Abstract
Granulomatous experimental autoimmune thyroiditis (G-EAT) is induced by mouse thyroglobulin-sensitized splenocytes activated in vitro with mouse thyroglobulin and interleukin-12. In wild-type (WT) DBA/1 recipients of WT donor splenocytes, thyroid lesions reach maximal severity at day 20, with ongoing inflammation and extensive fibrosis at day 60. Our previous studies indicated the site of expression of FLIP and Fas ligand [thyroid epithelial cells (TECs) versus inflammatory cells] differed in mice when lesions would resolve or progress to fibrosis. To test the hypothesis that expression of FLIP by TECs would promote earlier G-EAT resolution in DBA/1 mice, transgenic (Tg) DBA/1 mice expressing FLIP on TECs were generated. In FLIP Tg+ and Tg− littermate recipients of WT donor splenocytes, G-EAT severity was comparable at day 20, but fibrosis was decreased, and many lesions resolved by day 60 in Tg+ but not Tg− recipients. FLIP and Fas ligand were primarily expressed by TECs in Tg+ recipients and by inflammatory cells in Tg− recipients at day 60. Apoptosis of inflammatory cells was greater, and expression of proinflammatory cytokines was decreased in thyroids of Tg+ compared with Tg− recipients. These results are consistent with the hypothesis that transgenic expression of FLIP on thyroid epithelial cells promotes earlier resolution of granulomatous experimental autoimmune thyroiditis.
Experimental autoimmune thyroiditis (EAT), the conventional model to study thyroid autoimmunity in mice, is an organ-specific autoimmune disease that can be induced in genetically susceptible strains of mice by injection of mouse thyroglobulin (MTg) and adjuvant1 or by transfer of MTg-primed donor spleen cells activated with MTg in vitro.1,2,3 A more severe and histologically distinct granulomatous EAT (G-EAT) is induced when MTg-sensitized donor spleen cells are activated in vitro with MTg and interleukin (IL)-12.4,5 Thyroid lesions in G-EAT are characterized by thyroid epithelial cell (TEC) proliferation, large numbers of histiocytes, multinucleated giant cells, and variable numbers of neutrophils in addition to T lymphocytes.4,5 In wild-type (WT) DBA/1 recipients of WT donor splenocytes, thyroid lesions reached maximal severity at day 20, with ongoing inflammation and development of fibrosis by day 60.5 CD4+ T cells are the primary effector cells for G-EAT.4 Depletion of CD8+ T cells has no effect on G-EAT development but inhibits G-EAT resolution, indicating that resolution of G-EAT requires CD8+ T cells.6,7
By formation of a death-inducing signaling complex and initiation of a signaling cascade of caspases, Fas ligand (FasL) induces apoptosis of Fas-expressing cells.8 Apoptosis through the Fas/FasL pathway plays a critical role in termination of immune responses and maintenance of peripheral lymphocyte homeostasis.8,9 It also plays an important role in many human and murine autoimmune diseases, such as Hashimoto’s thyroiditis and Grave’s disease,10,11,12 multiple sclerosis,13 insulin-dependent diabetes,14 experimental allergic encephalomyelitis (EAE),15,16 type 1 diabetes in non-obese diabetic mice,17,18,19,20 and G-EAT.10,21,22,23,24 The Fas/FasL pathway can function to induce autoimmune damage17,18,19,20 and also to shut down autoimmune responses.15,16,23,25 Our previous studies showed that resolution of G-EAT involves apoptosis of CD4+ effector cells through the Fas/FasL pathway.23,25,26 Recovery from EAE is also associated with apoptotic elimination of effector T cells through the Fas/FasL pathway.15,16,27
Following the characterization of viral FLIP (vFLIP), the mammalian cellular homolog was identified and called FLICE-inhibitory protein (cFLIP).28,29,30,31 cFLIP inhibits Fas-mediated apoptosis through its ability to block activation of caspase-8.8,28 FLIP is therefore an important inhibitor of the initial upstream steps of Fas-mediated apoptosis.8 cFLIP expression was shown to be increased in lymphocytes of patients with active multiple sclerosis,28 in Graves’ disease32 and in T cells in animal models of inflammatory arthritis28,32 and in G-EAT.24
Recently, cFLIPL was found to divert Fas signals toward pathways leading to cell growth and differentiation.33 This suggests that cFLIP function is complex, and more studies are required to elucidate its role in regulating autoimmune inflammatory responses. We recently suggested that chronic inflammation in G-EAT was associated with predominant expression of FLIP by thyroid-infiltrating inflammatory cells, whereas increased FLIP expression by TECs may protect TECs from apoptosis and promote G-EAT resolution.24 To test directly the hypothesis that G-EAT resolution would be promoted if FLIP was primarily expressed by TECs, transgenic mice expressing FLIP on TECs were generated. Mice expressing cFLIPL on TECs had earlier resolution of G-EAT compared with nontransgenic littermates, supporting the hypothesis that FLIP expression on thyroid epithelial cells contributes to G-EAT resolution.
