Abstract
Objective
To estimate the prevalence and risk factors for DED among US men.
Methods
Cross-sectional prevalence survey among male participants aged 50y and older in the Physicians’ Health Studies I (N=18,596) and II (N=6,848). We defined DED as the presence of clinically diagnosed dry eye or severe symptoms (both dryness and irritation constantly or often). We calculated the age-standardized prevalence of DED adjusted to the age distribution of US men in 2004, and projected estimates forward to 2030. We compared DED prevalence to a similar cohort of women, and examined associations with possible risk factors.
Results
The prevalence of DED increased with age, from 3.90% among men 50–54y old to 7.67% among men 80y and older (P for trend <0.0001). High blood pressure (OR=1.28; CI=1.12–1.45) and benign prostatic hyperplasia (OR=1.25; CI=1.09–1.44) were associated with a higher risk of DED. Use of antidepressants, antihypertensives, and medications to treat benign prostatic hyperplasia were also associated with increased risk of DED. The age-standardized prevalence of DED was 4.34%, or 1.68 million men aged 50y or older, and is expected to affect over 2.79 million US men by 2030.
Conclusions
DED is prevalent and increases with age, hypertension, benign prostatic hyperplasia and antidepressants.
Keywords: dry eye disease, keratoconjunctivitis sicca, gender, epidemiology, prevalence, risk factors
Introduction
Dry eye is one of the most prevalent eye diseases and reasons for seeking eye care among older people. It is an important public health problem, causing increased risk of ocular infections and bothersome symptoms of ocular discomfort, fatigue, and visual disturbance that interfere with crucial activities such as reading, working on a computer, and driving a car (1–3). Studies over the past decade have identified older age, female sex, reduced androgen levels, exogenous estrogen use, and an imbalance in the dietary intake of essential fatty acids as important risk factors for dry eye disease (DED) (1, 4–7). A large-scale study of participants’ in the Women’s Health Study estimated that 3.25 million US women aged 50 and older are affected with clinically important DED (8). The present study examined the prevalence and risk factors for DED among 25,444 middle-aged and older men participating in the Physicians’ Health Studies I and/or II (PHS I and PHS II).
Design and Methods
Study Subjects
The PHS 1 began in 1982 as a randomized, double-masked, placebo-controlled trial of aspirin and beta-carotene for the primary prevention of cardiovascular disease and cancer among 22,071 US male physicians (9). The PHS II is a randomized, double-masked, placebo-controlled factorial trial of alternate day beta-carotene, alternate day vitamin E, daily vitamin C, and a daily multivitamin in the prevention of cardiovascular disease, total and prostate cancer, cataract and macular degeneration (10). Beginning in 1999, after the end of the randomized component of PHS I, the PHS II enrolled 7644 willing and eligible PHS I participants, as well as 7000 newly recruited US physicians aged 55 years and older. The remaining PHS I participants who elected not to enroll in PHS II continue to be followed with annual questionnaires. Because of their medical training, PHS participants have reliably reported specific details about their health. Participants receive mailed questionnaires every year on which they record a number of health-related exposures and any health outcomes experienced over the previous year. The PHS I and II are approved by the Human Subjects Committee at Brigham and Women’s Hospital, and all subjects gave informed consent.
Ascertainment of DED
Assessment of DED was conducted following the previously reported and validated protocol that was also used in the Women’s Health Study (4, 6, 8, 11). We asked participants three questions pertaining to diagnosis or symptoms of DED: 1) ‘Have you ever been diagnosed by a clinician as having dry eye syndrome?’ 2) ‘How often do your eyes feel dry (not wet enough)?’ 3) ‘How often do your eyes feel irritated?’ Possible answers to the two questions about symptoms included ‘constantly’, ‘often’, ‘sometimes’, or ‘never’. These questions were previously found to provide high specificity for the diagnosis of dry eye. For our primary analyses, we defined DED as the presence of either a previous clinical diagnosis of DED or severe symptoms (both dryness and irritation either constantly or often). We also analyzed separately clinically diagnosed DED and severe DED symptoms.
