dynamics. For the decoding role, the strongest effects are derived from modifications to the wobble anticodon (position 34 in the tRNA nomenclature) and to the residue to the immediate 3′ of the anticodon nucleotides (position 37). A-to-Inosine (I) modification occurs in at least eight human tRNA wobble positions that play an obvious role to expand the base pairing capability from A34-U to I34-U/I34-C and maybe even I34-A.64 2′O-methylation of G34/C34 (Gm/Cm) is another common wobble modification in human tRNAs, and it is likely to enhance codon–anticodon interactions on the ribosome.10,65 Elaborate U34 modifications, such as 5-methylaminomethyl-2-thiouridine (mcm5s2U), can confer decoding bias for the cognate codons.66,67 Not all known nucleotide 34 modifications have well-defined functions. For example, the human ALKBH8 is a member of the ALKB homolog family that includes the mRNA m6A demethylases ALKBH5 and FTO. It has a methyltransferase domain that methylates 5-carboxymethyl-U (cm5U) to 5-methoxycarbonylmethyl-U (mcm5U)68,69 and a hydroxylation domain that hydroxylates mcm5U34 in tRNAGly(UCC) to 5-(carboxyhydroxymethyl) methyl ester-U (mchm5U).70,71 The methylation reaction can also be carried out by another human enzyme, hTRMT9, and plays a role in enhanced decoding,72 whereas the function of the hydroxylation is not known. Nucleotide 37 is an A or G in tRNA, and its modifications are always at the Watson–Crick face. G37 modifications are typically N1-methylguanosine (m1G) and can be further modified to peroxywybutosine (W) in tRNAPhe.73 A37 modifications can be N6-threonylcarbamoyladenosine (t6A) or N6-isopentenyladenosine (i6A) with large chemical groups at the N6-position. A major function of the A/G37 modification is to prevent frameshifting.74–77
Modifications outside of the anticodon loop can perform many other tRNA-related functions. For base methylations alone, m1A and m1G9 between the acceptor and D-stem help fold mitochondrial tRNAs into their correct structures.78,79 This modification in the cytosolic tRNAs can also increase tRNA rigidity through enhanced base stacking.80,81 Similarly, the m22G26 modification between the D and anticodon stems increases the stiffness of the modified tRNA, which is likely to be important when tRNA on the ribosome needs to accommodate the conformational dynamics of the ribosome during translational elongation.82,83 Perhaps the best-studied modification outside of the anticodon loop is m1A58 located in the T loop of tRNA. M1A58 adds a positive charge to the nucleobase. It is present in almost all human tRNAs58,84 and is essential for the stability of the initiator tRNAMet.85 Interestingly, the m1A58 modification fraction on individual tRNAs can be variable in different cell types and under different cellular conditions.55,86 The fractional modification is likely maintained by the combined actions of the m1A58 methyltransferase, TRMT6/TRMT61A87 or TRMT61B,88 and the m1A58 demethylase, ALKBH1 in the cytosol86 and in mitochondria.55 A working hypothesis (Fig. 6) for this reversible modification for the cytosolic tRNA is that the m1A58-modified and unmodified tRNAs have differential affinity for the elongation factor EF1A. EF1A delivers tRNA into the A-site of the ribosome in protein synthesis. By adjusting the level of this single modification in specific tRNAs, cells can readily titrate the amount of these tRNAs available for translation through altering their affinity for EF1A and thus for the ribosome. Hypo-modified tRNAs that escape EF1A binding may bind other cellular proteins to coordinate translation and other cellular processes (see below). Although ALKBH1 is the only known “eraser” for tRNA so far, other human enzymes will likely be found that act on other types of tRNA modifications.
Fig. 6.
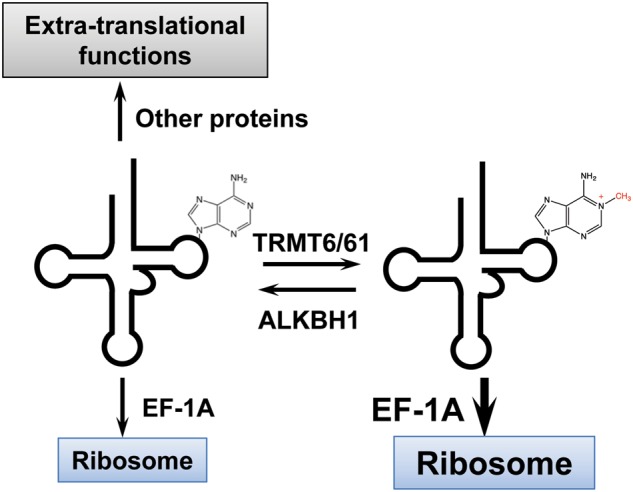
Working model for tRNA m1A58 demethylase (eraser) function. Modified tRNA has a higher affinity for the EF-1A protein, which delivers tRNA to the ribosome. Elevated ALKBH1 activity decreases tRNA availability for translation but may enhance tRNA binding to other cellular proteins due to reduced competition by EF-1A
Functional genomics
Translational regulation occurs at the initiation, elongation, and termination steps. Elongation is the step at which tRNA decoding can play the most fundamental roles. In total, 18 of the 20 amino acids have more than one codon in the genetic code and the distribution of codon usage varies widely in the coding sequences among different organisms. Because multiple codons are present for many amino acids, multiple tRNAs with distinct anticodons (i.e., isoacceptors) are needed to read these codons. In humans, tRNA isoacceptors are present for 12 amino acids, ranging from two (Glu, Lys, Gln), three (Ile, Val, Thr, Ala, Gly, Pro), four (Ser) to five (Leu, Arg). For the six amino acids (Phe, Tyr, His, Asn, Asp, Cys) that have just two codons each, a single tRNA is present in the genome. Here the wobble anticodon nucleotide of all these tRNAs, with the exception of tRNACys, can be modified to more efficiently decode both codons. One additional tRNA property used in decoding is aminoacylation or charging. A charged tRNA has a covalently attached 3′ amino acid and is required for protein synthesis.
