Abstract
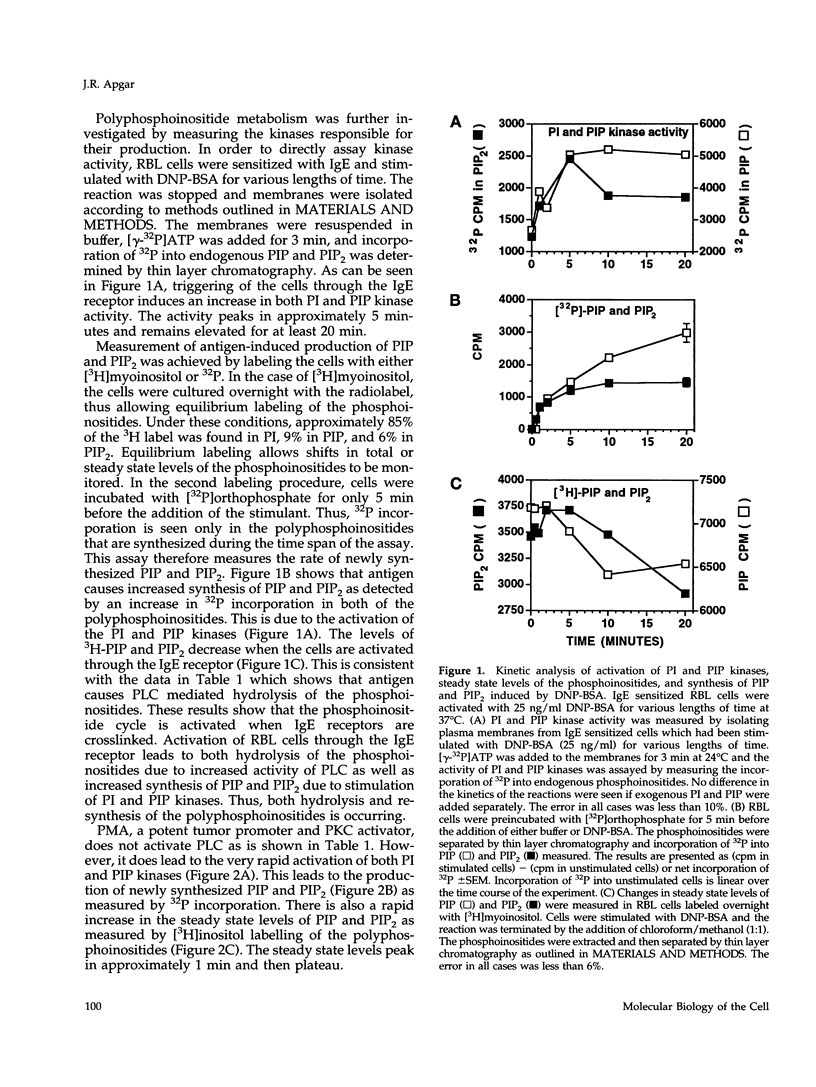
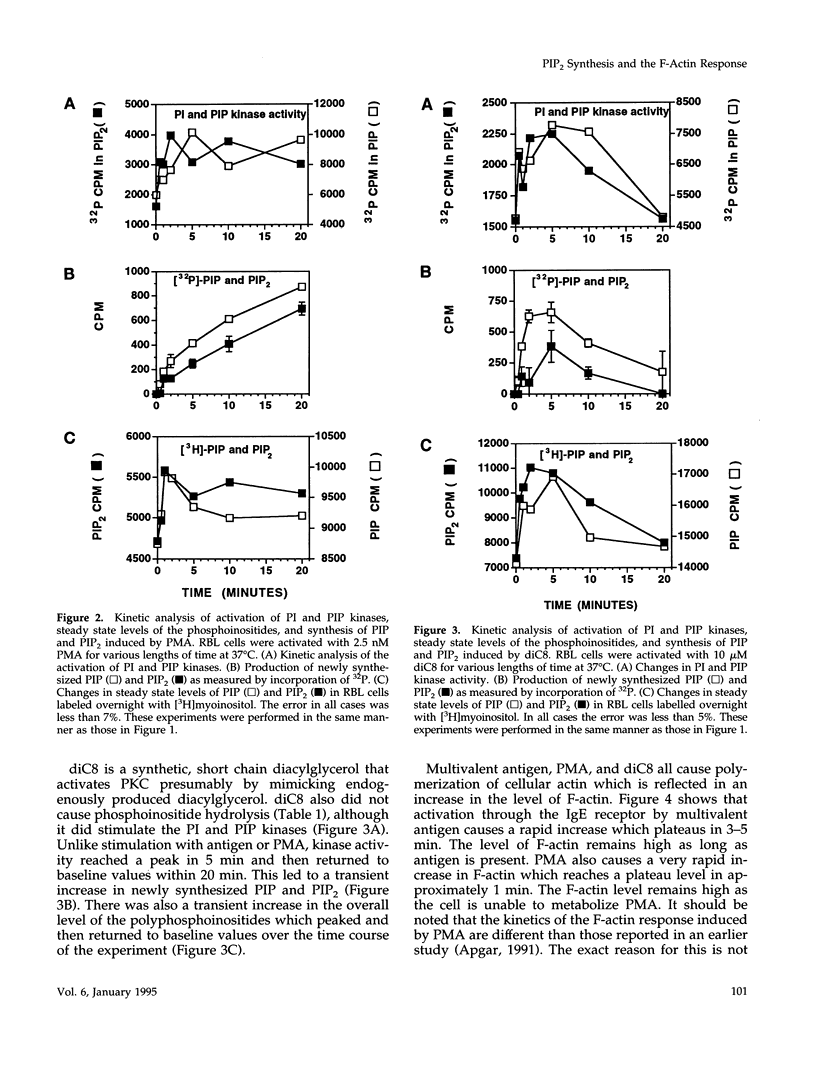
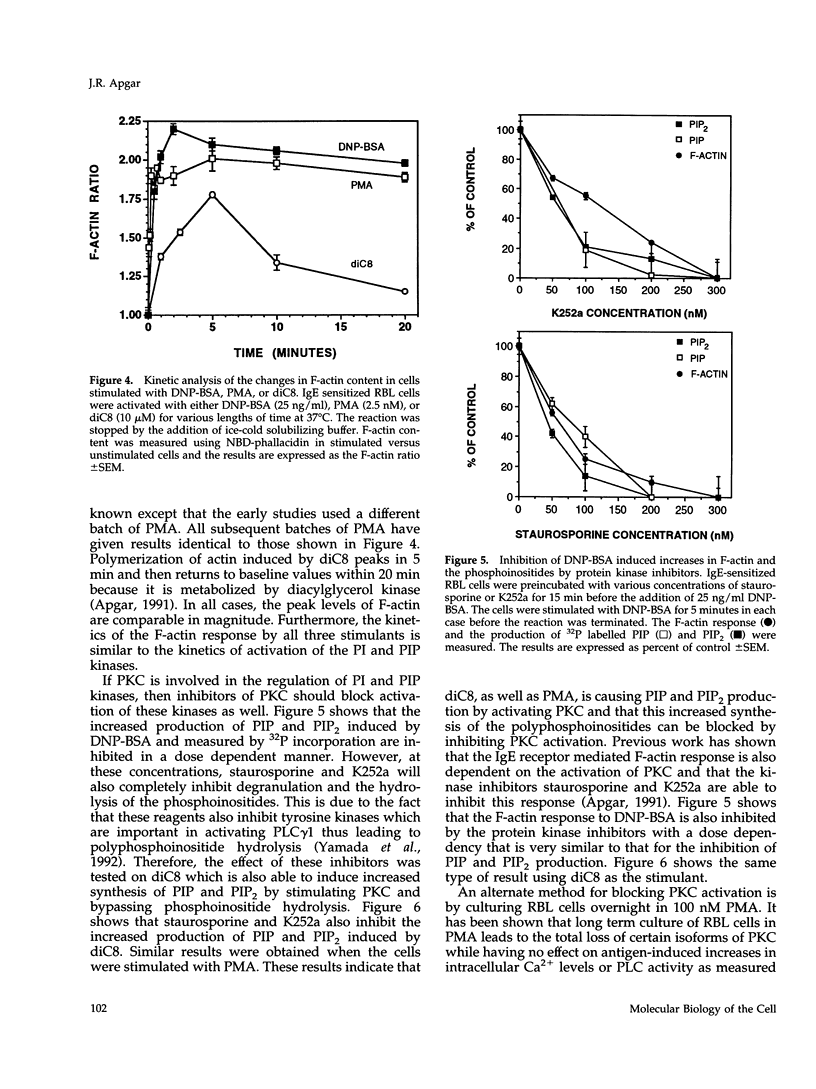
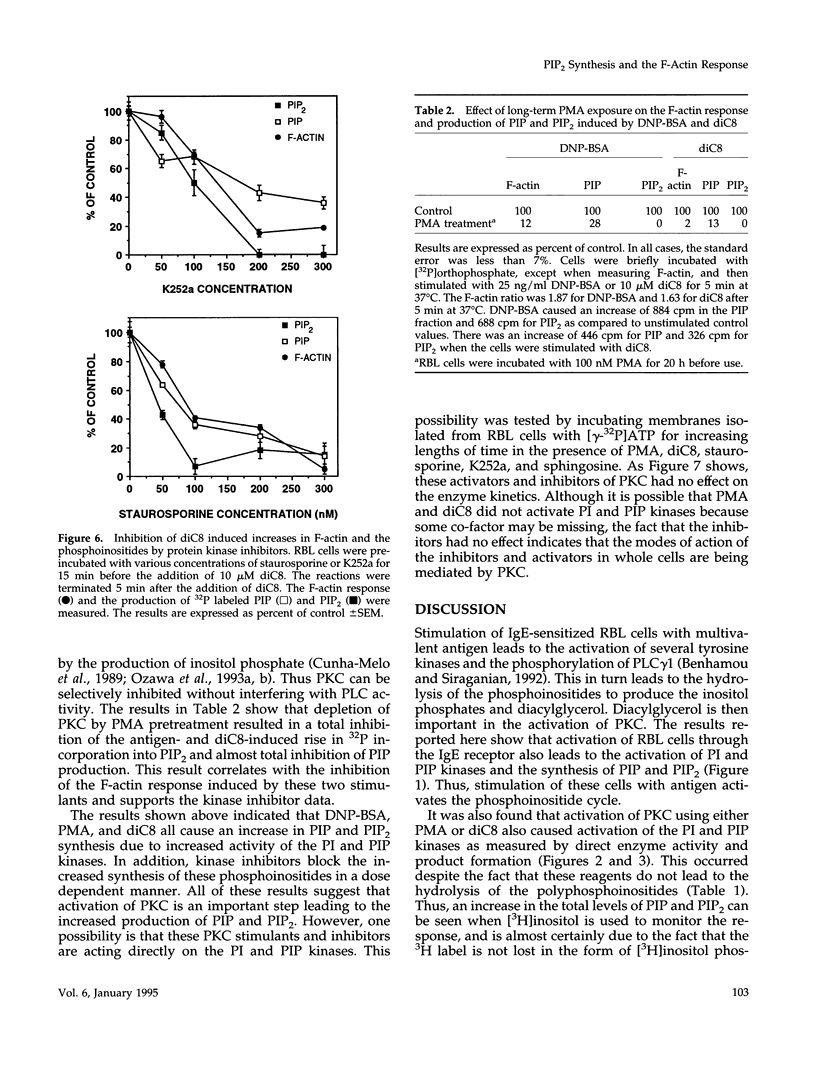
Cross-linking of the immunoglobulin E receptor on rat basophilic leukemia (RBL)1 cells by multivalent antigen activates phosphatidylinositol (PI) kinase and phosphatidylinositol 4-phosphate (PIP) kinase leading to the increased production of PIP and phosphatidylinositol 4,5-bisphosphate (PIP2). Activators of protein kinase C (PKC), such as phorbol myristate acetate (PMA) and the synthetic diacylglycerol, 1,2-dioctanoyl-sn-glycerol (diC8), were found to have the same effect even though PMA and diC8 do not cause the activation of phospholipase C. Although the kinetics are different depending on the stimulant, activation of PKC using multivalent antigen, PMA or diC8 also causes the polymerization of actin and an increase in the F-actin content of the cells. In all cases, a good correlation was observed between F-actin levels, activation of PI and PIP kinases, and the increased production of PIP and PIP2. However, in the case of antigen, there is no correlation between actin polymerization and the total amount of PIP and PIP2. Staurosporine, an inhibitor of protein kinases, blocks the F-actin response and the increased synthesis of PIP and PIP2 with similar dose dependencies. Furthermore, depletion of PKC activity through long-term exposure to PMA, inhibited both the F-actin response and the increased synthesis of PIP and PIP2 induced by either DNP-BSA or diC8. These results suggest that activation of PKC precedes the activation of PI and PIP kinases and that under certain circumstances activation of the kinases and the increased synthesis of PIP and PIP2 may be involved in the polymerization of actin in RBL cells, possibly through the interaction of the polyphosphoinositides with actin-binding proteins such as gelsolin and profilin.