Statistical Analysis
We combined data from the PHS I and PHS II and calculated the prevalence of DED in this study population overall, as well as according to age in 5-year categories, geographic region (South, West, Midwest, Northeast, or other, based on US census regions), and race/ethnicity (White, Black/African American, Hispanic, Asian/Pacific Islander, Native American/Alaskan Native, or other). Then, using US census figures for US men in 2004 and projected estimates for 2030, we used the direct-adjustment method to calculate age-adjusted prevalence of DED among US men currently, and projected estimates for the year 2030, respectively. Projections assumed current rates of DED. We also compared rates of DED in women versus men by directly standardizing and comparing rates derived from the Women’s Health Study (for women) and PHS (for men) using US census estimates for the total population in 2000. To evaluate potential risk factors, we constructed multivariable logistic regression models using a dichotomous outcome variable that indicated whether or not a study participant had DED, and indicator variables for each demographic characteristic or other potential risk factor. From these models, we obtained odds ratios (OR) and their corresponding ninety-five percent confidence intervals (CI). The risk factors evaluated were selected based on specific hypotheses regarding dry eye pathogenesis, and included age, benign prostatic hyperplasia, and history of hypertension (treated and untreated) and diabetes.
In the PHS II population, we had additional information on some commonly used medication groups that have biologically plausible associations with DED. Thus, in this subgroup, we further evaluated possible associations with reported use medications for treatment of benign prostatic hyperplasia and hypertension, as well as reported use of antidepressants and statin medications. Finally, we also had information on diagnosis of rheumatoid arthritis in this subgroup and evaluated its association with DED.
Results
We assessed the prevalence of dry eye among 25444 men, including 18596 original participants in the PHS I, and 6848 men who did not participate in PHS I but were randomized into the PHS II, representing 93.8% of men who were potentially eligible for this study of DED. The age range of the men in the study was from 50 to 99 years (median=64.4 years) and 90.6% of the men where white (Table 1). The men resided in all regions of the US, with the largest proportion living in the South (39.1%). A total of 1103 men (4.3%) reported a clinical diagnosis or severe symptoms of DED.
Table 1.
Demographic Characteristics of the N=25444 Participants in the Physicians’ Health Studies I and II with Information on Dry Eye Disease
| Characteristic | Total in Category | Number (%) With DED* |
|---|---|---|
| Age | ||
| 50 – 54 | 2513 | 98 (3.90) |
| 55 – 59 | 5936 | 197 (3.32) |
| 60 – 64 | 4805 | 145 (3.02) |
| 65 – 69 | 4326 | 173 (4.00) |
| 70 – 74 | 3554 | 184 (5.18) |
| 75 – 79 | 2433 | 162 (6.66) |
| 80 + | 1877 | 144 (7.67) |
| Race/ethnicity | ||
| White | 23056 | 993 (4.31) |
| African American | 609 | 30 (4.93) |
| Asian/Pacific Islander | 241 | 14 (5.81) |
| Hispanic | 1182 | 53 (4.48) |
| Unknown, Other | 356 | 13 (3.65) |
| Region of residence† | ||
| West | 5668 | 249 (4.39) |
| Midwest | 6561 | 284 (4.33) |
| Northeast | 5622 | 233 (4.14) |
| South | 7417 | 324 (4.37) |
| Outside of US (including Puerto Rico, Guam and other US territories) | 176 | 13 (7.39) |
DED = dry eye disease, defined as a reported clinical diagnosis or symptoms of both dryness and irritation either constantly or often
According to US census region
The distribution of symptoms in the population showed 204 men (0.8%) who experienced dryness constantly, 924 (3.6%) often, 4716 (18.5%) sometimes, and 19597 (77.0%) never. For symptoms of irritation, 126 (0.5%) men reported experiencing irritation constantly, 1050 (4.1%) often, 10622 (41.8%) sometimes, and 13644 (53.6%) never. Considering both symptoms together, the proportion of the population experiencing at least one symptom sometimes or more frequently was 49.2%, whereas 21.8% of men reported both symptoms at least sometimes. There were 6.8% of men who reported experiencing at least one symptom constantly or often, and 2.2% who reported experiencing both dryness and irritation either constantly or often. Less than half (40.7%) of the men with severe symptoms reported a history of a diagnosis of dry eye, and the probability of having such a diagnosis increased significantly with age (P<0.001). Overall, diagnosed dry eye was reported by 765 men (3.0%). Of the 765 men with diagnosed dry eye, 232 men (30.3%) also reported severe symptoms, 528 men (69.0%) had less severe symptoms, and 5 men (0.7%) did not provide data on dry eye symptoms. Among men with diagnosed dry eye, older men tended to be more likely than younger men to have a concurrent complaint of severe symptoms, but this trend was not statistically significant (P=0.14).