The three properties of tRNA—abundance, modification, and charging—are coordinated in protein synthesis according to cell states and conditions (Fig. 7). Translation is most active during the cell cycle when the cell doubles its protein mass. Under these conditions, most protein synthesis activity is devoted to the synthesis of the highly abundant house-keeping proteins, such as the ribosomal and cytoskeletal proteins. Here the abundance of tRNAs, and particularly the representation of tRNA isoacceptors, plays the most important role in translational regulation by accommodating the synthesis of those house-keeping proteins representing ~10–20% of human genes. Under these conditions, most tRNAs are highly charged,36 and the modification levels for most tRNAs are high, at least in the case of the Watson–Crick face methylations.58
Fig. 7.
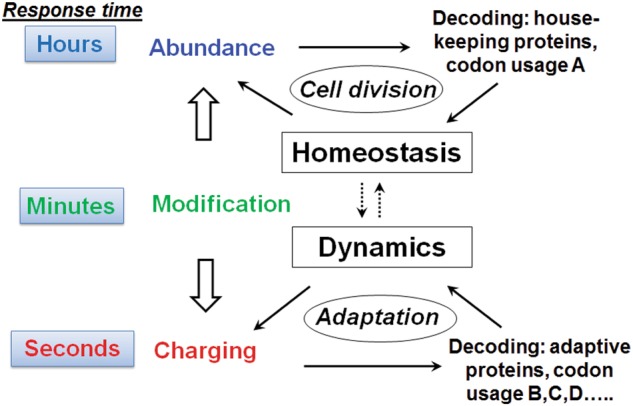
Dynamics of protein synthesis needs and tRNA response. During cell cycle progression, most translation activity is for the synthesis of highly abundant house-keeping proteins (~10–20% of all genes), and the tRNA abundance matters most. In stressful times or environmental fluctuations, translation activity switches to the synthesis of response or regulatory proteins (30–50% of all genes), to which tRNA charging significantly contributes. Modification is the “wild card” that tunes the regulation under all conditions
When cells are stressed or exposed to conditions where cell cycle progression is halted or severely slowed, the major protein synthetic activity switches from the production of house-keeping proteins to stress response or other regulatory proteins. Under stress, the level of charged tRNAs can rapidly change. Importantly, the charging levels among the tRNA isoacceptors for the same amino acid can become different and so become a useful parameter for the stress response or production of adaptive proteins for the new condition. At the transcriptomic level, selective or differential tRNA isoacceptor charging was first demonstrated directly in E. coli under amino acid starvation,89 which has significant consequences on the translational efficiency of mRNAs for many native E. coli proteins.90 Selective tRNA isoacceptor charging is derived in part by cells overexpressing certain isoacceptors beyond their needs for translating house-keeping proteins.91 These overproduced tRNA isoacceptors can remain at higher charging levels when stress or other changes to the environment halt the synthesis of house-keeping proteins. In a human cell line, starvation of an essential amino acid also leads to differential charging among tRNA isoacceptors for the corresponding starved amino acid (Fig. 8).
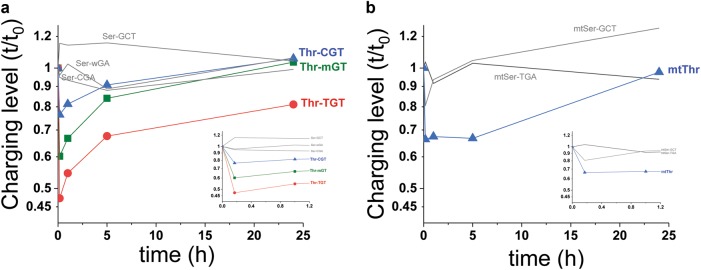
Fig. 8.
tRNA-charging level changes upon threonine starvation. Starvation of MDA-MB-231 cells was performed by exchanging the growth medium with the same medium lacking the amino acid threonine at t = 0. Total RNA was isolated at the indicated time points under mild acidic conditions (pH ~ 5) where tRNAs remained aminoacylated. Relative charging levels were measured using tRNA microarrays at the isoacceptor resolution.89, 111 a Nuclear-encoded tRNAThr and tRNASer. Thr-mGT corresponds to an array probe complementary to all Thr-AGT and three of the five Thr-CGT tRNA genes. Thr-CGT corresponds to two of the five Thr-CGT tRNA genes. b Mitochondrial-encoded tRNAThr and tRNASer. Inset shows the expanded view for early time points. Upon starvation, charging levels for all tRNAThr isoacceptors immediately dropped but to different degrees. Differential charging remained for several hours. Mitochondrial tRNAThr recovered more slowly than the cytosolic tRNAThr. Charging levels did not drop for tRNASer
Human tRNA modifications can also change reflecting culture conditions with different concentrations of glucose.86 Glucose deprivation increased the expression of the m1A58 eraser protein ALKBH1, resulting in decreased m1A levels for several tRNAs. As the m1A58 modification increased the effective concentration of tRNA for translation, a reduction of m1A58 levels decreased translation under low-glucose conditions. This study demonstrates that tRNA modification levels can respond to distinct culture conditions to adapt to the efficacy of translation.
The three main properties of tRNA exhibit dynamic changes with a distinct time scale (Fig. 7). Mature mammalian tRNAs are extremely stable with a half-life on the order of 100 h.92 Based on cellular tRNA/ribosome ratios (~10 on average) and protein synthesis rate on the ribosome (~2–5 peptide bonds per second on average), charged tRNAs should turn over every few seconds in a human cell when protein synthesis is fully active as occurs, for example, during cell cycle progression. Although tRNA modification dynamics remain to be determined in human cells, the presence of eraser enzymes would enable demodification reactions within a few minutes or less, as is typical for enzymatic reactions on macromolecules. The distinct dynamic range of distinct tRNA properties from seconds to hours or even days enables tRNAs to behave as either rapidly or slowly reacting responders to protein synthesis under wide ranging conditions.