Materials and Methods
Generation of cFLIPL Transgenic Mice
The rat thyroglobulin (RTg) promoter on pTgCAT7 was provided by Dr. Motoyasu Saji (Ohio State University, Columbus, OH), and the 1.5-kb FLAG-tagged murine cFLIPL on pBSKII (2.96 kb) was provided by Dr. Susanne Lens (Netherlands Cancer Institute, Amsterdam, The Netherlands). The 900-bp fragment of RTg promoter was obtained using HindIII and EcoRI (Invitrogen, Carlsbad, CA) and blunted. The pBSKII containing murine cFLIPL was linearized by NotI (Invitrogen), blunted, and then ligated with the RTg promoter using T4 DNA ligase. The correct orientation of the promoter in the recombined plasmid was confirmed by sequence determination using T7 primer (DNA Core Facility, University of Missouri, Columbia, MO). The recombined construct (3.5 kb) containing 1.5-kb FLAG-tagged mouse cFLIPL cDNA, β-globin polyA (500 bp), β-globin intron (600 bp), and RTg promoter (900 bp) was amplified in Escherichia coli and transfected into the thyroid PCCL3 cell line (provided by Dr. Daniel Altschuler, University of Pittsburgh, Pittsburgh, PA) using Fugene6 (Invitrogen), and FLIP protein expression was confirmed by immunohistochemical staining. The recombinant plasmid was digested using BssHII (Invitrogen), and the 3.5-kb fragment containing FLAG-tagged mouse cFLIPL cDNA, β-globin polyA, β-globin intron, and RTg promoter was microinjected into fertilized oocytes from FVB female mice (Transgenic Core Facility, University of Missouri). Transgenic founders were screened by polymerase chain reaction (PCR) of tail DNA using the following primers specific to RTg promoter and cFLIPL recombined construct: sense, 5′-CTGGTCATCATCCTGCCTTTCTC-3′; and antisense, 5′-TGCAGCCAGGTTCTCAGTCAC-3′. Dot blot of tail DNA was also used to confirm expression of the transgene in some mice. Two FVB transgenic founders (one male and one female) were obtained. Because FVB mice are less EAT susceptible than DBA/1 mice, even though both strains express the same H-2q MHC haplotype (our unpublished results), the transgenic female FVB founder was crossed with an EAT-susceptible DBA/1 male, and Tg+ F1 offspring were selected by PCR analysis of tail DNA. Tg+ F1 mice were backcrossed to DBA/1 mice. After backcrossing three times to DBA/1 mice and selecting for expression of the transgene and the DBA/1 coat color at each generation, Tg+ and Tg− littermates used as recipients of activated DBA/1 splenocytes (see below) developed maximal G-EAT lesions 20 days after cell transfer that were indistinguishable histologically and in severity from those developing in WT DBA/1 recipients of the same cells. FLIP Tg+ and Tg− mice from the third backcross generation also transferred severe G-EAT to DBA/1 recipients when immunized and used as donors for adoptive transfer of G-EAT (our unpublished results). Mice used for these experiments were from the third and fourth backcross generations, and identical results were obtained with both backcross groups. In all experiments, Tg+ and Tg− littermates were used as recipients of sensitized donor cells from WT DBA/1 mice. Immunohistochemistry (IHC) using rabbit anti-FLIP polyclonal antibody (Ab) (Abcam, Cambridge, MA) and anti-FLAG polyclonal Ab (Abcam) and Western blot using anti-FLIP monoclonal antibody (mAb) (Alexis, San Diego, CA) and anti-FLAG were used to confirm expression of the transgene on TECs but not on other cell types.
Induction of G-EAT
WT DBA/1 mice generated in our breeding colony were used as donors for all experiments. DBA/1 donors were injected intravenously twice at 10-day intervals with 150 μg of MTg prepared as previously described2 and 15 μg of lipopolysaccharide LPS (Sigma, St. Louis, MO). Seven days later, donor spleen cells were activated in vitro with 25 μg/ml MTg and 5 ng/ml IL-12 (Peprotech, Rocky Hill, NJ) as previously described.5 Cells were harvested after 72 hours and washed twice, and 3 × 107 cells were transferred intravenously to 500-Rad irradiated FLIP Tg+ or Tg− littermate recipients. Recipient thyroids were evaluated 20 (peak of disease), 40, or 60 days after cell transfer.5,23 Both male and female mice were used for these experiments. Donor DBA/1 mice were generally 8 to 10 weeks old at the time of immunization, and recipients were 7 to 9 weeks old. All mice were bred and maintained in accordance with University of Missouri institutional guidelines for animal care.
Evaluation of G-EAT Histopathology and Fibrosis
Thyroids were removed from groups of six to eight FLIP Tg+ or Tg− recipients 20, 40, or 60 days after cell transfer, and one lobe of each thyroid was fixed in formalin. Formalin-fixed paraffin-embedded tissues were sectioned and stained with hematoxylin and eosin. Thyroids were scored quantitatively for G-EAT severity using a scale of 1+ to 5+ according to previously established criteria.4,5 Thyroiditis of 1+ is defined as an infiltrate of at least 125 cells in one or several foci, 2+ is 10 to 20 foci of cellular infiltration involving up to 25% of the gland, 3+ indicates that 25 to 50% of the gland is infiltrated, 4+ indicates that >50% of the gland is destroyed, and 5+ indicates virtually complete destruction of the gland, with few or no remaining follicles. At day 20, thyroids with 5+ severity scores are greatly enlarged (10- to 20-fold), with granuloma formation, large numbers of histiocytes, lymphocytes, and neutrophils, microabscess formation, necrosis, and fibrosis.5 At days 40 to 60, fibrosis is extensive, and thyroids are very small and atrophic (approximately one-fifth to one-tenth the size of a normal thyroid). For evaluation of collagen deposition (fibrosis), some thyroid sections from FLIP Tg− and Tg+ recipients were stained using Masson’s Trichrome.34 All slides were evaluated by at least two investigators, one of whom had no knowledge of the experimental treatments. Differences in interpretation were very rare.
Serum Thyroxine (T4) Assay
Serum T4 levels were determined using a T4 enzyme immunoassay kit (Biotecx Labs, Houston, TX) according to the manufacturer’s instructions. Results are expressed as micrograms of T4 per decaliter of serum. Using this assay, T4 values for normal mouse serum ranged from 4 to 10 μg/dl; values <3 μg/dl are considered low.35
IHC
IHC staining for FLIP was described previously.23 The same protocol was used for FLAG staining. IHC staining for CD4 and CD8 was described previously.24,35 Briefly, cryostat sections were fixed in acetone for 10 minutes at 4°C. After treatment for 30 minutes with 0.1% saponin in 1% bovine serum albumin (FLIP and FLAG) or 1% bovine serum albumin alone (CD4 and CD8), slides were incubated with polyclonal anti-FLIP or anti-FLAG, rat anti-CD4 (GK1.5; American Type Culture Collection, Manassas, VA) and rat anti-CD8 mAb (53.6; American Type Culture Collection) for 60 minutes at room temperature. After incubation with a secondary biotinylated Ab (Jackson ImmunoResearch Laboratories Inc., West Grove, PA), immunoreactivity was demonstrated using the avidin-biotin complex immunoperoxidase system (Vector, Burlingame, CA) and developed using NovaRED (Vector) as the chromogen. Slides were counterstained with hematoxylin. As a negative control, primary Ab was replaced with an equal amount of normal rabbit, goat, or rat IgG; these controls were always negative. To quantify the number of CD4+ or CD8+ cells, all CD4+ or CD8+ cells in five to six randomly selected high-power fields (magnification, ×400) were manually counted, and the average number of cells per field for three individual animals per group were determined (Figure 3G).