Men aged 75 years and older were more likely to have DED. The observed prevalence rose from 3.9% among men aged 50–54 years to 7.7% among men aged 80 and older (P for trend<0.0001). Adjusting the observed age-specific prevalence of DED in the PHS to US census estimates for men in 2004 resulted in an overall population adjusted estimate of 4.4% for men aged 50 and older in the US. This translated into an estimated 1.68 million men aged 50 and older in the US with dry eye. Given that DED becomes more prevalent among the 75 years and older group, we were interested to see what impact the projected growth of the older segments of our population would have on the magnitude of the DED among the US male population. Estimates obtained using projected 2030 census figures and the observed prevalence of DED in the PHS, showed a predicted growth to 2.79 million US men affected by DED in 2030.
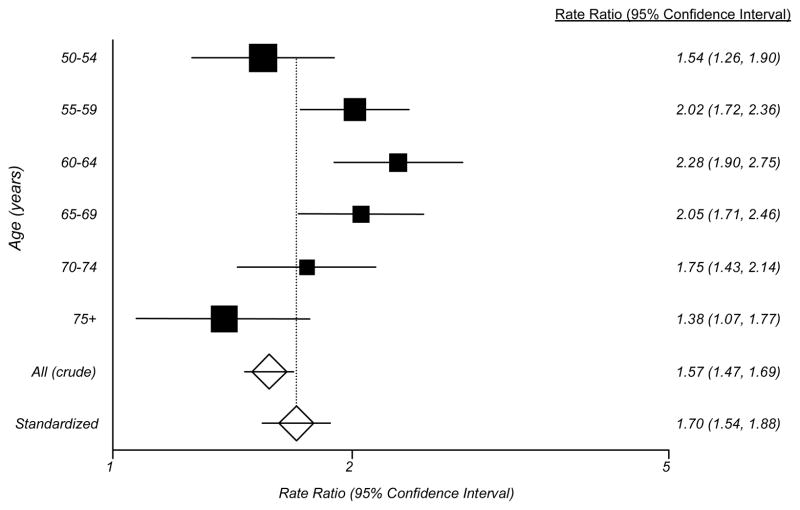
For comparison of the prevalence of DED in men and women, we calculated the ratio of prevalence of dry eye among women versus men in each age group and used the age distribution of the US population as a whole (men and women) as a common standard to calculate an age-standardized prevalence ratio. These analyses demonstrated a significantly higher prevalence of DED among women in all age groups (Figure), and the age-standardized ratio reflected a 70% higher prevalence among women versus men in the 50 years and older group as a whole (Figure).
Figure 1.
The Figure depicts the rate ratio for dry eye disease within 5-year age groups among 36,995 women participating in the Women’s Health Study [REF] compared to the prevalence observed in the present study of 25,444 men participating in the Physicians’ Health Studies I and II. The black boxes indicate the rate ratios and the horizontal bars show the 95% confidence interval for the rate ratio within each age group. The size of the box is proportional to the number of subjects in each age group. The unfilled diamonds show the overall rate ratio comparing the prevalence of dry eye disease in women versus men in these two studies based on the observed data (top diamond), an age-standardized estimate derived by using the age distribution of the US population as a whole (men and women) as a common standard to calculate an age-standardized prevalence ratio (bottom diamond). The horizontal lines passing through the diamonds depict the 95% confidence intervals for these rate ratios.
After controlling for age and other variables, there were no substantial differences in the prevalence of DED among men by either race or geographic region of the US (Table 2). There was also no significant difference in the prevalence of DED among men with and without diabetes mellitus. In contrast, men with treated or untreated hypertension (OR=1.28, CI=1.12 to 1.45) and men with benign prostatic hyperplasia (OR=1.26, CI=1.09 to 1.44) were significantly more likely to have DED. In an alternative model in which we considered only treated cases of hypertension, we also observed a significant association between use of antihypertensive medications and DED (OR=1.28, CI=1.12 to 1.46).
Table 2.