Our efforts to develop high-throughput sequencing methods for tRNA now allow the measurement of all three tRNA properties in a single-sequencing reaction36,58,62 (Fig. 9). As mentioned above, the DM-tRNA-seq approach combines enzymatic demethylation and a processive thermophilic RT for measurement of tRNA abundance and modification. Extending this method to tRNA charging involves a periodate oxidation/β-elimination reaction of the total RNA before demethylase treatment. This chemical reaction removes the 3′A residue in all uncharged tRNAs, while charged tRNAs remain intact. Since all tRNAs end with 3′CCA, uncharged tRNAs end with 3′C, while charged tRNAs end with 3′A in this “one-pot” sequencing reaction. This method is applicable to any RNA sample from cell lines and tissues.
Fig. 9.
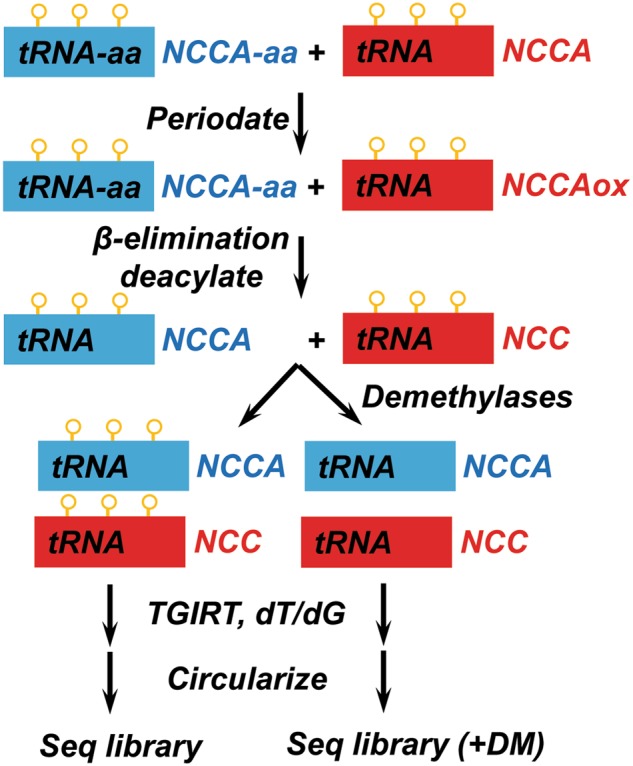
DM-tRNA-seq can measure tRNA abundance, modification, and charging in a single-sequencing library. Periodate only oxidizes uncharged tRNA. β-Elimination removes the oxidized 3′ nucleotides and deacylates charged tRNAs at the same time. The sample is then split into two, and one part treated with demethylases to remove m1A, m3C, and m1G (orange circles). Both samples are subjected to cDNA synthesis by a processive RT with both dT/dG-ending template-switching primers in case of TGIRT. cDNAs are circularized, followed by PCR to build sequencing libraries
Extra–translational tRNA–protein interactions
At a cellular concentration of tens of millions of copies (up to 100 µM) per cell, tRNAs are well suited for interactions with proteins not directly involved in protein synthesis, even at µM affinity. The GCN2 kinase–tRNA interaction is perhaps the best-studied system that serves an extratranslational function for tRNA.93–97 GCN2 is a protein kinase with a kinase domain that phosphorylates eIF2α and a regulatory domain that senses uncharged tRNA levels to activate the kinase domain. eIF2α is a conserved eukaryotic initiation factor involved in delivering the initiator tRNAMet to the scanning 40S ribosome. Phosphorylated eIF2α is inactive; therefore, the phospho-eIF2α level regulates the global translation activity. Proliferating cells have full translation activity and little uncharged tRNAs are available to activate the GCN2 kinase. Under stress or nutrient limitation, the uncharged tRNA level increases, which results in GCN2 activation and increased eIF2α phosphorylation either through a direct tRNA–GCN2 interaction or mediated through the ribosome. This decreases the global translation activity in time of stress or nutrient limitation.
Cell apoptosis can be triggered by the mitochondrial release of the cytochrome C protein to the cytosol that activates caspases. tRNA can bind the cytochrome C protein to block its binding to the caspase activator Apaf-1.98 tRNA binding prevents caspase activation under conditions where spurious disruption of mitochondria would not result in apoptosis, thereby ensuring that apoptosis only takes place when the cytosolic cytochrome C concentration has reached a certain threshold.
The interferon response to infection is mediated by the interferon-induced tetratricopeptide repeat (IFIT) protein family. The IFIT5 protein binds tRNA and may modulate the activity of the RIG-I protein, a cellular sensor for viruses.99,100 The cellular HIV restriction factors Sfn11 and Hili bind tRNA to reduce HIV virus replication.101,102 Both proteins reduce the translation of the HIV Gag protein in a codon usage-dependent manner. Since high levels of Gag protein translation are required for HIV replication, and because HIV genes have codon usages distinct from human genes, the interaction of these proteins with tRNA may selectively reduce the available amount of “rare” tRNAs to below a threshold for efficient Gag protein translation.
My laboratory has developed a systematic approach to identify protein candidates that interact with tRNA in cells.103 Our approach began with a computational, machine-learning analysis in which the known crystal structures of tRNA–protein complexes were used to compose Chemical Context Profiles (CCP). The CCP value scores the relevance of individual amino acid and nucleotide functional groups in the RNA–protein interaction interface. To identify candidates of new tRNA-binding proteins, 100,000 three-dimensional (3D) tRNA–protein decoys were built for each of >800 mammalian proteins or protein domains in the protein database. These 3D models were then scored according to the CCPs obtained from the learning set above. This approach identified 37 mammalian proteins as candidates for tRNA binding; none was previously known to be an RNA-binding protein. Using the ultraviolet crosslinking and immunoprecipitation approach104 commonly used in the field to study protein–RNA interactions, we experimentally validated tRNA binding for six of these predicted proteins in HEK293T cells.