Figure 3.
Collagen deposition (fibrosis) and CD4+ and CD8+ T cells in thyroids of FLIP Tg+ and Tg− littermate recipients with G-EAT. A and B: Masson’s Trichrome staining for collagen (blue) in thyroids of FLIP Tg+ (right) and Tg− recipients (left) at day 60. There was minimal deposition of collagen (blue, arrows) in thyroids of FLIP Tg+ recipients compared with Tg− littermates. C and D: CD4+ T cells (red, arrows) in thyroids of FLIP Tg+ (right) and Tg− recipients (left) at day 60. E and F: CD8+ T cells (red, arrows) in thyroids of FLIP Tg+ (right) and Tg− recipients (left) at day 60. Results are representative areas on slides from at least three individual mice examined in each group. G: CD4+ and CD8+ T cells in five to six randomly selected high-power fields of three individual mice/group (magnification, ×400) were manually counted. CD4+ T cells outnumbered CD8+ T cells in FLIP Tg− recipients with ongoing inflammation (4 to 5+ severity scores) and fibrosis at day 60 (G, left), whereas CD8+ T cells outnumbered CD4+ T cells in FLIP Tg+ recipients when lesions were beginning to resolve (4+ severity scores) (G, right). A significant difference is indicated by the asterisk. Magnification, ×100 (in A, C, and E); ×400 (B, D, and F).
Confocal Laser Scanning Double-Immunofluorescence Microscopy
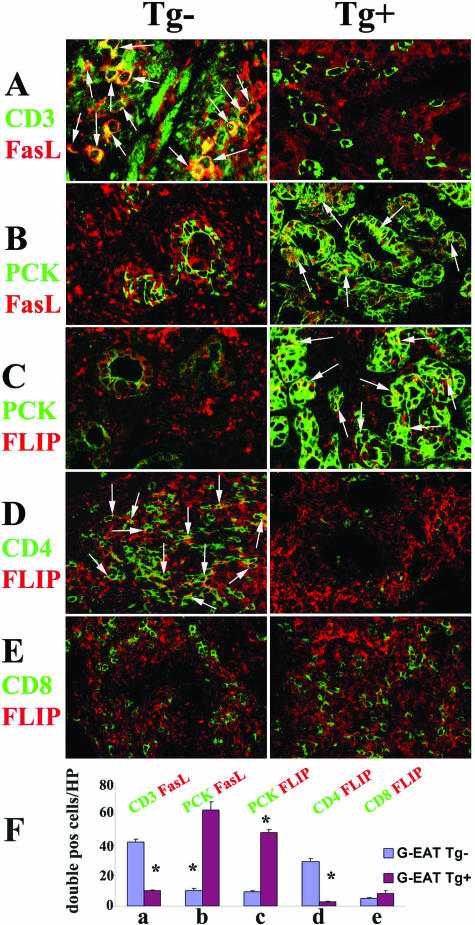
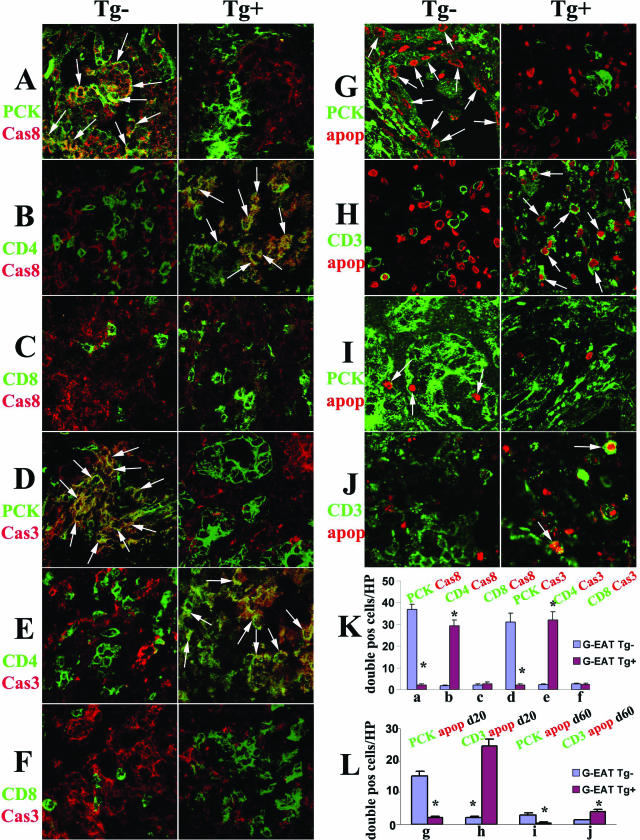
To detect differential expression of FLIP, FasL, active caspase-8, and active caspase-3 by CD4+ and CD8+ T cells or TECs, dual-color immunofluorescence and confocal laser scanning microscopy was performed using the same protocol as previously described23,24,34,35,36 using rat anti-CD4 and -CD8 mAb and polyclonal anti-FLIP (described above), anti-FasL mAb (Santa Cruz Biotechnology, Santa Cruz, CA), anti-active caspase-8 (IMGENEX, San Diego, CA), anti-active caspase-3 (BD Pharmingen, San Diego, CA), and anti-CD3 (DakoCytomation, Carpinteria, CA) for T cells or pan-cytokeratin-26 (PCK; Sigma) for TECs. Frozen sections of thyroids were used except for dual staining of FasL and CD3 or PCK (formalin-fixed paraffin sections were used). FLIP, FasL, active caspase-8, and active caspase-3 were visualized with Alexa 568 (red; Molecular Probes, Eugene, OR). Cytokeratin (TECs), CD3, CD4, and CD8 molecules were visualized by Alexa 488 (green; Molecular Probes). Apoptosis was detected using an in situ cell death kit (red; Roche, Nutley, NJ) with formalin-fixed paraffin sections of thyroids as previously described.36 Slides were observed with a Bio-Rad Radiance 2000 confocal system (Bio-Rad, Hercules, CA) coupled to an Olympus IX70 inverted microscope (Olympus, Tokyo, Japan). To quantify the number of double positive cells, all double positive cells in four to five randomly selected high-power fields (magnification, ×600) were manually counted, and the average number of cells per field for three individual animals per group was determined (Figure 4F and Figure 5, K and L).
