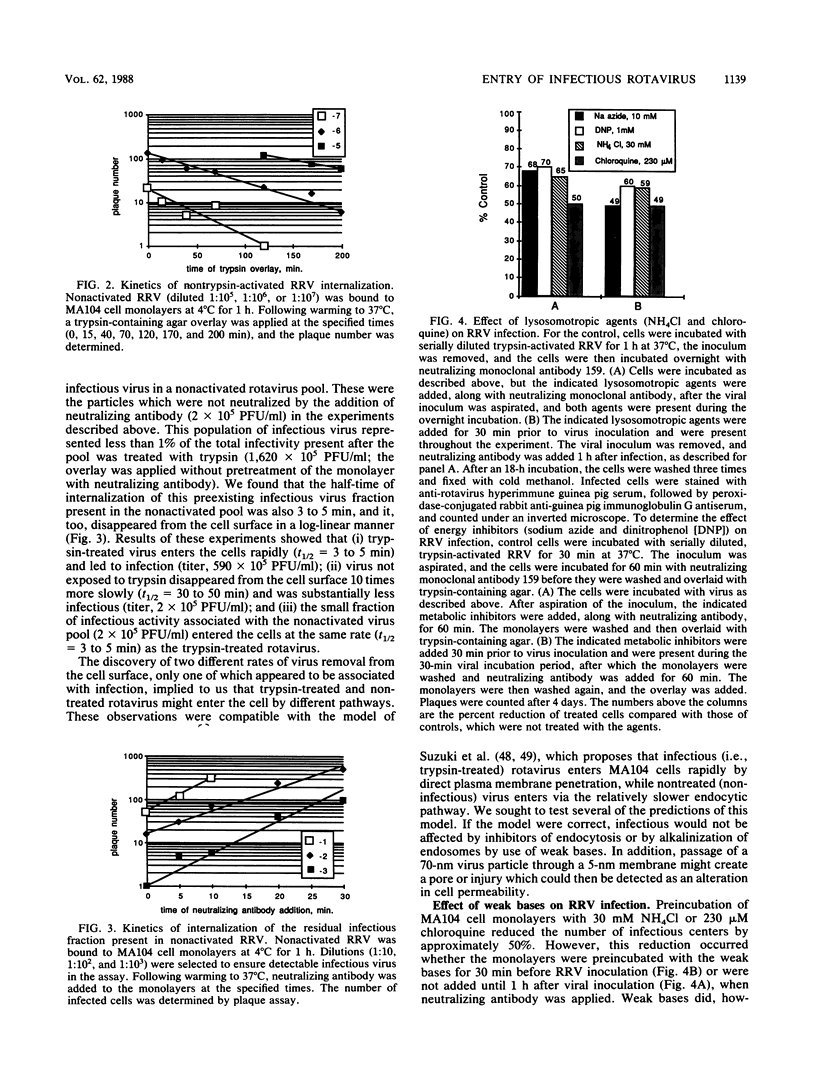
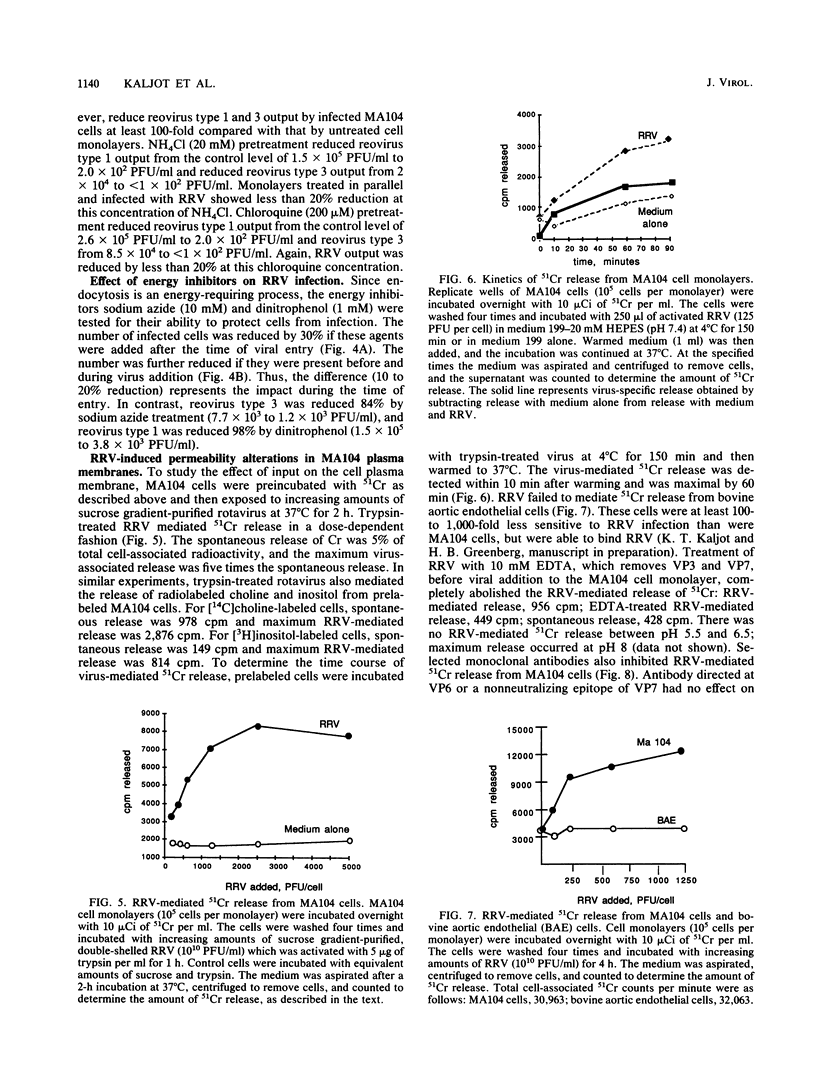
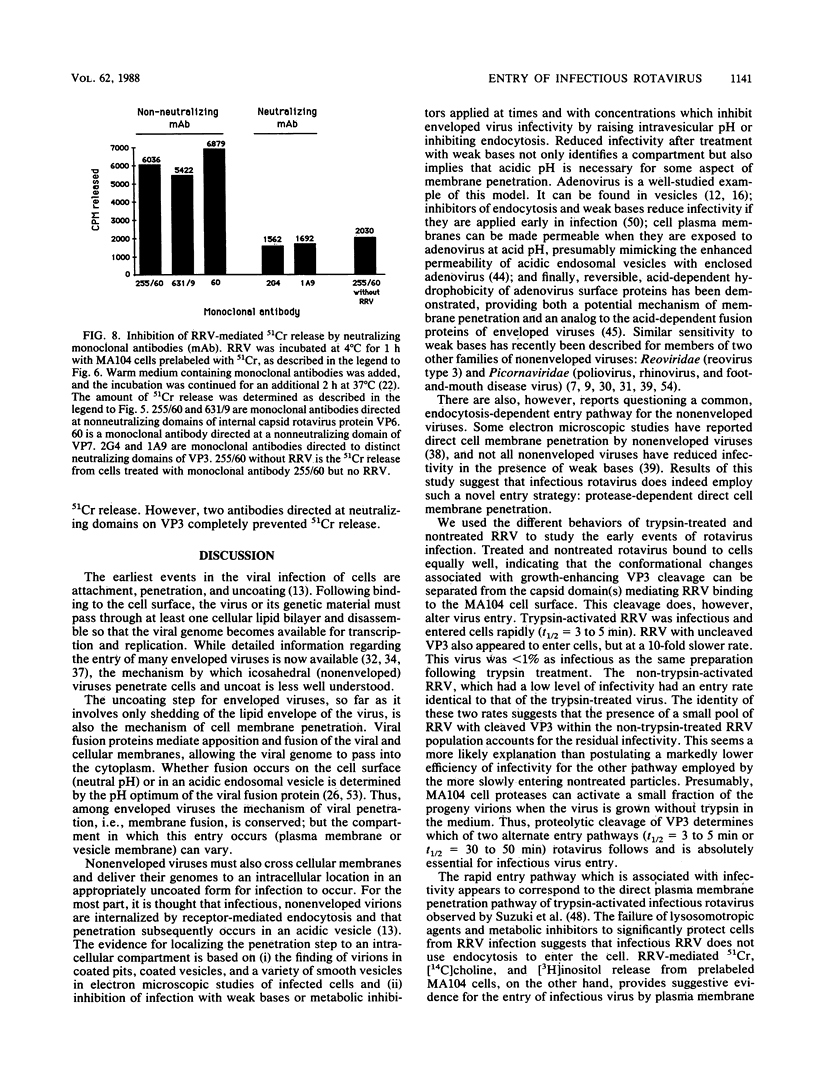
Abstract
Rotaviruses are icosahedral viruses with a segmented, double-stranded RNA genome. They are the major cause of severe infantile infectious diarrhea. Rotavirus growth in tissue culture is markedly enhanced by pretreatment of virus with trypsin. Trypsin activation is associated with cleavage of the viral hemagglutinin (viral protein 3 [VP3]; 88 kilodaltons) into two fragments (60 and 28 kilodaltons). The mechanism by which proteolytic cleavage leads to enhanced growth is unknown. Cleavage of VP3 does not alter viral binding to cell monolayers. In previous electron microscopic studies of infected cell cultures, it has been demonstrated that rotavirus particles enter cells by both endocytosis and direct cell membrane penetration. To determine whether trypsin treatment affected rotavirus internalization, we studied the kinetics of entry of infectious rhesus rotavirus (RRV) into MA104 cells. Trypsin-activated RRV was internalized with a half-time of 3 to 5 min, while nonactivated virus disappeared from the cell surface with a half-time of 30 to 50 min. In contrast to trypsin-activated RRV, loss of nonactivated RRV from the cell surface did not result in the appearance of infection, as measured by plaque formation. Endocytosis inhibitors (sodium azide, dinitrophenol) and lysosomotropic agents (ammonium chloride, chloroquine) had a limited effect on the entry of infectious virus into cells. Purified trypsin-activated RRV added to cell monolayers at pH 7.4 medicated 51Cr, [14C]choline, and [3H]inositol released from prelabeled MA104 cells. This release could be specifically blocked by neutralizing antibodies to VP3. These results suggest that MA104 cell infection follows the rapid entry of trypsin-activated RRV by direct cell membrane penetration. Cell membrane penetration of infectious RRV is initiated by trypsin cleavage of VP3. Neutralizing antibodies can inhibit this direct membrane penetration.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Almeida J. D., Hall T., Banatvala J. E., Totterdell B. M., Chrystie I. L. The effect of trypsin on the growth of rotavirus. J Gen Virol. 1978 Jul;40(1):213–218. doi: 10.1099/0022-1317-40-1-213. [DOI] [PubMed] [Google Scholar]