One of the new tRNA-binding proteins is the mitogen-activated protein kinase kinase (MEK), which is required for cell cycle progression. We further investigated the property and potential function of the MEK–tRNA interaction in vitro and in several cell lines.105 We found that tRNA inhibits MEK2 kinase activity, consistent with our working hypothesis that tRNA provides an interface between translation activity and cell cycle progression. Cell cycle progression requires high levels of translation because of the synthesis of the very abundant house-keeping proteins. High-level translation utilizes most of the cellular tRNAs so that only a small amount of non-ribosomal-associated tRNAs would be available for interaction with other proteins. When translation activity decreases for any reason, more “free” tRNA becomes available for interacting with proteins that regulate other cellular processes. The tRNA-mediated inhibition of MEK protein kinase activity would slow down cell cycle progression, enabling cells to wait until translation can resume at full speed.
Future perspectives and challenges
A major challenge in human tRNA biology is to elucidate the function of tRNA isodecoders. The conservation of isodecoder genes in the mammalian genomes and the demonstration of a CNS-specific function of the tRNAArg(UCU) isodecoder indicate that many isodecoders are likely expressed uniquely in different tissues and cell types. Specific tRNA isodecoders could enhance cell type-specific translation in a codon or even mRNA-dependent manner. Other tRNA isodecoders may show preferred interactions with other cellular proteins for extratranslational functions.
Another major future challenge is the complete mapping of human tRNA modifications and assignment of their roles in biological regulation. As of today, <50% of nuclear-encoded human tRNAs have their modifications mapped in just one cell type. The complete modification map of the other human tRNAs remains to be charted. For each tRNA, the modification fraction at some sites may vary in a cell type- and cell state-dependent manner. How dynamic changes of tRNA modifications affect different biological processes remains to be determined.
A third challenge is to reveal how cells integrate the three tRNA properties of abundance, modification, and charging to achieve robust regulation of translation under all conditions. Translational regulation involves the temporal and spatial interplay between all three tRNA properties. So far, the primary focus has been on the role of tRNA abundance in translation. How rapidly changing charging levels and the dynamics of tRNA modification complement tRNA abundance to achieve optimal regulation remains to be determined.
Although not covered in this article, tRNA fragments have become increasingly important in a wide range of biological processes. These small RNAs are derived from the conditional cleavage of mature tRNAs or tRNA precursors by cellular nucleases. Given the extremely high levels of tRNA in a human cell, converting just a few percentage of mature tRNA into fragments would generate millions of small tRNA-derived molecules that can directly interact with cellular proteins or mRNAs. How much a role modifications play in tRNA fragment biology is a fascinating question that is only beginning to emerge.
Finally, the field requires a major technological advance to carry out single-cell tRNA sequencing. Single-cell transcriptome sequencing has become a mainstream for biological studies and has uncovered many surprising cellular physiologies such as new subtypes of cells in organs that are previously considered homogenous.106,107 Currently, tRNA-seq is much more difficult to carry out than mRNA-seq and so facilitating such studies would be a major technical achievement. The effort should well be worth it, as single-cell tRNA-seq may reveal unique expression and/or modification patterns that reflect regulatory mechanisms to adjust demand for protein synthesis in individual cells.
Acknowledgements
I thank NIH (DP1GM105386) and DoD/CDMRP (BC160450) for their generous support for our tRNA research. I thank Dr. Mariana Pavon Eternod for the tRNA charging results in Fig. 8.
Competing interests
The author declares that he has no conflict of interest.
References
- 1.Pavon-Eternod M, et al. tRNA over-expression in breast cancer and functional consequences. Nucleic Acids Res. 2009;37:7268–7280. doi: 10.1093/nar/gkp787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gingold H, et al. A dual program for translation regulation in cellular proliferation and differentiation. Cell. 2014;158:1281–1292. doi: 10.1016/j.cell.2014.08.011. [DOI] [PubMed] [Google Scholar]
- 3.Goodarzi H, et al. Modulated expression of specific tRNAs drives gene expression and cancer progression. Cell. 2016;165:1416–1427. doi: 10.1016/j.cell.2016.05.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Abbott JA, Francklyn CS, Robey-Bond SM. Transfer RNA and human disease. Front. Genet. 2014;5:158. doi: 10.3389/fgene.2014.00158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brandon MC, et al. MITOMAP: a human mitochondrial genome database–2004 update. Nucleic Acids Res. 2005;33:D611–D613. doi: 10.1093/nar/gki079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goto Y, Nonaka I, Horai S. A mutation in the tRNA(Leu)(UUR) gene associated with the MELAS subgroup of mitochondrial encephalomyopathies. Nature. 1990;348:651–653. doi: 10.1038/348651a0. [DOI] [PubMed] [Google Scholar]
- 7.Shoffner JM, et al. Myoclonic epilepsy and ragged-red fiber disease (MERRF) is associated with a mitochondrial DNA tRNA(Lys) mutation. Cell. 1990;61:931–937. doi: 10.1016/0092-8674(90)90059-n. [DOI] [PubMed] [Google Scholar]