Figure 4.
FLIP and FasL distribution in thyroids of FLIP Tg+ and Tg− recipients at day 60. A–C: CD4+ and CD8+ T cells were identified by CD3 (A, green) and TECs by PCK (B and C, green) on paraffin sections of thyroids. Although TECs and inflammatory cells both expressed FasL (A and B, red) in FLIP Tg− (left) and Tg+ recipients (right), FasL was mainly expressed by CD3+ inflammatory cells in FLIP Tg− recipients (A, left, yellow, overlay, arrows) and by PCK + TECs in FLIP Tg+ recipients (B, right, yellow, overlay, arrows). TECs and inflammatory cells both expressed FLIP (C, red) in FLIP Tg− (left) and Tg+ recipients (right); FLIP was mainly expressed by PKC + TECs in FLIP Tg+ recipients (C, right, yellow, overlay, arrows). D and E: Confocal staining of CD4 (D, green) or CD8 (E, green) on frozen sections with FLIP (D and E, red) showed that FLIP was mainly expressed by CD4+ T cells in FLIP Tg− recipients (D, left, yellow, overlay, arrows), whereas very few CD4+ T cells in Tg+ recipients expressed FLIP (D, right, yellow, overlay). F: Double positive cells (yellow, overlay, in A–E) in four to five randomly selected high-power fields of three individual mice/group (magnification, ×600) were manually counted and summarized in F (bars a–e correspond to panels A–E). A significant difference between G-EAT FLIP Tg− recipients and Tg+ recipients is indicated by the asterisk (P < 0.05). Magnification: A, ×800, B–E, ×600. Results are representative areas on slides from at least three individual mice examined per group. Mice in both groups had comparable G-EAT severity scores (4 to 5+).
Figure 5.
Evaluation of active caspase-8, active capase-3, and apoptosis in thyroids of FLIP Tg+ and Tg− recipients at day 20. A–F: Confocal staining of TECs, CD4, or CD8 on frozen sections with active caspase-8 or active caspase-3. CD4+ and CD8+ T cells (B, C, E, and F, green) and PCK + TECs (A and D, green) both expressed active caspase-8 (A–C, red) in FLIP Tg− (left) and Tg+ recipients (right), and active caspase-8 was mainly expressed by TECs in FLIP Tg− recipients (A, left, yellow, overlay, arrows) and by CD4+ T cells in FLIP Tg+ recipients (B, right, yellow, overlay, arrows). Active caspase-3 was primarily expressed by TECs in FLIP Tg− recipients (D, left, yellow, overlay, arrows) and by CD4+ T cells in FLIP Tg+ recipients at day 20 (E, right, yellow, overlay, arrows). G–J: Confocal staining of TECs and CD3+ T cells (green) on paraffin sections with apoptosis (red nuclear staining). There were many apoptotic cells in thyroids of both FLIP Tg+ and Tg− littermates with 4 to 5+ G-EAT severity scores at day 20 (red, G and H) and many fewer apoptotic cells in thyroids of both groups at day 60 (red, I and J). Apoptosis (red) was mainly in TECs in FLIP Tg− recipients both at days 20 (G, left, arrows) and 60 (I, left, arrows) and mainly in inflammatory cells in FLIP Tg+ recipients both at days 20 (H, right, arrows) and 60 (J, right, arrows). K and L: Double positive cells (yellow, overlay, in A–F; or red circled by green in G–J) in four to five randomly selected high-power fields of three individual mice/group (magnification, ×800) were manually counted and summarized in K and L (bars a–j correspond to panels A–J). A significant difference between G-EAT FLIP Tg− recipients and Tg+ recipients is indicated by the asterisk (P < 0.05). Magnification, ×800 (A–J). Shown are representative areas on slides of thyroids of at least three individual mice per group.
Reverse Transcriptase (RT)-PCR
Thyroid lobes were removed from individual mice at different times after adoptive transfer, and one lobe was stored at −80°C before processing. Frozen thyroids were homogenized in TRIzol (Invitrogen), and RNA was extracted and reverse transcribed as previously described in detail.37,38 Diluted cDNA was amplified using the following conditions: 94°C for 30 seconds for denaturing, 60°C for 30 seconds for annealing, and 72°C for 1 minute for extension. To determine the relative initial amounts of target cDNA, each cDNA sample was serially diluted one-fifth and one-twenty-fifth and amplified with specific primers.37,38 Hypoxanthine phosphoribosyltransferase was used as a housekeeping gene to verify that the same amount of RNA was amplified. PCR products were electrophoresed in 2% agarose gel, visualized by UV light after staining with ethidium bromide, and normalized between samples relative to levels of hypoxanthine phosphoribosyltransferase using an IS-1000 Digital Imaging System (Life Sciences, St. Louis, MO). Most gene primers used in this study have been described previously.34,37,38 IL-17 primers were: sense, 5′-GGTCAACCTCAAAGTCTTTAACTC-3′, and antisense, 5′-TTAAAAATGCAAGTAAGTTTGCTG-3′. Foxp3 primers were: sense, 5′-GGCCCTTCTCCAGGACAGA-3′, and antisense, 5′-GCTGATCATGGCTGGGTTGT-3′.