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apgar J. R. Polymerization of actin in RBL-2H3 cells can be triggered through either the IgE receptor or the adenosine receptor but different signaling pathways are used. Mol Biol Cell. 1994 Mar;5(3):313–322. doi: 10.1091/mbc.5.3.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
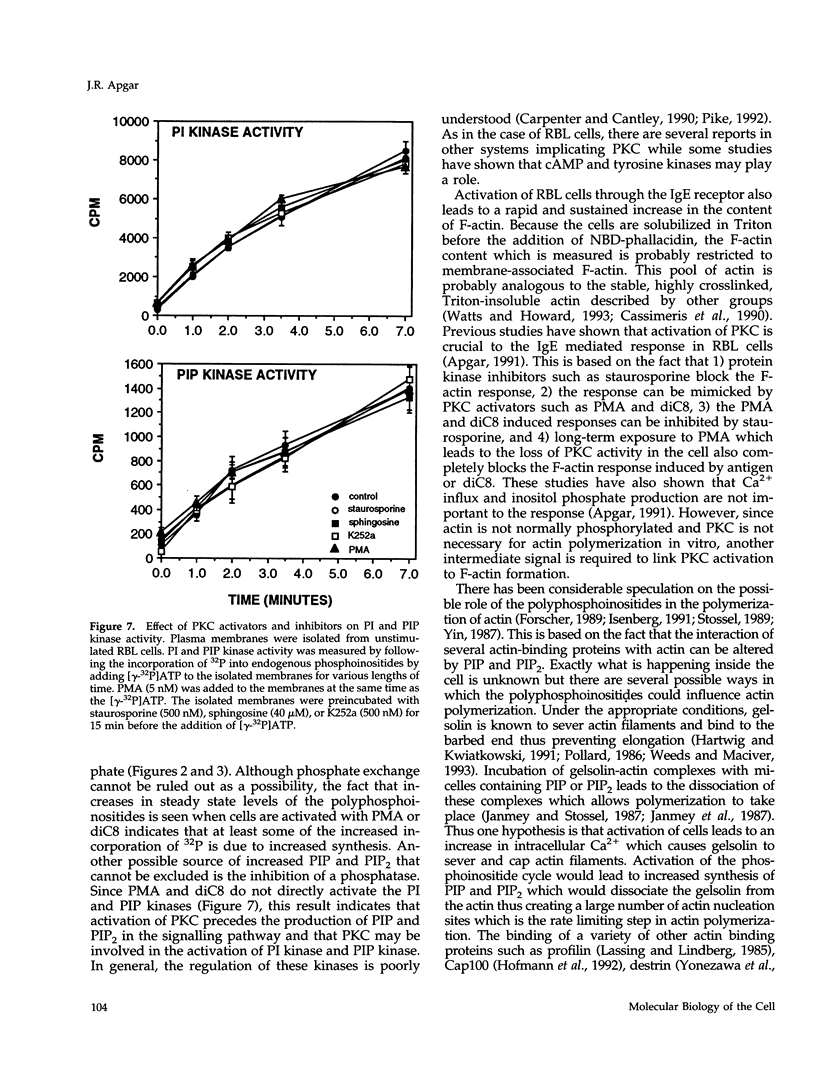
- Apgar J. R. Regulation of the antigen-induced F-actin response in rat basophilic leukemia cells by protein kinase C. J Cell Biol. 1991 Mar;112(6):1157–1163. doi: 10.1083/jcb.112.6.1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaven M. A., Cunha-Melo J. R. Membrane phosphoinositide-activated signals in mast cells and basophils. Prog Allergy. 1988;42:123–184. [PubMed] [Google Scholar]
- Beaven M. A., Metzger H. Signal transduction by Fc receptors: the Fc epsilon RI case. Immunol Today. 1993 May;14(5):222–226. doi: 10.1016/0167-5699(93)90167-j. [DOI] [PubMed] [Google Scholar]
- Benhamou M., Siraganian R. P. Protein-tyrosine phosphorylation: an essential component of Fc epsilon RI signaling. Immunol Today. 1992 Jun;13(6):195–197. doi: 10.1016/0167-5699(92)90152-w. [DOI] [PubMed] [Google Scholar]
- Berridge M. J., Downes C. P., Hanley M. R. Lithium amplifies agonist-dependent phosphatidylinositol responses in brain and salivary glands. Biochem J. 1982 Sep 15;206(3):587–595. doi: 10.1042/bj2060587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carpenter C. L., Cantley L. C. Phosphoinositide kinases. Biochemistry. 1990 Dec 25;29(51):11147–11156. doi: 10.1021/bi00503a001. [DOI] [PubMed] [Google Scholar]