- 8.Blanco S, et al. Aberrant methylation of tRNAs links cellular stress to neuro-developmental disorders. EMBO J. 2014;33:2020–2039. doi: 10.15252/embj.201489282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Najmabadi H, et al. Deep sequencing reveals 50 novel genes for recessive cognitive disorders. Nature. 2011;478:57–63. doi: 10.1038/nature10423. [DOI] [PubMed] [Google Scholar]
- 10.Guy MP, et al. Defects in tRNA anticodon loop 2′-O-methylation are implicated in nonsyndromic X-linked intellectual disability due to mutations in FTSJ1. Hum. Mutat. 2015;36:1176–1187. doi: 10.1002/humu.22897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Michaud J, et al. Isolation and characterization of a human chromosome 21q22.3 gene (WDR4) and its mouse homologue that code for a WD-repeat protein. Genomics. 2000;68:71–79. doi: 10.1006/geno.2000.6258. [DOI] [PubMed] [Google Scholar]
- 12.Shaheen R, et al. A homozygous truncating mutation in PUS3 expands the role of tRNA modification in normal cognition. Hum. Genet. 2016;135:707–713. doi: 10.1007/s00439-016-1665-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yamasaki S, Ivanov P, Hu GF, Anderson P. Angiogenin cleaves tRNA and promotes stress-induced translational repression. J. Cell Biol. 2009;185:35–42. doi: 10.1083/jcb.200811106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ivanov P, Emara MM, Villen J, Gygi SP, Anderson P. Angiogenin-induced tRNA fragments inhibit translation initiation. Mol. Cell. 2011;43:613–623. doi: 10.1016/j.molcel.2011.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maute RL, et al. tRNA-derived microRNA modulates proliferation and the DNA damage response and is down-regulated in B cell lymphoma. Proc. Natl. Acad. Sci. USA. 2013;110:1404–1409. doi: 10.1073/pnas.1206761110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Saikia M, et al. Angiogenin-cleaved tRNA halves interact with cytochrome c, protecting cells from apoptosis during osmotic stress. Mol. Cell Biol. 2014;34:2450–2463. doi: 10.1128/MCB.00136-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Goodarzi H, et al. Endogenous tRNA-derived fragments suppress breast cancer progression via YBX1 displacement. Cell. 2015;161:790–802. doi: 10.1016/j.cell.2015.02.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schaffer AE, et al. CLP1 founder mutation links tRNA splicing and maturation to cerebellar development and neurodegeneration. Cell. 2014;157:651–663. doi: 10.1016/j.cell.2014.03.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Honda S, et al. Sex hormone-dependent tRNA halves enhance cell proliferation in breast and prostate cancers. Proc. Natl. Acad. Sci. USA. 2015;112:E3816–E3825. doi: 10.1073/pnas.1510077112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sharma U, et al. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science. 2016;351:391–396. doi: 10.1126/science.aad6780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen Q, et al. Sperm tsRNAs contribute to intergenerational inheritance of an acquired metabolic disorder. Science. 2016;351:397–400. doi: 10.1126/science.aad7977. [DOI] [PubMed] [Google Scholar]
- 22.Kirchner S, Ignatova Z. Emerging roles of tRNA in adaptive translation, signalling dynamics and disease. Nat. Rev. Genet. 2015;16:98–112. doi: 10.1038/nrg3861. [DOI] [PubMed] [Google Scholar]
- 23.Suzuki T, Nagao A. Human mitochondrial tRNAs: biogenesis, function, structural aspects, and diseases. Annu Rev. Genet. 2012;45:299–329. doi: 10.1146/annurev-genet-110410-132531. [DOI] [PubMed] [Google Scholar]
- 24.Torres AG, Batlle E, Ribas de Pouplana L. Role of tRNA modifications in human diseases. Trends Mol. Med. 2014;20:306–314. doi: 10.1016/j.molmed.2014.01.008. [DOI] [PubMed] [Google Scholar]
- 25.Anderson P, Ivanov P. tRNA fragments in human health and disease. FEBS Lett. 2014;588:4297–4304. doi: 10.1016/j.febslet.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schimmel P. The emerging complexity of the tRNA world: mammalian tRNAs beyond protein synthesis. Nat. Rev. Mol. Cell Biol. 2018;19:45–58. doi: 10.1038/nrm.2017.77. [DOI] [PubMed] [Google Scholar]
- 27.Bednarova A, et al. Lost in translation: defects in transfer RNA modifications and neurological disorders. Front. Mol. Neurosci. 2017;10:135. doi: 10.3389/fnmol.2017.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kumar P, Kuscu C, Dutta A. Biogenesis and function of transfer RNA-related fragments (tRFs) Trends Biochem. Sci. 2016;41:679–689. doi: 10.1016/j.tibs.2016.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chan PP, Lowe TM. GtRNAdb 2.0: an expanded database of transfer RNA genes identified in complete and draft genomes. Nucleic Acids Res. 2016;44:D184–D189. doi: 10.1093/nar/gkv1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Parisien M, Wang X, Pan T. Diversity of human tRNA genes from the 1000-genomes project. RNA Biol. 2013;10:1853–1867. doi: 10.4161/rna.27361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lowe TM, Eddy SR. tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res. 1997;25:955–964. doi: 10.1093/nar/25.5.955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rudinger-Thirion J, Lescure A, Paulus C, Frugier M. A misfolded tRNA isodecoder binds and neutralizes a 3′ UTR-embedded Alu element. Proc. Natl. Acad. Sci. USA. 2011;108:794–802. doi: 10.1073/pnas.1103698108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goodenbour JM, Pan T. Diversity of tRNA genes in eukaryotes. Nucleic Acids Res. 2006;34:6137–6146. doi: 10.1093/nar/gkl725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schmitt BM, et al. High-resolution mapping of transcriptional dynamics across tissue development reveals a stable mRNA-tRNA interface. Genome Res. 2014;24:1797–1807. doi: 10.1101/gr.176784.