- Askaa J., Bloch B. Infection in piglets with a porcine rotavirus-like virus. Experimental inoculation and ultrastructural examination. Arch Virol. 1984;80(4):291–303. doi: 10.1007/BF01311220. [DOI] [PubMed] [Google Scholar]
- Babiuk L. A., Mohammed K., Spence L., Fauvel M., Petro R. Rotavirus isolation and cultivation in the presence of trypsin. J Clin Microbiol. 1977 Dec;6(6):610–617. doi: 10.1128/jcm.6.6.610-617.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banatvala J. E., Totterdell B., Chrystie I. L., Woode G. N. In-vitro detection of human rotaviruses. Lancet. 1975 Oct 25;2(7939):821–821. doi: 10.1016/s0140-6736(75)80057-2. [DOI] [PubMed] [Google Scholar]
- Barnett B. B., Spendlove R. S., Clark M. L. Effect of enzymes on rotavirus infectivity. J Clin Microbiol. 1979 Jul;10(1):111–113. doi: 10.1128/jcm.10.1.111-113.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsa J., Morash B. D., Sargent M. D., Copps T. P., Lievaart P. A., Szekely J. G. Two modes of entry of reovirus particles into L cells. J Gen Virol. 1979 Oct;45(1):161–170. doi: 10.1099/0022-1317-45-1-161. [DOI] [PubMed] [Google Scholar]
- Canning W. M., Fields B. N. Ammonium chloride prevents lytic growth of reovirus and helps to establish persistent infection in mouse L cells. Science. 1983 Feb 25;219(4587):987–988. doi: 10.1126/science.6297010. [DOI] [PubMed] [Google Scholar]
- Carrasco L. Modification of membrane permeability induced by animal viruses early in infection. Virology. 1981 Sep;113(2):623–629. doi: 10.1016/0042-6822(81)90190-2. [DOI] [PubMed] [Google Scholar]
- Carrillo E. C., Giachetti C., Campos R. H. Effect of lysosomotropic agents on the foot-and-mouth disease virus replication. Virology. 1984 Jun;135(2):542–545. doi: 10.1016/0042-6822(84)90208-3. [DOI] [PubMed] [Google Scholar]
- Chasey D., Banks J. Replication of atypical ovine rotavirus in small intestine and cell culture. J Gen Virol. 1986 Mar;67(Pt 3):567–576. doi: 10.1099/0022-1317-67-3-567. [DOI] [PubMed] [Google Scholar]
- Clark S. M., Roth J. R., Clark M. L., Barnett B. B., Spendlove R. S. Trypsin enhancement of rotavirus infectivity: mechanism of enhancement. J Virol. 1981 Sep;39(3):816–822. doi: 10.1128/jvi.39.3.816-822.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dales S. Early events in cell-animal virus interactions. Bacteriol Rev. 1973 Jun;37(2):103–135. doi: 10.1128/br.37.2.103-135.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dimmock N. J. Review article initial stages in infection with animal viruses. J Gen Virol. 1982 Mar;59(Pt 1):1–22. doi: 10.1099/0022-1317-59-1-1. [DOI] [PubMed] [Google Scholar]