- Carson M., Weber A., Zigmond S. H. An actin-nucleating activity in polymorphonuclear leukocytes is modulated by chemotactic peptides. J Cell Biol. 1986 Dec;103(6 Pt 2):2707–2714. doi: 10.1083/jcb.103.6.2707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassimeris L., McNeill H., Zigmond S. H. Chemoattractant-stimulated polymorphonuclear leukocytes contain two populations of actin filaments that differ in their spatial distributions and relative stabilities. J Cell Biol. 1990 Apr;110(4):1067–1075. doi: 10.1083/jcb.110.4.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunha-Melo J. R., Gonzaga H. M., Ali H., Huang F. L., Huang K. P., Beaven M. A. Studies of protein kinase C in the rat basophilic leukemia (RBL-2H3) cell reveal that antigen-induced signals are not mimicked by the actions of phorbol myristate acetate and Ca2+ ionophore. J Immunol. 1989 Oct 15;143(8):2617–2625. [PubMed] [Google Scholar]
- Dadabay C. Y., Patton E., Cooper J. A., Pike L. J. Lack of correlation between changes in polyphosphoinositide levels and actin/gelsolin complexes in A431 cells treated with epidermal growth factor. J Cell Biol. 1991 Mar;112(6):1151–1156. doi: 10.1083/jcb.112.6.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Downey G. P., Chan C. K., Lea P., Takai A., Grinstein S. Phorbol ester-induced actin assembly in neutrophils: role of protein kinase C. J Cell Biol. 1992 Feb;116(3):695–706. doi: 10.1083/jcb.116.3.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberle M., Traynor-Kaplan A. E., Sklar L. A., Norgauer J. Is there a relationship between phosphatidylinositol trisphosphate and F-actin polymerization in human neutrophils? J Biol Chem. 1990 Oct 5;265(28):16725–16728. [PubMed] [Google Scholar]
- Forscher P. Calcium and polyphosphoinositide control of cytoskeletal dynamics. Trends Neurosci. 1989 Nov;12(11):468–474. doi: 10.1016/0166-2236(89)90098-2. [DOI] [PubMed] [Google Scholar]
- Geiger B. Membrane-cytoskeleton interaction. Biochim Biophys Acta. 1983 Aug 11;737(3-4):305–341. doi: 10.1016/0304-4157(83)90005-9. [DOI] [PubMed] [Google Scholar]