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kutter C, et al. Pol III binding in six mammals shows conservation among amino acid isotypes despite divergence among tRNA genes. Nat. Genet. 2011;43:948–955. doi: 10.1038/ng.906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Evans ME, Clark WC, Zheng G, Pan T. Determination of tRNA aminoacylation levels by high-throughput sequencing. Nucleic Acids Res. 2017;45:e133. doi: 10.1093/nar/gkx514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Geslain R, Pan T. Functional analysis of human tRNA isodecoders. J. Mol. Biol. 2010;396:821–831. doi: 10.1016/j.jmb.2009.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ishimura R, et al. Ribosome stalling induced by mutation of a CNS-specific tRNA causes neurodegeneration. Science. 2014;345:455–459. doi: 10.1126/science.1249749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mungall AJ, et al. The DNA sequence and analysis of human chromosome 6. Nature. 2003;425:805–811. doi: 10.1038/nature02055. [DOI] [PubMed] [Google Scholar]
- 40.Horton R, et al. Gene map of the extended human MHC. Nat. Rev. Genet. 2004;5:889–899. doi: 10.1038/nrg1489. [DOI] [PubMed] [Google Scholar]
- 41.Mercer TR, et al. The human mitochondrial transcriptome. Cell. 2011;146:645–658. doi: 10.1016/j.cell.2011.06.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dittmar KA, Goodenbour JM, Pan T. Tissue-specific differences in human transfer RNA expression. PLoS Genet. 2006;2:e221. doi: 10.1371/journal.pgen.0020221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.van Tol H, Stange N, Gross HJ, Beier H. A human and a plant intron-containing tRNATyr gene are both transcribed in a HeLa cell extract but spliced along different pathways. EMBO J. 1987;6:35–41. doi: 10.1002/j.1460-2075.1987.tb04715.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kato N, Hoshino H, Harada F. Minor serine tRNA containing anticodon NCA (C4 RNA) from human and mouse cells. Biochem. Int. 1983;7:635–645. [PubMed] [Google Scholar]
- 45.Powell CA, Nicholls TJ, Minczuk M. Nuclear-encoded factors involved in post-transcriptional processing and modification of mitochondrial tRNAs in human disease. Front. Genet. 2015;6:79. doi: 10.3389/fgene.2015.00079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Edmonds CG, Vestal ML, McCloskey JA. Thermospray liquid chromatography-mass spectrometry of nucleosides and of enzymatic hydrolysates of nucleic acids. Nucleic Acids Res. 1985;13:8197–8206. doi: 10.1093/nar/13.22.8197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhou S, Sitaramaiah D, Pomerantz SC, Crain PF, McCloskey JA. Tandem mass spectrometry for structure assignments of wye nucleosides from transfer RNA. Nucleosides Nucleotides Nucleic Acids. 2004;23:41–50. doi: 10.1081/ncn-120027816. [DOI] [PubMed] [Google Scholar]
- 48.Suzuki T, Suzuki T, Wada T, Saigo K, Watanabe K. Taurine as a constituent of mitochondrial tRNAs: new insights into the functions of taurine and human mitochondrial diseases. EMBO J. 2002;21:6581–6589. doi: 10.1093/emboj/cdf656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cai WM, et al. A platform for discovery and quantification of modified ribonucleosides in RNA: application to stress-induced reprogramming of tRNA modifications. Methods Enzymol. 2015;560:29–71. doi: 10.1016/bs.mie.2015.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Suzuki T, Suzuki T. A complete landscape of post-transcriptional modifications in mammalian mitochondrial tRNAs. Nucleic Acids Res. 2014;42:7346–7357. doi: 10.1093/nar/gku390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chan CT, et al. A quantitative systems approach reveals dynamic control of tRNA modifications during cellular stress. PLoS Genet. 2010;6:e1001247. doi: 10.1371/journal.pgen.1001247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gu C, Begley TJ, Dedon PC. tRNA modifications regulate translation during cellular stress. FEBS Lett. 2014;588:4287–4296. doi: 10.1016/j.febslet.2014.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chan CT, et al. Reprogramming of tRNA modifications controls the oxidative stress response by codon-biased translation of proteins. Nat. Commun. 2012;3:937. doi: 10.1038/ncomms1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Roundtree IA, Evans ME, Pan T, He C. Dynamic RNA modifications in gene expression regulation. Cell. 2017;169:1187–1200. doi: 10.1016/j.cell.2017.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kawarada L, et al. ALKBH1 is an RNA dioxygenase responsible for cytoplasmic and mitochondrial tRNA modifications. Nucleic Acids Res. 2017;45:7401–7415. doi: 10.1093/nar/gkx354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ebhardt HA, et al. Meta-analysis of small RNA-sequencing errors reveals ubiquitous post-transcriptional RNA modifications. Nucleic Acids Res. 2009;37:2461–2470. doi: 10.1093/nar/gkp093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ryvkin P. HAMR: high-throughput annotation of modified ribonucleotides. RNA. 2013;19:1684–1692. doi: 10.1261/rna.036806.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Clark WC, Evans ME, Dominissini D, Zheng G, Pan T. tRNA base methylation identification and quantification via high-throughput sequencing. RNA. 2016;22:1771–1784. doi: 10.1261/rna.056531.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hauenschild R, et al. The reverse transcription signature of N-1-methyladenosine in RNA-Seq is sequence dependent. Nucleic Acids Res. 2015;43:9950–9964. doi: 10.1093/nar/gkv895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kuksa PP, et al. In silico identification of RNA modifications from high-throughput sequencing data using HAMR. Methods Mol. Biol. 2017;1562:211–229. doi: 10.1007/978-1-4939-6807-7_14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mohr S, et al. Thermostable group II intron reverse transcriptase fusion proteins and their use in cDNA synthesis and next-generation RNA sequencing. RNA. 2013;19:958–970. doi: 10.1261/rna.039743.