Western Blot
Thyroids were homogenized in lysis buffer (50 mmol/L HEPES, 250 mmol/L NaCl, 5 mmol/L ethylenediamine tetraacetic acid, and 0.1% Triton). After centrifugation at 15,000 rpm for 15 minutes at 4°C, supernatants were collected and electrophoresed by adding 30 μg of protein to a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel. After transfer onto nitrocellulose membranes, they were incubated overnight at 4°C in Tris-buffered saline containing 5% nonfat dry milk and 0.05% Tween 20 and probed 2 hours at room temperature with rat anti-FLIP mAb or rabbit anti-FLAG polyclonal Ab (Sigma). After washing, membranes were incubated with a peroxidase-conjugated goat anti-rat or goat anti-rabbit IgG (1/5000) for 1 hour at room temperature, and the reaction was detected with a chemiluminescence detection kit (Pierce, Rockford, IL).24 For normalization of signals, the membranes were stripped by incubating at 60°C for 30 minutes in stripping buffer (62.5 mmol/L Tris-HCl, 100 mmol/L 2-ME, and 2% sodium dodecyl sulfate), washed in Tris-buffered saline/Tween 20, and reprobed with 1/2000 rabbit anti-actin primary Ab (Santa Cruz Biotechnology) and 1/2000 horseradish peroxidase-conjugated anti-rabbit IgG as secondary Ab. Bands were scanned and quantitated using Quality One-4.41 software. Each lane in the figures represents protein extracted from individual thyroids of normal FLIP transgenic mice or Tg− littermate controls (Figure 1D) or G-EAT Tg− and G-EAT Tg+ recipients (Figure 6B).
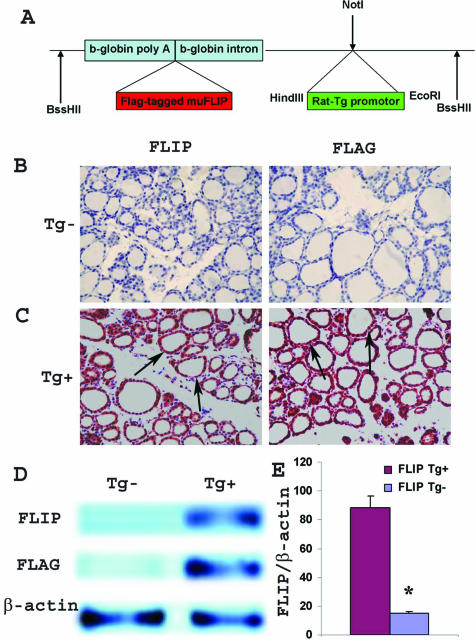
Figure 1.
Generation of cFLIPL transgenic mice and expression of FLIP transgene. A: Map of the cFLIP transgene. FLAG-tagged mouse cFLIPL (1.5 kb) was cloned into a targeting vector (pBSKII), driven by the 900-bp RTg promoter. The 3.5-kb fragment was microinjected into fertilized oocytes of FVB female mice. B and C: IHC of FLIP (left) and FLAG (right) in thyroids of normal DBA/1 FLIP Tg− and Tg+ littermates. Both FLIP and FLAG were highly expressed in thyroids of naïve Tg+ mice (arrows) but were not detected in thyroids of naïve Tg− mice (magnification, ×400). D: Protein expression of FLIP and FLAG in thyroids of FLIP Tg+ and Tg− littermates. Thirty micrograms of protein was loaded in each lane. Results are representative of three experiments. E: Dot blot analysis of tail DNA from naïve FLIP Tg+ and Tg− littermates. Twenty micrograms of DNA was loaded, and 1.5-kb murine FLIP Klenow was used as probe template. β-Actin was used for data normalization. FLIP transgene level in Tg+ littermates was nearly six times higher than in Tg− littermates. Bars are means of data for thyroids of Tg− littermates (four to six individuals) and Tg+ littermates (16 individuals) ± SEM. Results are expressed as the mean ratio of densitometric U/β-actin ± SEM (×100) and are representative of three independent experiments. A significant difference is indicated by the asterisk.
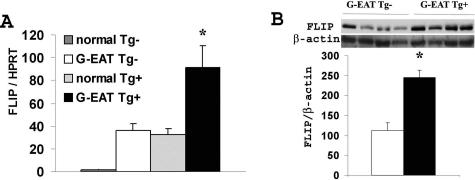
Figure 6.
FLIP protein and mRNA expression in thyroids of FLIP Tg+ and Tg− recipients at day 20. A: FLIP mRNA was undetectable in thyroids of normal unimmunized Tg− mice but was significantly up-regulated (P < 0.05) in thyroids of FLIP Tg− recipients with severe (4 to 5+) G-EAT at day 20. FLIP mRNA expression in thyroids of normal FLIP Tg+ mice was similar to that in thyroids of FLIP Tg− recipients with severe G-EAT and was further up-regulated (P < 0.05) in thyroids of FLIP Tg+ recipients with 4 to 5+ G-EAT at day 20. Results are expressed as the mean ratio of FLIP densitometric U/hypoxanthine phosphoribosyltransferase ± SEM (×100) of five to six individual mice per group and are representative of three independent experiments. B: FLIP protein was significantly higher (P < 0.05) in thyroids of FLIP Tg+ recipients compared with Tg− recipients with 4 to 5+ G-EAT at day 20. Thirty micrograms of protein from thyroids of the FLIP Tg+ (four individuals) and Tg− recipients (four individuals) was loaded in each lane, and results for FLIP and β-actin are shown at the top in B. Results are expressed as the mean ratio of densitometric U/β-actin ± SEM (×100) and are representative of three independent experiments. A significant difference between G-EAT FLIP Tg− recipients and Tg+ recipients is indicated by the asterisk (P < 0.05).