- Goldschmidt-Clermont P. J., Furman M. I., Wachsstock D., Safer D., Nachmias V. T., Pollard T. D. The control of actin nucleotide exchange by thymosin beta 4 and profilin. A potential regulatory mechanism for actin polymerization in cells. Mol Biol Cell. 1992 Sep;3(9):1015–1024. doi: 10.1091/mbc.3.9.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldschmidt-Clermont P. J., Janmey P. A. Profilin, a weak CAP for actin and RAS. Cell. 1991 Aug 9;66(3):419–421. doi: 10.1016/0092-8674(81)90002-7. [DOI] [PubMed] [Google Scholar]
- Goldschmidt-Clermont P. J., Kim J. W., Machesky L. M., Rhee S. G., Pollard T. D. Regulation of phospholipase C-gamma 1 by profilin and tyrosine phosphorylation. Science. 1991 Mar 8;251(4998):1231–1233. doi: 10.1126/science.1848725. [DOI] [PubMed] [Google Scholar]
- Goldschmidt-Clermont P. J., Machesky L. M., Baldassare J. J., Pollard T. D. The actin-binding protein profilin binds to PIP2 and inhibits its hydrolysis by phospholipase C. Science. 1990 Mar 30;247(4950):1575–1578. doi: 10.1126/science.2157283. [DOI] [PubMed] [Google Scholar]
- Ha K. S., Exton J. H. Activation of actin polymerization by phosphatidic acid derived from phosphatidylcholine in IIC9 fibroblasts. J Cell Biol. 1993 Dec;123(6 Pt 2):1789–1796. doi: 10.1083/jcb.123.6.1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartwig J. H., Kwiatkowski D. J. Actin-binding proteins. Curr Opin Cell Biol. 1991 Feb;3(1):87–97. doi: 10.1016/0955-0674(91)90170-4. [DOI] [PubMed] [Google Scholar]
- Hofmann A., Eichinger L., André E., Rieger D., Schleicher M. Cap100, a novel phosphatidylinositol 4,5-bisphosphate-regulated protein that caps actin filaments but does not nucleate actin assembly. Cell Motil Cytoskeleton. 1992;23(2):133–144. doi: 10.1002/cm.970230206. [DOI] [PubMed] [Google Scholar]