- Espejo R. T., López S., Arias C. Structural polypeptides of simian rotavirus SA11 and the effect of trypsin. J Virol. 1981 Jan;37(1):156–160. doi: 10.1128/jvi.37.1.156-160.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estes M. K., Graham D. Y., Mason B. B. Proteolytic enhancement of rotavirus infectivity: molecular mechanisms. J Virol. 1981 Sep;39(3):879–888. doi: 10.1128/jvi.39.3.879-888.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FitzGerald D. J., Padmanabhan R., Pastan I., Willingham M. C. Adenovirus-induced release of epidermal growth factor and pseudomonas toxin into the cytosol of KB cells during receptor-mediated endocytosis. Cell. 1983 Feb;32(2):607–617. doi: 10.1016/0092-8674(83)90480-4. [DOI] [PubMed] [Google Scholar]
- Gollins S. W., Porterfield J. S. A new mechanism for the neutralization of enveloped viruses by antiviral antibody. Nature. 1986 May 15;321(6067):244–246. doi: 10.1038/321244a0. [DOI] [PubMed] [Google Scholar]
- Gorziglia M., Hoshino Y., Buckler-White A., Blumentals I., Glass R., Flores J., Kapikian A. Z., Chanock R. M. Conservation of amino acid sequence of VP8 and cleavage region of 84-kDa outer capsid protein among rotaviruses recovered from asymptomatic neonatal infection. Proc Natl Acad Sci U S A. 1986 Sep;83(18):7039–7043. doi: 10.1073/pnas.83.18.7039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham D. Y., Estes M. K. Proteolytic enhancement of rotavirus infectivity: biology mechanism. Virology. 1980 Mar;101(2):432–439. doi: 10.1016/0042-6822(80)90456-0. [DOI] [PubMed] [Google Scholar]
- Hsu M., Scheid A., Choppin P. W. Activation of the Sendai virus fusion protein (f) involves a conformational change with exposure of a new hydrophobic region. J Biol Chem. 1981 Apr 10;256(7):3557–3563. [PubMed] [Google Scholar]
- Joklik W. K. Studies on the effect of chymotrypsin on reovirions. Virology. 1972 Sep;49(3):700–715. doi: 10.1016/0042-6822(72)90527-2. [DOI] [PubMed] [Google Scholar]