Dot Blot Analysis
Dot blot analysis was used to assess the level of FLIP transgene in FLIP Tg+ mice. After denaturation, 20 μg of tail DNA was immobilized onto nitrocellulose membranes. FLAG-tagged murine FLIP Klenow (1.5 kb) was used as the template, [α-32P]dCTP-labeled DNA probes were generated using strip-EZ probe synthesis and removal kits (Ambion, Austin, TX). Prehybridization was performed for 2 to 4 hours, hybridization was performed overnight at 42°C, and washes were done according to instructions. Blots were exposed either 8 hours or 2 days at −80°C to blue X-ray films. For normalization of signals, the membranes were stripped by incubating at 90°C for 30 minutes in stripping buffer, washed, and reprobed with a β-actin probe.
Statistical Analysis
All experiments were repeated at least three times. Statistical analysis of data was performed using an unpaired two-tailed Student’s t-test or/and the Mann-Whitney rank sum test. A value of P < 0.05 was considered significant.
Results
Generation of Transgenic Mice Expressing Murine cFLIPL on TECs
To test the hypothesis that overexpression of FLIP by TECs would promote earlier resolution of G-EAT, we generated transgenic mice expressing mouse cFLIPL under control of the RTg promoter (Figure 1A). The FVB transgenic founder female was crossed with an MHC-compatible, EAT-susceptible DBA/1 male, and the Tg+ offspring were backcrossed to DBA/1 as described in Materials and Methods. Both IHC (Figure 1, B and C) and Western blot (Figure 1D) showed that neither FLIP nor FLAG-tagged protein was detected in thyroids of naïve Tg− mice. However, FLIP and FLAG were both highly expressed by TECs in the founder and all Tg+ mice at each backcross generation (Figure 1, B–D; data not shown) but not in other tissues such as spleen and cervical lymph nodes (data not shown). Some mice were typed for FLIP transgene expression by both PCR analysis of tail DNA and dot blot analysis, and results using both assays were consistent. Dot blot indicated that the FLIP transgene level in normal FLIP Tg+ mice was nearly six times higher than in Tg− littermates (Figure 1E).
Distinct Outcomes of G-EAT in FLIP Tg+ and FLIP Tg− DBA/1 Recipients
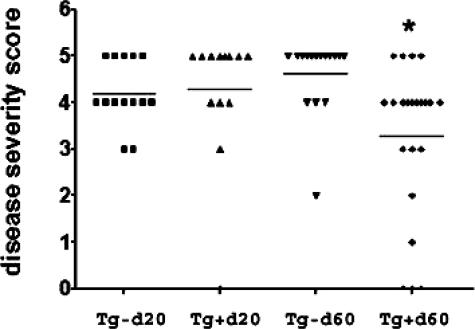
G-EAT was induced by adoptive transfer of spleen cells from MTg-sensitized DBA/1 donors activated with MTg and IL-12 in vitro. G-EAT lesions in both Tg+ and Tg− recipients of DBA/1 spleen cells reached maximal severity 20 days after cell transfer, and disease severity scores were similar (4 to 5+) in both FLIP Tg+ and Tg− recipients (Figure 2). At day 40, severity scores of 4 to 5+ were maintained in both groups (data not shown), although thyroids of most Tg+ recipients were larger (less atrophic) and had less fibrosis (Table 1; data not shown). By day 60, thyroid lesions in all Tg− recipients were fibrotic, with an average severity score of 4.5, whereas thyroid lesions in all but four of the Tg+ recipients had less fibrosis and had begun to resolve with an average severity score of 3.3 (Figure 2). The difference between the two groups at day 60 was highly significant (P < 0.001, using Student’s t-test and the Mann-Whitney rank sum test). Collagen deposition in the thyroid, an indicator of fibrosis, was assessed by Masson’s Trichrome staining. As shown in Figure 3, A and B, there was extensive deposition of collagen (blue) at day 60 in thyroids of FLIP Tg− recipients (left panels), whereas collagen deposition was considerably reduced in thyroids of FLIP Tg+ recipients (right panels) at day 60. The decreased fibrosis in thyroids of Tg+ versus Tg− recipients was also evident at days 20 and 40, as summarized in Table 1. Despite almost complete destruction of thyroids at day 20, most Tg+ and Tg− recipients had normal serum T4 at day 20 (Table 1). Serum T4 was low in almost all mice with 4 to 5+ severity scores at days 40 and 60 but returned to normal in Tg+ recipients with 1 to 3+ severity scores at day 60 (Table 1; data not shown). Because high TSH levels could promote thyroid regeneration in mice with low serum T4, it is important to emphasize that both Tg+ and Tg− mice developed severe G-EAT at day 20 and had low serum T4 at day 40. T4 levels returned to normal only in mice with substantial resolution of their thyroid lesions (Table 1; data not shown). Therefore, thyroid regeneration in this model actually occurs more quickly when T4 levels have returned to normal (Table 1) or when mice develop less severe G-EAT and maintain normal serum T4 throughout the course of disease.6,7,39 These results indicate that the outcome of G-EAT differed in FLIP Tg− and Tg+ littermate recipients given the same pool of WT donor cells; ie, thyroid lesions resolved earlier, and there was less fibrosis in FLIP Tg+ recipients, whereas thyroids of FLIP Tg− recipients had ongoing inflammation and extensive fibrosis.
Figure 2.
Adoptive transfer of MTg-activated WT DBA/1 spleen cells into FLIP Tg+ and FLIP Tg− littermate recipients. G-EAT severity scores of individual mice 20 and 60 days after cell transfer are shown. The severity of thyroid lesions in Tg+ recipients at day 60 was significantly decreased (average severity score, 3.3) compared with those in Tg− littermates (average severity score, 4.5), P < 0.001 at day 60 using both two-tailed Student’s t-test and the Mann-Whitney rank sum test. A significant difference is indicated by the asterisk. Results are pooled from three separate representative experiments.