- Howard T. H., Oresajo C. O. A method for quantifying F-actin in chemotactic peptide activated neutrophils: study of the effect of tBOC peptide. Cell Motil. 1985;5(6):545–557. doi: 10.1002/cm.970050609. [DOI] [PubMed] [Google Scholar]
- Howard T. H., Wang D. Calcium ionophore, phorbol ester, and chemotactic peptide-induced cytoskeleton reorganization in human neutrophils. J Clin Invest. 1987 May;79(5):1359–1364. doi: 10.1172/JCI112962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard T., Chaponnier C., Yin H., Stossel T. Gelsolin-actin interaction and actin polymerization in human neutrophils. J Cell Biol. 1990 Jun;110(6):1983–1991. doi: 10.1083/jcb.110.6.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isenberg G. Actin binding proteins--lipid interactions. J Muscle Res Cell Motil. 1991 Apr;12(2):136–144. doi: 10.1007/BF01774032. [DOI] [PubMed] [Google Scholar]
- Ishikawa H. Plasmalemmal undercoat: the cytoskeleton supporting the plasmalemma. Arch Histol Cytol. 1988 May;51(2):127–145. doi: 10.1679/aohc.51.127. [DOI] [PubMed] [Google Scholar]
- Jacobson B. S. Interaction of the plasma membrane with the cytoskeleton: an overview. Tissue Cell. 1983;15(6):829–852. doi: 10.1016/0040-8166(83)90053-8. [DOI] [PubMed] [Google Scholar]
- Janmey P. A., Iida K., Yin H. L., Stossel T. P. Polyphosphoinositide micelles and polyphosphoinositide-containing vesicles dissociate endogenous gelsolin-actin complexes and promote actin assembly from the fast-growing end of actin filaments blocked by gelsolin. J Biol Chem. 1987 Sep 5;262(25):12228–12236. [PubMed] [Google Scholar]
- Janmey P. A., Stossel T. P. Modulation of gelsolin function by phosphatidylinositol 4,5-bisphosphate. Nature. 1987 Jan 22;325(6102):362–364. doi: 10.1038/325362a0. [DOI] [PubMed] [Google Scholar]
- Lassing I., Lindberg U. Specific interaction between phosphatidylinositol 4,5-bisphosphate and profilactin. Nature. 1985 Apr 4;314(6010):472–474. doi: 10.1038/314472a0. [DOI] [PubMed] [Google Scholar]