- Kalica A. R., Flores J., Greenberg H. B. Identification of the rotaviral gene that codes for hemagglutination and protease-enhanced plaque formation. Virology. 1983 Feb;125(1):194–205. doi: 10.1016/0042-6822(83)90073-9. [DOI] [PubMed] [Google Scholar]
- Kantharidis P., Dyall-Smith M. L., Holmes I. H. Marked sequence variation between segment 4 genes of human RV-5 and simian SA 11 rotaviruses. Arch Virol. 1987;93(1-2):111–121. doi: 10.1007/BF01313897. [DOI] [PubMed] [Google Scholar]
- Leitman D. C., Fiscus R. R., Murad F. Forskolin, phosphodiesterase inhibitors, and cyclic AMP analogs inhibit proliferation of cultured bovine aortic endothelial cells. J Cell Physiol. 1986 May;127(2):237–243. doi: 10.1002/jcp.1041270208. [DOI] [PubMed] [Google Scholar]
- Lenard J., Miller D. K. Uncoating of enveloped viruses. Cell. 1982 Jan;28(1):5–6. doi: 10.1016/0092-8674(82)90368-3. [DOI] [PubMed] [Google Scholar]
- López S., Arias C. F., Bell J. R., Strauss J. H., Espejo R. T. Primary structure of the cleavage site associated with trypsin enhancement of rotavirus SA11 infectivity. Virology. 1985 Jul 15;144(1):11–19. doi: 10.1016/0042-6822(85)90300-9. [DOI] [PubMed] [Google Scholar]
- López S., Arias C. F., Méndez E., Espejo R. T. Conservation in rotaviruses of the protein region containing the two sites associated with trypsin enhancement of infectivity. Virology. 1986 Oct 15;154(1):224–227. doi: 10.1016/0042-6822(86)90445-9. [DOI] [PubMed] [Google Scholar]
- Madshus I. H., Olsnes S., Sandvig K. Different pH requirements for entry of the two picornaviruses, human rhinovirus 2 and murine encephalomyocarditis virus. Virology. 1984 Dec;139(2):346–357. doi: 10.1016/0042-6822(84)90380-5. [DOI] [PubMed] [Google Scholar]
- Madshus I. H., Olsnes S., Sandvig K. Mechanism of entry into the cytosol of poliovirus type 1: requirement for low pH. J Cell Biol. 1984 Apr;98(4):1194–1200. doi: 10.1083/jcb.98.4.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maratos-Flier E., Goodman M. J., Murray A. H., Kahn C. R. Ammonium inhibits processing and cytotoxicity of reovirus, a nonenveloped virus. J Clin Invest. 1986 Oct;78(4):1003–1007. doi: 10.1172/JCI112653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsh M., Helenius A. Adsorptive endocytosis of Semliki Forest virus. J Mol Biol. 1980 Sep 25;142(3):439–454. doi: 10.1016/0022-2836(80)90281-8. [DOI] [PubMed] [Google Scholar]