Table 1.
Expression of Transgenic FLIP Reduces Thyroid Fibrosis
| Day of assessment* | Fibrosis† | Mice with low serum T4/total‡ | ||
|---|---|---|---|---|
| Tg− | Tg+ | Tg− | Tg+ | |
| 20 | +/++ | −/+ | 2/6 | 1/8 |
| 40 | ++/+++ | + | 6/7 | 7/9 |
| 60 | +++ | −/+ | 8/8 | 10/14 |
Splenocytes from MTg-immunized DBA/1 donors were activated in vitro with MTg and IL-12, and 3 × 107 cells were transferred to 500-rad DBA/1 FLIP Tg− and Tg+ littermate recipients. Thyroids were removed 20, 40, and 60 days after cell transfer as indicated. Results are from some of the mice shown in Figure 2.
Intensity of fibrosis between FLIP Tg− and Tg+ recipients determined by Trichrome staining of slides from four to five mice per group was graded as follows: −, not detectable; +, weak; ++, moderate; and +++, strong. The range of fibrosis intensity for each group is indicated (eg, ++/+++). Figure 3, A and B (left panels), are examples of a +++ score, and the right panels in Figure 3, A and B, are examples of a + score.
Numbers of FLIP Tg− or Tg+ recipients with low serum T4level/total numbers of randomly selected recipients tested in each group (eg, 2/6 indicates that two of six recipients had low serum T4level at the day evaluated). Low serum T4level is defined as <3.0 μg/dl.
Ratio of CD4+ versus CD8+ T Cells Differs in G-EAT Thyroids That Will Resolve or Progress to Fibrosis
Our previous studies showed that thyroids of mice with 3 to 4+ G-EAT severity scores that would resolve had more CD8+ T cells than CD4+ T cells at day 20, whereas CD4+ T cells outnumbered CD8+ T cells at day 20 in thyroids with 5+ severity scores that would progress to fibrosis.39 IHC was used to determine whether the ratio of CD4+ versus CD8+ T cells differed in thyroids of FLIP Tg+ and Tg− recipients. Consistent with our previous study,39 CD4+ T cells outnumbered CD8+ T cells at day 20 in FLIP Tg− recipients, which had ongoing inflammation and fibrosis at day 60, whereas CD8+ T cells outnumbered CD4+ T cells at day 20 in FLIP Tg+ recipients whose lesions would resolve by day 60 (data not shown). These differences were even more striking at day 60 (Figure 3). CD4+ T cells (Figure 3, C and D) outnumbered CD8+ T cells (Figure 3, E and F) in FLIP Tg− recipients (Figure 3, C–F, left panels) with ongoing inflammation and fibrosis at day 60 (Figure 3G, left panel), whereas there were fewer T cells in thyroids of FLIP Tg+ recipients, and most were CD8+ (Figure 3, C–F, right panels) at day 60 (Figure 3G, right panel). Numbers of CD4+ and CD8+ T cells were assessed semiquantitatively by counting five to six random high-power fields on slides of three mice in each group with comparable 4 to 5+ G-EAT severity scores (Figure 3G). The more completely resolved thyroids (1 to 3+) had many fewer inflammatory cells at day 60, but most were CD8+ T cells (data not shown). These results are consistent with our previous studies indicating that the ratio of CD4+ versus CD8+ T cells in G-EAT thyroids correlates with and can be a good predictor of the outcome of the autoimmune inflammatory response.39
Outcome of G-EAT in FLIP Tg+ and Tg− Recipients Correlates with the Site of Expression of FLIP and FasL
Our previous studies suggested that up-regulation of FLIP by inflammatory cells might contribute to chronic inflammation by blocking apoptosis of FasL-positive inflammatory cells,24 whereas expression of FLIP by FasL-positive TECs might protect TECs from apoptosis, contributing to resolution.24 Confocal microscopy was used to determine whether the site of expression of FasL (paraffin sections) and FLIP (frozen sections) by TECs versus inflammatory cells differed in thyroids of FLIP Tg+ and Tg− recipients. Our unpublished data showed that FasL staining did not work well on frozen sections, and FLIP staining worked well both on frozen and paraffin sections. Because CD4 and CD8 cannot be stained on paraffin sections, CD4+ and CD8+ T cells, the predominant inflammatory cells in G-EAT thyroids, were identified by cell surface expression of CD3 on paraffin sections (Figure 4A, green), and TECs were identified by PCK staining (Figure 4, B and C, green). Although TECs and inflammatory cells both expressed FasL (Figure 4, A and B, red) in FLIP Tg− (left panels) and Tg+ recipients (right panels), FasL was mainly expressed by CD3+ inflammatory cells in FLIP Tg-recipients (Figure 4A, left panel, yellow, overlay) and by TECs in FLIP Tg+ recipients (Figure 4B, right panel, yellow, overlay). Although TECs and inflammatory cells both expressed FLIP (Figure 4C, red) in FLIP Tg− (left panel) and Tg+ recipients (right panel), FLIP was mainly expressed by PCK + TECs in FLIP Tg+ recipients (Figure 4C, right panel, yellow, overlay). Confocal staining of CD4 (Figure 4D, green) or CD8 (Figure 4E, green) on frozen sections with FLIP (Figure 4, D and E, red) showed that, consistent with IHC (Figure 3), CD4+ T cells were predominant in thyroids of FLIP Tg− recipients (Figure 4, D and E, left panels), whereas CD8+ T cells were predominant in thyroids of FLIP Tg+ recipients (Figure 4, D and E, right panels) at day 60. FLIP was mainly expressed by CD4+ T cells in FLIP Tg− recipients (Figure 4D, left panel, yellow, overlay), whereas very few CD4+ T cells in Tg+ recipients expressed FLIP (Figure 4D, right panel, yellow, overlay). Double positive cells (yellow, overlay) in four to five randomly selected high-power fields of three individual mice per group (magnification, ×600) were manually counted, and the results are summarized in Figure 4F (bars a–e correspond to Figure 4, A–E). Results are representative areas on slides from at least three individual mice examined per group.