- Lin P. Y., Wiggan G. A., Gilfillan A. M. Activation of phospholipase D in a rat mast (RBL 2H3) cell line. A possible unifying mechanism for IgE-dependent degranulation and arachidonic acid metabolite release. J Immunol. 1991 Mar 1;146(5):1609–1616. [PubMed] [Google Scholar]
- Lind S. E., Janmey P. A., Chaponnier C., Herbert T. J., Stossel T. P. Reversible binding of actin to gelsolin and profilin in human platelet extracts. J Cell Biol. 1987 Aug;105(2):833–842. doi: 10.1083/jcb.105.2.833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu F. T., Bohn J. W., Ferry E. L., Yamamoto H., Molinaro C. A., Sherman L. A., Klinman N. R., Katz D. H. Monoclonal dinitrophenyl-specific murine IgE antibody: preparation, isolation, and characterization. J Immunol. 1980 Jun;124(6):2728–2737. [PubMed] [Google Scholar]
- Metzger H., Alcaraz G., Hohman R., Kinet J. P., Pribluda V., Quarto R. The receptor with high affinity for immunoglobulin E. Annu Rev Immunol. 1986;4:419–470. doi: 10.1146/annurev.iy.04.040186.002223. [DOI] [PubMed] [Google Scholar]
- Metzger H. The receptor with high affinity for IgE. Immunol Rev. 1992 Feb;125:37–48. doi: 10.1111/j.1600-065x.1992.tb00624.x. [DOI] [PubMed] [Google Scholar]
- Moritz A., De Graan P. N., Gispen W. H., Wirtz K. W. Phosphatidic acid is a specific activator of phosphatidylinositol-4-phosphate kinase. J Biol Chem. 1992 Apr 15;267(11):7207–7210. [PubMed] [Google Scholar]
- Nachmias V. T. Small actin-binding proteins: the beta-thymosin family. Curr Opin Cell Biol. 1993 Feb;5(1):56–62. doi: 10.1016/s0955-0674(05)80008-0. [DOI] [PubMed] [Google Scholar]