Predictors of Clinically Important Dry Eye Disease in US Men
| Variable | Odds Ratio | 95% Confidence Interval |
|---|---|---|
| Age | ||
| 50 – 54 | 1.00 | |
| 55 – 59 | 0.81 | 0.64–1.04 |
| 60 – 64 | 0.72 | 0.55–0.93 |
| 65 – 69 | 0.92 | 0.71–1.20 |
| 70 – 74 | 1.18 | 0.92–1.53 |
| 75 – 79 | 1.51 | 1.15–1.97 |
| 80 + | 1.76 | 1.34–2.32 |
| Race/ethnicity | ||
| White | 1.00 | |
| African American | 1.13 | 0.76–1.68 |
| Asian/Pacific Islander | 1.36 | 0.79–2.35 |
| Hispanic | 1.25 | 0.93–1.67 |
| Unknown, Other | 0.93 | 0.53–1.63 |
| Region of residence | ||
| South | 1.00 | |
| West | 0.93 | 0.53–1.63 |
| Midwest | 1.01 | 0.85–1.18 |
| Northeast | 0.96 | 0.81–1.14 |
| Other | 1.61 | 0.85–3.04 |
| Hypertension | ||
| No | 1.00 | |
| Yes | 1.28 | 1.12–1.45 |
| Benign Prostatic Hyperplasia | ||
| No | 1.00 | |
| Yes | 1.26 | 1.09–1.44 |
| Diabetes Mellitus | ||
| No | 1.00 | |
| Yes | 0.97 | 0.74–1.24 |
Information on medication use and rheumatoid arthritis was available among 6034 men, among whom 446 had DED. After controlling for age and other variables, we observed a significant association between the use of medications to treat benign prostatic hyperplasia (OR=1.35, CI=1.01 to 1.80) and DED. In addition, men who used antidepressants had a nearly two-fold higher risk of DED compared to men who did not use these drugs (OR=1.90, CI=1.39 to 2.61). On the other hand, there was no significant association between use of antihypertensive medications and DED among this subgroup of men (OR=1.15, CI=0.93 to 1.43). Use of statin drugs was not associated with DED (OR=1.07, CI=0.85 to 1.34). Finally, there was a non-significant trend toward a higher risk of DED men with rheumatoid arthritis (OR=1.90, CI=0.90 to 4.02), however the confidence interval was wide due to the small number of men with this diagnosis (n=53).
Discussion
The present study estimates that approximately 1.68 million men aged 50 and older are affected with DED in the US. These data derived from studying over 25000 men, show a significantly lower prevalence of DED than was found in a similar study using the same methodology among US women, among whom the prevalence was estimated at 3.23 million women (8). Nonetheless, there is a significant increase in the prevalence of DED among with age among men, as is the case among women; and there is a predicted growth to 2.79 million US men affected by DED in 2030. There are no significant variations in the prevalence of DED among the racial/ethnic groups represented in the PHS, or within US geographical regions. Hypertension and benign prostatic hyperplasia are associated with an increase in the prevalence of DED in this population. Medications associated with DED included drugs to treat hypertension and benign prostatic hyperplasia, and antidepressants.
Since the prevalence of DED is related to factors such as hypertension and benign prostatic hyperplasia, the prevalence estimates we present might differ somewhat from the true prevalence among US men because our study was carried out in a cohort of initially healthy male physicians. The PHS population includes men from across the US, however, and although participants were required to be free of cardiovascular disease and cancer at the start of the PHS, many of them had developed comorbid conditions during their many years of participation in the PHS and prior to our assessment of DED. Other epidemiological studies of DED have been restricted to single towns or cities (12–15) and these populations are also likely to differ from the general US population in important ways. Nevertheless, since one prior study found a higher prevalence of DED among people with a higher number of comorbidities (14), and we also find significant associations with two common medical conditions in this age group, it is important to keep in mind that prevalence estimates may be affected by the prevalence of comorbid conditions in any population studied.