- Marsh M., Matlin K., Simons K., Reggio H., White J., Kartenbeck J., Helenius A. Are lysosomes a site of enveloped-virus penetration? Cold Spring Harb Symp Quant Biol. 1982;46(Pt 2):835–843. doi: 10.1101/sqb.1982.046.01.078. [DOI] [PubMed] [Google Scholar]
- Marsh M. The entry of enveloped viruses into cells by endocytosis. Biochem J. 1984 Feb 15;218(1):1–10. doi: 10.1042/bj2180001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason B. B., Graham D. Y., Estes M. K. Biochemical mapping of the simian rotavirus SA11 genome. J Virol. 1983 May;46(2):413–423. doi: 10.1128/jvi.46.2.413-423.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuno S., Inouye S., Kono R. Plaque assay of neonatal calf diarrhea virus and the neutralizing antibody in human sera. J Clin Microbiol. 1977 Jan;5(1):1–4. doi: 10.1128/jcm.5.1.1-4.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller D. K., Lenard J. Antihistaminics, local anesthetics, and other amines as antiviral agents. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3605–3609. doi: 10.1073/pnas.78.6.3605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan C., Rosenkranz H. S., Mednis B. Structure and development of viruses as observed in the electron microscope. V. Entry and uncoating of adenovirus. J Virol. 1969 Nov;4(5):777–796. doi: 10.1128/jvi.4.5.777-796.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrie B. L., Graham D. Y., Estes M. K. Identification of rotavirus particle types. Intervirology. 1981;16(1):20–28. doi: 10.1159/000149243. [DOI] [PubMed] [Google Scholar]
- Quan C. M., Doane F. W. Ultrastructural evidence for the cellular uptake of rotavirus by endocytosis. Intervirology. 1983;20(4):223–231. doi: 10.1159/000149395. [DOI] [PubMed] [Google Scholar]
- Richardson C. D., Scheid A., Choppin P. W. Specific inhibition of paramyxovirus and myxovirus replication by oligopeptides with amino acid sequences similar to those at the N-termini of the F1 or HA2 viral polypeptides. Virology. 1980 Aug;105(1):205–222. doi: 10.1016/0042-6822(80)90168-3. [DOI] [PubMed] [Google Scholar]
- Rubin D. H., Kornstein M. J., Anderson A. O. Reovirus serotype 1 intestinal infection: a novel replicative cycle with ileal disease. J Virol. 1985 Feb;53(2):391–398. doi: 10.1128/jvi.53.2.391-398.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seth P., Pastan I., Willingham M. C. Adenovirus-dependent increase in cell membrane permeability. J Biol Chem. 1985 Aug 15;260(17):9598–9602. [PubMed] [Google Scholar]