- Norgauer J., Eberle M., Lemke H. D., Aktories K. Activation of human neutrophils by mastoparan. Reorganization of the cytoskeleton, formation of phosphatidylinositol 3,4,5-trisphosphate, secretion up-regulation of complement receptor type 3 and superoxide anion production are stimulated by mastoparan. Biochem J. 1992 Mar 1;282(Pt 2):393–397. doi: 10.1042/bj2820393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliver J. M., Berlin R. D. Mechanisms that regulate the structural and functional architecture of cell surfaces. Int Rev Cytol. 1982;74:55–94. doi: 10.1016/s0074-7696(08)61169-9. [DOI] [PubMed] [Google Scholar]
- Oliver J. M., Seagrave J., Stump R. F., Pfeiffer J. R., Deanin G. G. Signal transduction and cellular response in RBL-2H3 mast cells. Prog Allergy. 1988;42:185–245. [PubMed] [Google Scholar]
- Ozawa K., Szallasi Z., Kazanietz M. G., Blumberg P. M., Mischak H., Mushinski J. F., Beaven M. A. Ca(2+)-dependent and Ca(2+)-independent isozymes of protein kinase C mediate exocytosis in antigen-stimulated rat basophilic RBL-2H3 cells. Reconstitution of secretory responses with Ca2+ and purified isozymes in washed permeabilized cells. J Biol Chem. 1993 Jan 25;268(3):1749–1756. [PubMed] [Google Scholar]
- Ozawa K., Yamada K., Kazanietz M. G., Blumberg P. M., Beaven M. A. Different isozymes of protein kinase C mediate feedback inhibition of phospholipase C and stimulatory signals for exocytosis in rat RBL-2H3 cells. J Biol Chem. 1993 Feb 5;268(4):2280–2283. [PubMed] [Google Scholar]
- Pantaloni D., Carlier M. F. How profilin promotes actin filament assembly in the presence of thymosin beta 4. Cell. 1993 Dec 3;75(5):1007–1014. doi: 10.1016/0092-8674(93)90544-z. [DOI] [PubMed] [Google Scholar]
- Pfeiffer J. R., Seagrave J. C., Davis B. H., Deanin G. G., Oliver J. M. Membrane and cytoskeletal changes associated with IgE-mediated serotonin release from rat basophilic leukemia cells. J Cell Biol. 1985 Dec;101(6):2145–2155. doi: 10.1083/jcb.101.6.2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pike L. J. Phosphatidylinositol 4-kinases and the role of polyphosphoinositides in cellular regulation. Endocr Rev. 1992 Nov;13(4):692–706. doi: 10.1210/edrv-13-4-692. [DOI] [PubMed] [Google Scholar]