Another potential limitation relates to our assessment of DED using self-reports of clinically diagnosed dry eye and severe symptoms. There is growing international consensus, however, that such methods are valuable for studies of DED and provide useful information about its public health significance because these symptoms have a substantial impact on vision related quality of life in DED patients (1, 2, 16). Clinicians in practice also rely heavily on the evaluation of dry eye symptoms for both the diagnosis of DED and patients’ responses to treatment (17, 18). Our validated questionnaire and strict criteria to identify men as having DED has been shown to achieve a good balance of sensitivity and specificity versus commonly used clinical tests for DED (11). It remains possible that some of the men in our study may have experienced symptoms of both dryness and irritation from something other than dry eye, but it was not possible to perform standardized clinical examinations in such a large and geographically dispersed cohort. Several other factors such as the lack of correspondence between symptoms and other clinical signs of DED, under-diagnosis of the condition in the US population, the healthy nature of our study populations, and the high threshold for our definition of DED based on symptoms alone, suggest that our estimates might represent a lower bound of the prevalence of DED in the population and that the true prevalence, particularly of less severe disease, is likely to be substantially higher. The estimates we provide of the prevalence of DED are based on the presence of either severe symptoms or a reported clinical diagnosis of dry eye and should accordingly encompass many clinically significant cases with only milder symptoms as well as undiagnosed cases of DED with severe symptoms.
DED has received recognition as a common ocular problem for older women, but there have been fewer data to describe the magnitude of the problem and its risk factors among US men. Estimates from the present study, the largest study to date on DED among men, show that 1.68 million men in the US have DED of sufficient severity to meet our definition. Because of the use of similar populations and methodologies, we were able to directly compare this estimate to our prior estimate of the prevalence of DED among women and these analyses confirm the lower prevalence of DED among men. The reason for this is not entirely clear, but several lines of evidence implicate a role for sex steroid hormone levels.
The meibomian gland is a large modified sebaceous gland that produces the tear film lipid layer. Aging is associated with the development of meibomian gland dysfunction, which promotes tear film instability and evaporative dry eye. Of interest, some of the anatomical and physiological modifications of the meibomian gland during aging have been linked to androgen deficiency (19), concurring with androgenic control of sebaceous glands in the skin (20). Both the observations of a higher prevalence of dry eye among women and an increasing prevalence of DED with aging in both sexes are also consistent with the hypothesis that loss of androgen support can contribute to an increased risk of DED. Androgen levels are higher in men than in women throughout life, and both sexes experience a decline in bioavailable androgen levels during aging (21–24). The range of effects of the decline in androgen bioavailability with aging are only beginning to be elucidated, and it seems plausible that these age-related declines in the androgen pool might be related to the development of DED.
We also found that men with a diagnosis of benign prostatic hyperplasia and those using medications to treat this condition were more likely to have DED. We speculate that these findings could possibly relate to use of anti-androgen therapy, as a prior clinical study showed that men receiving anti-androgen drugs had significant adverse changes in several measures of ocular surface health (25, 26). Indeed, androgen deficiency could provide a unifying explanation for observations of a higher prevalence of DED in aging, menopause, complete androgen insufficiency syndrome, and Sjogren’s syndrome (27–30). The lack of data specific to the use of anti-androgen medications in the present study is a major limitation, however, that leaves the door open to confounding or chance. In that regard, the use of alpha-1 adrenergic receptor blockers is an even more common treatment for benign prostatic hyperplasia than anti-androgens are. As the conjunctiva is reported to express alpha-1 receptors (31), potential effects of these drugs on the ocular surface may be plausible, though it is unclear whether they would be beneficial or harmful. Future study of the possible effects of these medications on the ocular surface is needed to address these issues.
We observed a strong association between the use of antidepressant medications and risk of DED, a finding consistent with data from at least two other epidemiological studies (32, 33). In contrast, dry eye is not common in the reporting of clinical trials of antidepressant drugs, whereas dry mouth is prevalent (34, 35). However, this may be a methodological issue, as side effects in such trials are often assessed by spontaneous reporting by patients or by closed-ended lists that do not include dry eye (36). For example, a recent study presented patients with a list of 17 common side effects reported by patients taking antidepressants. The only ocular symptom on the list was blurred vision, which was reported by 11% of patients. In light of the knowledge that DED is known to cause fluctuating visual blur, one wonders whether the blurred vision reported by patients could have been due at least in part to DED.
Men with hypertension were also more likely to report dry eye in the present study, and treated hypertension was significant associated with DED in the full cohort. A previous cross-sectional study found a significantly higher prevalence of symptoms of dry eye or dry mouth among people who took diuretics or other hypotensive medications (33). A more recent study of found a borderline increased risk of DED with use of diuretics, but a decreased risk of DED with use of ACE inhibitors (32). In our subgroup analysis on a smaller group of men, we did not find a significant association with the use of antihypertensive medications generally, but specific data on which medications were being used (i.e. diuretics versus ACE inhibitors or other types) was not available and may have diluted underlying associations.