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zheng G. Efficient and quantitative high-throughput tRNA sequencing. Nat. Methods. 2015;12:835–837. doi: 10.1038/nmeth.3478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wei FY, et al. Deficit of tRNA(Lys) modification by Cdkal1 causes the development of type 2 diabetes in mice. J. Clin. Invest. 2011;121:3598–3608. doi: 10.1172/JCI58056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Novoa EM, Pavon-Eternod M, Pan T, Ribas de Pouplana L. A role for tRNA modifications in genome structure and codon usage. Cell. 2012;149:202–213. doi: 10.1016/j.cell.2012.01.050. [DOI] [PubMed] [Google Scholar]
- 65.Liu RJ, et al. The tRNA recognition mechanism of the minimalist SPOUT methyltransferase, TrmL. Nucleic Acids Res. 2013;41:7828–7842. doi: 10.1093/nar/gkt568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bjork GR, Huang B, Persson OP, Bystrom AS. A conserved modified wobble nucleoside (mcm5s2U) in lysyl-tRNA is required for viability in yeast. RNA. 2007;13:1245–1255. doi: 10.1261/rna.558707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nedialkova DD, Leidel SA. Optimization of codon translation rates via tRNA modifications maintains proteome integrity. Cell. 2015;161:1606–1618. doi: 10.1016/j.cell.2015.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Fu D, et al. Human AlkB homolog ABH8 Is a tRNA methyltransferase required for wobble uridine modification and DNA damage survival. Mol. Cell Biol. 2010;30:2449–2459. doi: 10.1128/MCB.01604-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Songe-Moller L, et al. Mammalian ALKBH8 possesses tRNA methyltransferase activity required for the biogenesis of multiple wobble uridine modifications implicated in translational decoding. Mol. Cell Biol. 2010;30:1814–1827. doi: 10.1128/MCB.01602-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.van den Born E, et al. ALKBH8-mediated formation of a novel diastereomeric pair of wobble nucleosides in mammalian tRNA. Nat. Commun. 2011;2:172. doi: 10.1038/ncomms1173. [DOI] [PubMed] [Google Scholar]
- 71.Fu Y, et al. The AlkB domain of mammalian ABH8 catalyzes hydroxylation of 5-methoxycarbonylmethyluridine at the wobble position of tRNA. Angew. Chem. Int. Ed. Engl. 2010;49:8885–8888. doi: 10.1002/anie.201001242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Begley U, et al. A human tRNA methyltransferase 9-like protein prevents tumour growth by regulating LIN9 and HIF1-alpha. EMBO Mol. Med. 2013;5:366–383. doi: 10.1002/emmm.201201161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Roe BA, et al. Sequence studies on tRNAPhe from placenta: comparison with known sequences of tRNAPhe from other normal mammalian tissues. Biochem. Biophys. Res. Commun. 1975;66:1097–1105. doi: 10.1016/0006-291x(75)90470-2. [DOI] [PubMed] [Google Scholar]
- 74.Bjork GR, Wikstrom PM, Bystrom AS. Prevention of translational frameshifting by the modified nucleoside 1-methylguanosine. Science. 1989;244:986–989. doi: 10.1126/science.2471265. [DOI] [PubMed] [Google Scholar]
- 75.Christian T, Evilia C, Williams S, Hou YM. Distinct origins of tRNA(m1G37) methyltransferase. J. Mol. Biol. 2004;339:707–719. doi: 10.1016/j.jmb.2004.04.025. [DOI] [PubMed] [Google Scholar]
- 76.Klassen R, Bruch A, Schaffrath R. Independent suppression of ribosomal + 1 frameshifts by different tRNA anticodon loop modifications. RNA Biol. 2016;14:1–8. doi: 10.1080/15476286.2016.1267098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Powell CA, et al. TRMT5 mutations cause a defect in post-transcriptional modification of mitochondrial tRNA associated with multiple respiratory-chain deficiencies. Am. J. Hum. Genet. 2015;97:319–328. doi: 10.1016/j.ajhg.2015.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Voigts-Hoffmann F, et al. A methyl group controls conformational equilibrium in human mitochondrial tRNA(Lys) J. Am. Chem. Soc. 2007;129:13382–13383. doi: 10.1021/ja075520+. [DOI] [PubMed] [Google Scholar]
- 79.Helm M, Giege R, Florentz C. A Watson-Crick base-pair-disrupting methyl group (m1A9) is sufficient for cloverleaf folding of human mitochondrial tRNALys. Biochemistry. 1999;38:13338–13346. doi: 10.1021/bi991061g. [DOI] [PubMed] [Google Scholar]
- 80.Igoillo-Esteve M, et al. tRNA methyltransferase homolog gene TRMT10A mutation in young onset diabetes and primary microcephaly in humans. PLoS Genet. 2013;9:e1003888. doi: 10.1371/journal.pgen.1003888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shao Z, et al. Crystal structure of tRNA m1G9 methyltransferase Trm10: insight into the catalytic mechanism and recognition of tRNA substrate. Nucleic Acids Res. 2014;42:509–525. doi: 10.1093/nar/gkt869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Liu J, Straby KB. The human tRNA(m(2)(2)G(26))dimethyltransferase: functional expression and characterization of a cloned hTRM1 gene. Nucleic Acids Res. 2000;28:3445–3451. doi: 10.1093/nar/28.18.3445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Arimbasseri AG, et al. RNA polymerase III output is functionally linked to tRNA dimethyl-G26 modification. PLoS Genet. 2015;11:e1005671. doi: 10.1371/journal.pgen.1005671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Cozen AE, et al. ARM-seq: AlkB-facilitated RNA methylation sequencing reveals a complex landscape of modified tRNA fragments. Nat. Methods. 