- Pike M. C., Costello K., Southwick F. S. Stimulation of human polymorphonuclear leukocyte phosphatidylinositol-4-phosphate kinase by concanavalin A and formyl-methionyl-leucyl-phenylalanine is calcium-independent. Correlation with maintenance of actin assembly. J Immunol. 1991 Oct 1;147(7):2270–2275. [PubMed] [Google Scholar]
- Pollard T. D., Cooper J. A. Actin and actin-binding proteins. A critical evaluation of mechanisms and functions. Annu Rev Biochem. 1986;55:987–1035. doi: 10.1146/annurev.bi.55.070186.005011. [DOI] [PubMed] [Google Scholar]
- Safer D. The interaction of actin with thymosin beta 4. J Muscle Res Cell Motil. 1992 Jun;13(3):269–271. doi: 10.1007/BF01766454. [DOI] [PubMed] [Google Scholar]
- Stossel T. P. From signal to pseudopod. How cells control cytoplasmic actin assembly. J Biol Chem. 1989 Nov 5;264(31):18261–18264. [PubMed] [Google Scholar]
- Theriot J. A., Mitchison T. J. The three faces of profilin. Cell. 1993 Dec 3;75(5):835–838. doi: 10.1016/0092-8674(93)90527-w. [DOI] [PubMed] [Google Scholar]
- Watts R. G., Howard T. H. Mechanisms for actin reorganization in chemotactic factor-activated polymorphonuclear leukocytes. Blood. 1993 May 15;81(10):2750–2757. [PubMed] [Google Scholar]
- Weatherbee J. A. Membranes and cell movement: interactions of membranes with the proteins of the cytoskeleton. Int Rev Cytol Suppl. 1981;12:113–176. doi: 10.1016/b978-0-12-364373-5.50014-7. [DOI] [PubMed] [Google Scholar]
- Yonezawa N., Nishida E., Iida K., Yahara I., Sakai H. Inhibition of the interactions of cofilin, destrin, and deoxyribonuclease I with actin by phosphoinositides. J Biol Chem. 1990 May 25;265(15):8382–8386. [PubMed] [Google Scholar]