In the present study there was no association between use of statins and risk of DED. We examined this association in view of reported immunosuppressive effects of statins that could impact development of DED (37). In addition, statins were shown to decrease ocular surface damage in graft-versus-host disease, a T-cell mediated condition in which dry eye is a frequent complication (38). However, although T cells are also implicated in mediating development of DED (39, 40), we did not observe an association with statins in this study. Future studies of systemic medications and risk of DED would be informative.
In summary, we estimate that 1.68 million US men aged 50 and older currently suffer from clinically important DED, and this number is likely to grow to an estimated 2.79 million by 2030. Although this number is only about half the estimated prevalence of DED among women in the same age group, it points to a substantial burden of disease in this segment of the US population. Our data indicating a high frequency of less severe symptoms, suggest that the inclusion of milder cases of DED would swell prevalence estimates considerably. Given the increasing recognition of the adverse visual impact of DED, and the high level of bother patients report due to its irritative symptoms, we hope that these data from a large and well-characterized group of US men will provide further motivation for clinicians and researchers to understand this disease and develop more effective and targeted interventions for patients.
Acknowledgments
Financial Support: NIH grants EY00365, CA40360; and The Joint Clinical Research Center, Massachusetts Eye and Ear Infirmary and Schepens Eye Research Institute, Boston MA.
References
- 1.The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007) Ocul Surf. 2007;5(2):93–107. doi: 10.1016/s1542-0124(12)70082-4. [DOI] [PubMed] [Google Scholar]
- 2.The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007) Ocul Surf. 2007;5(2):75–92. doi: 10.1016/s1542-0124(12)70081-2. [DOI] [PubMed] [Google Scholar]
- 3.Miljanovic B, Dana R, Sullivan DA, Schaumberg DA. Impact of dry eye syndrome on vision-related quality of life. Am J Ophthalmol. 2007;143(3):409–15. doi: 10.1016/j.ajo.2006.11.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Miljanovic B, Trivedi KA, Dana MR, et al. Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. Am J Clin Nutr. 2005;82(4):887–93. doi: 10.1093/ajcn/82.4.887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schaumberg DA, Buring JE, Sullivan DA, Dana MR. The epidemiology of dry eye syndrome. Cornea. 2000;19(Suppl):S120. [Google Scholar]
- 6.Schaumberg DA, Buring JE, Sullivan DA, Dana MR. Hormone replacement therapy and dry eye syndrome. JAMA. 2001;286(17):2114–9. doi: 10.1001/jama.286.17.2114. [DOI] [PubMed] [Google Scholar]
- 7.Schaumberg DA, Sullivan DA, Dana MR. The relationship of hormone replacement therapy with the prevalence of dry eye syndrome in a large cohort of women. Ophthalmology. 2000;107(Suppl):105. [Google Scholar]
- 8.Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003;136(2):318–26. doi: 10.1016/s0002-9394(03)00218-6. [DOI] [PubMed] [Google Scholar]
- 9.Steering Committee of the Physicians’ Health Study Research Group. Final report on the aspirin component of the ongoing Physicians’ Health Study [see comments] N Engl J Med. 1989;321(3):129–35. doi: 10.1056/NEJM198907203210301. [DOI] [PubMed] [Google Scholar]