2015;12:879–884. doi: 10.1038/nmeth.3508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kadaba S, et al. Nuclear surveillance and degradation of hypomodified initiator tRNAMet in S. cerevisiae. Genes Dev. 2004;18:1227–1240. doi: 10.1101/gad.1183804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Liu F, Clark W, Luo G, et al. ALKBH1-mediated tRNA demethylation regulates translation. Cell. 2016;167:816–828. doi: 10.1016/j.cell.2016.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ozanick S, Krecic A, Andersland J, Anderson JT. The bipartite structure of the tRNA m1A58 methyltransferase from S. cerevisiae is conserved in humans. RNA. 2005;11:1281–1290. doi: 10.1261/rna.5040605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chujo T, Suzuki T. Trmt61B is a methyltransferase responsible for 1-methyladenosine at position 58 of human mitochondrial tRNAs. RNA. 2012;18:2269–2276. doi: 10.1261/rna.035600.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Dittmar KA, Sorensen MA, Elf J, Ehrenberg M, Pan T. Selective charging of tRNA isoacceptors induced by amino-acid starvation. EMBO Rep. 2005;6:151–7.. doi: 10.1038/sj.embor.7400341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Subramaniam AR, Pan T, Cluzel P. Environmental perturbations lift the degeneracy of the genetic code to regulate protein levels in bacteria. Proc. Natl. Acad. Sci. USA. 2013;110:2419–2424. doi: 10.1073/pnas.1211077110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Elf J, Nilsson D, Tenson T, Ehrenberg M. Selective charging of tRNA isoacceptors explains patterns of codon usage. Science. 2003;300:1718–1722. doi: 10.1126/science.1083811. [DOI] [PubMed] [Google Scholar]
- 92.Choe BK, Taylor MW. Kinetics of synthesis and characterization of transfer-RNA precursors in mammalian-cells. Biochim. Biophys. Acta. 1972;272:275–287. [Google Scholar]
- 93.Wek RC, Jiang HY, Anthony TG. Coping with stress: eIF2 kinases and translational control. Biochem. Soc. Trans. 2006;34:7–11. doi: 10.1042/BST20060007. [DOI] [PubMed] [Google Scholar]
- 94.Castilho BA, et al. Keeping the eIF2 alpha kinase Gcn2 in check. Biochim. Biophys. Acta. 2014;1843:1948–1968. doi: 10.1016/j.bbamcr.2014.04.006. [DOI] [PubMed] [Google Scholar]
- 95.Grallert B, Boye E. GCN2, an old dog with new tricks. Biochem Soc. Trans. 2013;41:1687–1691. doi: 10.1042/BST20130210. [DOI] [PubMed] [Google Scholar]
- 96.Ishimura R, Nagy G, Dotu I, Chuang JH, Ackerman SL. Activation of GCN2 kinase by ribosome stalling links translation elongation with translation initiation. Elife. 2016;5:e14295. doi: 10.7554/eLife.14295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Dong J, Qiu H, Garcia-Barrio M, Anderson J, Hinnebusch AG. Uncharged tRNA activates GCN2 by displacing the protein kinase moiety from a bipartite tRNA-binding domain. Mol. Cell. 2000;6:269–279. doi: 10.1016/s1097-2765(00)00028-9. [DOI] [PubMed] [Google Scholar]
- 98.Mei Y, Yong J, Liu H, et al. tRNA binds to cytochrome c and inhibits caspase activation. Mol. Cell. 2010;37:668–678. doi: 10.1016/j.molcel.2010.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Katibah GE, et al. tRNA binding, structure, and localization of the human interferon-induced protein IFIT5. Mol. Cell. 2013;49:743–750. doi: 10.1016/j.molcel.2012.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Katibah GE, et al. Broad and adaptable RNA structure recognition by the human interferon-induced tetratricopeptide repeat protein IFIT5. Proc. Natl. Acad. Sci. USA. 2014;111:12025–12030. doi: 10.1073/pnas.1412842111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Li M, Kao E, Gao X, et al. Codon-usage-based inhibition of HIV protein synthesis by human schlafen 11. Nature. 2012;491:125–128. doi: 10.1038/nature11433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Peterlin BM, et al. Hili inhibits HIV replication in activated T cells. J. Virol. 2017;91:e00237–17. doi: 10.1128/JVI.00237-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Parisien M. Discovering RNA-protein interactome by using chemical context profiling of the RNA-protein interface. Cell Rep. 2013;3:1703–1713. doi: 10.1016/j.celrep.2013.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ule J, Jensen K, Mele A, Darnell RB. CLIP: a method for identifying protein-RNA interaction sites in living cells. Methods. 2005;37:376–386. doi: 10.1016/j.ymeth.2005.07.018. [DOI] [PubMed] [Google Scholar]
- 105.Wang X, et al. Interaction of tRNA with MEK2 in pancreatic cancer cells. Sci. Rep. 2016;6:28260. doi: 10.1038/srep28260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Stubbington MJT, Rozenblatt-Rosen O, Regev A, Teichmann SA. Single-cell transcriptomics to explore the immune system in health and disease. Science. 2017;358:58–63. doi: 10.1126/science.aan6828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lein E, Borm LE, Linnarsson S. The promise of spatial transcriptomics for neuroscience in the era of molecular cell typing. Science. 2017;358:64–69. doi: 10.1126/science.aan6827. [DOI] [PubMed] [Google Scholar]
- 108.Sprinzl M, Vassilenko KS. Compilation of tRNA sequences and sequences of tRNA genes. Nucleic Acids Res. 2005;33:D139–D140. doi: 10.1093/nar/gki012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Cantara WA, et al. The RNA Modification Database, RNAMDB: 2011 update. Nucleic Acids Res. 2011;39:D195–D201. doi: 10.1093/nar/gkq1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Machnicka MA, et al. MODOMICS: a database of RNA modification pathways–2013 update. Nucleic Acids Res. 2013;41:D262–D267. doi: 10.1093/nar/gks1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Zaborske JM, et al. Genome-wide analysis of tRNA charging and activation of the eIF2 kinase Gcn2p. J. Biol. Chem. 2009;284:25254–25267. doi: 10.1074/jbc.M109.000877. [DOI] [PMC free article] [PubMed] [Google Scholar]