- Seth P., Willingham M. C., Pastan I. Adenovirus-dependent release of 51Cr from KB cells at an acidic pH. J Biol Chem. 1984 Dec 10;259(23):14350–14353. [PubMed] [Google Scholar]
- Seth P., Willingham M. C., Pastan I. Binding of adenovirus and its external proteins to Triton X-114. Dependence on pH. J Biol Chem. 1985 Nov 25;260(27):14431–14434. [PubMed] [Google Scholar]
- Shaw R. D., Vo P. T., Offit P. A., Coulson B. S., Greenberg H. B. Antigenic mapping of the surface proteins of rhesus rotavirus. Virology. 1986 Dec;155(2):434–451. doi: 10.1016/0042-6822(86)90205-9. [DOI] [PubMed] [Google Scholar]
- Suzuki H., Kitaoka S., Konno T., Sato T., Ishida N. Two modes of human rotavirus entry into MA 104 cells. Arch Virol. 1985;85(1-2):25–34. doi: 10.1007/BF01317003. [DOI] [PubMed] [Google Scholar]
- Suzuki H., Kitaoka S., Sato T., Konno T., Iwasaki Y., Numazaki Y., Ishida N. Further investigation on the mode of entry of human rotavirus into cells. Arch Virol. 1986;91(1-2):135–144. doi: 10.1007/BF01316734. [DOI] [PubMed] [Google Scholar]
- Svensson U. Role of vesicles during adenovirus 2 internalization into HeLa cells. J Virol. 1985 Aug;55(2):442–449. doi: 10.1128/jvi.55.2.442-449.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theil K. W., Bohl E. H., Agnes A. G. Cell culture propagation of porcine rotavirus (reovirus-like agent). Am J Vet Res. 1977 Nov;38(11):1765–1768. [PubMed] [Google Scholar]
- Vonderfecht S. L., Huber A. C., Eiden J., Mader L. C., Yolken R. H. Infectious diarrhea of infant rats produced by a rotavirus-like agent. J Virol. 1984 Oct;52(1):94–98. doi: 10.1128/jvi.52.1.94-98.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White J., Kielian M., Helenius A. Membrane fusion proteins of enveloped animal viruses. Q Rev Biophys. 1983 May;16(2):151–195. doi: 10.1017/s0033583500005072. [DOI] [PubMed] [Google Scholar]
- Zeichhardt H., Wetz K., Willingmann P., Habermehl K. O. Entry of poliovirus type 1 and Mouse Elberfeld (ME) virus into HEp-2 cells: receptor-mediated endocytosis and endosomal or lysosomal uncoating. J Gen Virol. 1985 Mar;66(Pt 3):483–492. doi: 10.1099/0022-1317-66-3-483. [DOI] [PubMed] [Google Scholar]