- 10.Christen WG, Gaziano JM, Hennekens CH. Design of Physicians’ Health Study II--a randomized trial of beta-carotene, vitamins E and C, and multivitamins, in prevention of cancer, cardiovascular disease, and eye disease, and review of results of completed trials. Ann Epidemiol. 2000;10(2):125–34. doi: 10.1016/s1047-2797(99)00042-3. [DOI] [PubMed] [Google Scholar]
- 11.Gulati A, Sullivan R, Buring JE, et al. Validation and repeatability of a short questionnaire for dry eye syndrome. Am J Ophthalmol. 2006;142(1):125–131. doi: 10.1016/j.ajo.2006.02.038. [DOI] [PubMed] [Google Scholar]
- 12.Bjerrum KB. Keratoconjunctivitis sicca and primary Sjogren’s syndrome in a Danish population aged 30–60 years. Acta Ophthalmol Scand. 1997;75(3):281–6. doi: 10.1111/j.1600-0420.1997.tb00774.x. [DOI] [PubMed] [Google Scholar]
- 13.McCarty CA, Bansal AK, Livingston PM, Stanislavsky YL, Taylor HR. The epidemiology of dry eye in Melbourne, Australia. Ophthalmology. 1998;105(6):1114–9. doi: 10.1016/S0161-6420(98)96016-X. [DOI] [PubMed] [Google Scholar]
- 14.Schein OD, Munoz B, Tielsch JM, Bandeen-Roche K, West S. Prevalence of dry eye among the elderly. Am J Ophthalmol. 1997;124(6):723–8. doi: 10.1016/s0002-9394(14)71688-5. [DOI] [PubMed] [Google Scholar]
- 15.Moss SE, Klein R, Klein BE. Prevalence of and risk factors for dry eye syndrome. Arch Ophthalmol. 2000;118(9):1264–8. doi: 10.1001/archopht.118.9.1264. [DOI] [PubMed] [Google Scholar]
- 16.Methodologies to diagnose and monitor dry eye disease: report of the Diagnostic Methodology Subcommittee of the International Dry Eye WorkShop (2007) Ocul Surf. 2007;5(2):108–52. doi: 10.1016/s1542-0124(12)70083-6. [DOI] [PubMed] [Google Scholar]
- 17.Nichols KK, Nichols JJ, Zadnik K. Frequency of dry eye diagnostic test procedures used in various modes of ophthalmic practice. Cornea. 2000;19(4):477–82. doi: 10.1097/00003226-200007000-00015. [DOI] [PubMed] [Google Scholar]
- 18.Korb DR. Survey of preferred tests for diagnosis of the tear film and dry eye. Cornea. 2000;19(4):483–6. doi: 10.1097/00003226-200007000-00016. [DOI] [PubMed] [Google Scholar]
- 19.Sullivan BD, Evans JE, Dana MR, Sullivan DA. Influence of aging on the polar and neutral lipid profiles in human meibomian gland secretions. Arch Ophthalmol. 2006;124(9):1286–92. doi: 10.1001/archopht.124.9.1286. [DOI] [PubMed] [Google Scholar]
- 20.Fritsch M, Orfanos CE, Zouboulis CC. Sebocytes are the key regulators of androgen homeostasis in human skin. J Invest Dermatol. 2001;116(5):793–800. doi: 10.1046/j.1523-1747.2001.01312.x. [DOI] [PubMed] [Google Scholar]
- 21.Belanger A, Candas B, Dupont A, et al. Changes in serum concentrations of conjugated and unconjugated steroids in 40- to 80-year-old men. J Clin Endocrinol Metab. 1994;79(4):1086–90. doi: 10.1210/jcem.79.4.7962278. [DOI] [PubMed] [Google Scholar]
- 22.Labrie F, Belanger A, Cusan L, Gomez JL, Candas B. Marked decline in serum concentrations of adrenal C19 sex steroid precursors and conjugated androgen metabolites during aging. J Clin Endocrinol Metab. 1997;82(8):2396–402. doi: 10.1210/jcem.82.8.4160. [DOI] [PubMed] [Google Scholar]
- 23.Labrie F, Luu-The V, Belanger A, et al. Is dehydroepiandrosterone a hormone? J Endocrinol. 2005;187(2):169–96. doi: 10.1677/joe.1.06264. [DOI] [PubMed] [Google Scholar]
- 24.Liu PY, Beilin J, Meier C, et al. Age-related changes in serum testosterone and sex hormone binding globulin in Australian men: longitudinal analyses of two geographically separate regional cohorts. J Clin Endocrinol Metab. 2007;92(9):3599–603. doi: 10.1210/jc.2007-0862. [DOI] [PubMed] [Google Scholar]
- 25.Krenzer KL, Reza Dana M, Ullman MD, et al. Effect of androgen deficiency on the human meibomian gland and ocular surface. J Clin Endocrinol Metab. 2000;85(12):4874–82. doi: 10.1210/jcem.85.12.7072. [DOI] [PubMed] [Google Scholar]
- 26.Sullivan BD, Evans JE, Krenzer KL, Reza Dana M, Sullivan DA. Impact of antiandrogen treatment on the fatty acid profile of neutral lipids in human meibomian gland secretions. J Clin Endocrinol Metab. 2000;85(12):4866–73. doi: 10.1210/jcem.85.12.7066. [DOI] [PubMed] [Google Scholar]
- 27.Sullivan DA, Sullivan BD, Evans JE, et al. Androgen deficiency, Meibomian gland dysfunction, and evaporative dry eye. Ann N Y Acad Sci. 2002;966:211–22. doi: 10.1111/j.1749-6632.2002.tb04217.x. [DOI] [PubMed] [Google Scholar]
- 28.Sullivan DA, Yamagami H, Liu M, et al. Sex steroids, the meibomian gland and evaporative dry eye. Adv Exp Med Biol. 2002;506(Pt A):389–99. doi: 10.1007/978-1-4615-0717-8_56. [DOI] [PubMed] [Google Scholar]
- 29.Cermak JM, Krenzer KL, Sullivan RM, Dana MR, Sullivan DA. Is complete androgen insensitivity syndrome associated with alterations in the meibomian gland and ocular surface? Cornea. 2003;22(6):516–21. doi: 10.1097/00003226-200308000-00006. [DOI] [PubMed] [Google Scholar]
- 30.Sullivan BD, Evans JE, Cermak JM, et al. Complete androgen insensitivity syndrome: effect on human meibomian gland secretions. Arch Ophthalmol. 2002;120(12):1689–99. doi: 10.1001/archopht.120.12.1689. [DOI] [PubMed] [Google Scholar]
- 31.Enriquez de Salamanca A, Siemasko KF, Diebold Y, et al. Expression of muscarinic and adrenergic receptors in normal human conjunctival epithelium. Invest Ophthalmol Vis Sci. 2005;46(2):504–13. doi: 10.1167/iovs.04-0665. [DOI] [PubMed] [Google Scholar]
- 32.Moss SE, Klein R, Klein BE. Long-term Incidence of Dry Eye in an Older Population. Optom Vis Sci. 2008;85(8):668–74. doi: 10.1097/OPX.0b013e318181a947. [DOI] [PubMed] [Google Scholar]
- 33.Schein OD, Hochberg MC, Munoz B, et al. Dry eye and dry mouth in the elderly: a population-based assessment. Arch Intern Med. 1999;159(12):1359–63. doi: 10.1001/archinte.159.12.1359. [DOI] [PubMed] [Google Scholar]
- 34.Wilkes S. Bupropion. Drugs Today (Barc) 2006;42(10):671–81. doi: 10.1358/dot.2006.42.10.1025701. [DOI] [PubMed] [Google Scholar]
- 35.Hudson JI, Perahia DG, Gilaberte I, et al. Duloxetine in the treatment of major depressive disorder: an open-label study. BMC Psychiatry. 2007;7:43. doi: 10.1186/1471-244X-7-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hu XH, Bull SA, Hunkeler EM, et al. Incidence and duration of side effects and those rated as bothersome with selective serotonin reuptake inhibitor treatment for depression: patient report versus physician estimate. J Clin Psychiatry. 2004;65(7):959–65. doi: 10.4088/jcp.v65n0712. [DOI] [PubMed] [Google Scholar]
- 37.Kuipers HF, van den Elsen PJ. Immunomodulation by statins: inhibition of cholesterol vs. isoprenoid biosynthesis. Biomed Pharmacother. 2007;61(7):400–7. doi: 10.1016/j.biopha.2007.06.005. [DOI] [PubMed] [Google Scholar]
- 38.Hori A, Kanda Y, Goyama S, et al. A prospective trial to evaluate the safety and efficacy of pravastatin for the treatment of refractory chronic graft-versus-host disease. Transplantation. 2005;79(3):372–4. doi: 10.1097/01.tp.0000151001.64189.1d. [DOI] [PubMed] [Google Scholar]
- 39.Stern ME, Gao J, Schwalb TA, et al. Conjunctival T-cell subpopulations in Sjogren’s and non-Sjogren’s patients with dry eye. Invest Ophthalmol Vis Sci. 2002;43(8):2609–14. [PubMed] [Google Scholar]
- 40.Stern ME, Siemasko KF, Gao J, et al. Evaluation of ocular surface inflammation in the presence of dry eye and allergic conjunctival disease. Ocul Surf. 2005;3(4 Suppl):S161–4. doi: 10.1016/s1542-0124(12)70246-x. [DOI] [PubMed] [Google Scholar]