Evaluation of Active Caspase-8, Active Capase-3, and Apoptosis in Thyroids of FLIP Tg+ and Tg− Recipients
Fas/FasL-mediated apoptosis involves activation of caspase-8 and its downstream effector, caspase-3, and FLIP inhibits Fas/FasL-mediated apoptosis through its ability to block activation of caspase-8.12,15,16,22,25 Confocal microscopy was used to determine whether the site of expression of active caspase-8 and active caspase-3 by TECs versus inflammatory cells differed in thyroids of FLIP Tg+ and Tg− recipients. CD4+ and CD8+ T cells were identified by cell surface expression of CD4 and CD8 (Figure 5, B, C, E and F, green), and TECs were identified by PCK (Figure 5, A and D, green). Although at day 20, TECs and inflammatory cells both expressed active caspase-8 (Figure 5, A–C, red) in FLIP Tg− (left panels) and Tg+ recipients (right panels), active caspase-8 was mainly expressed by TECs in FLIP Tg− recipients (Figure 5A, left panel, yellow, overlay) and by CD4+ T cells in FLIP Tg+ recipients (Figure 5B, right panel, yellow, overlay). Active caspase-3 was also expressed primarily by TECs in FLIP Tg− recipients (Figure 5D, left panel, yellow, overlay) and by CD4+ T cells in FLIP Tg+ recipients at day 20 (Figure 5E, right panel, yellow, overlay). Very few active caspase-8 or caspase-3+ cells were detected at day 60 (data not shown).
We previously showed that resolution of G-EAT was at least in part due to increased apoptosis of CD4+ effector T cells by a FasL-dependent mechanism.23,24,39 A fluorescence apoptosis kit was used to determine whether there was increased apoptosis of T cells in thyroids of FLIP Tg+ versus Tg− recipients and to determine the distribution of apoptotic cells in these thyroids (Figure 5, G–J). Because detection of apoptotic cells was reduced on frozen sections (our unpublished results), it was necessary to use paraffin sections for these experiments. TECs were identified by PCK (green), T cells were identified by CD3 (green), and apoptosis was detected as red nuclear staining. Many apoptotic cells were detected in thyroids of both FLIP Tg+ and Tg− recipients with G-EAT at day 20 (Figure 5, G and H), whereas only a few apoptotic cells were detected at day 60 (Figure 5, I and J). Apoptosis (red) was mainly in inflammatory cells in FLIP Tg+ recipients both at days 20 (Figure 5H, right panel) and 60 (Figure 5J, right panel) and mainly in TECs in FLIP Tg− recipients at days 20 (Figure 5G, left panel) and 60 (Figure 5I, left panel). Double positive cells (yellow, overlay in Figure 5, A–F; or red circled by green in Figure 5, G–J) in four to five randomly selected high-power fields of three individual mice per group (magnification, ×800) were manually counted and summarized in Figure 5, K and L (bars a–j correspond to Figure 5, A–J). Shown are representative areas on slides of thyroids of at least three individual mice per group. These results are consistent with cell distribution patterns of active caspase-8 and active caspase-3 and support the idea that transgenic FLIP on TECs may function, at least in part, to promote earlier resolution of G-EAT by protecting TECs from apoptosis and inducing increased apoptosis of effector CD4+ T cells through the caspase cascade pathway.
FLIP Protein and mRNA Expression in Thyroids of FLIP Tg+ and Tg− Mice at Day 20
To compare the levels of FLIP in thyroids of FLIP Tg+ and Tg− recipients with G-EAT at day 20, FLIP mRNA and protein expression levels were determined by semiquantitative RT-PCR and Western blot (Figure 6). FLIP mRNA was undetectable in normal Tg− thyroids but was significantly up-regulated in thyroids of FLIP Tg− recipients with severe G-EAT. FLIP mRNA expression in thyroids of normal FLIP Tg+ mice was similar to that in thyroids of FLIP Tg− recipients with severe G-EAT and was further up-regulated in thyroids of FLIP Tg+ recipients with G-EAT at day 20 (Figure 6A). Consistent with the FLIP mRNA expression level, FLIP protein was undetectable in normal Tg− thyroids (Figure 1D) but was significantly up-regulated in thyroids of FLIP Tg− recipients with severe G-EAT and was further increased in thyroids of FLIP Tg+ recipients with severe G-EAT (Figure 6B).
Cytokine and Foxp3 mRNA Expression in Thyroids of FLIP Tg+ and Tg− Mice
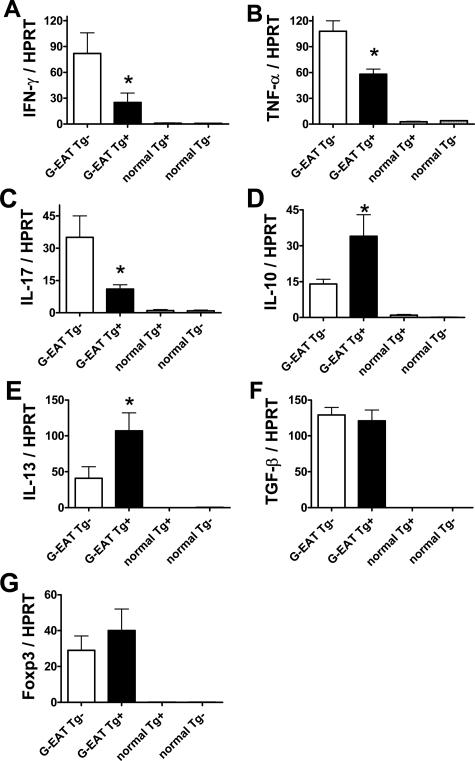
The balance between proinflammatory cytokines such as interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α), and IL-17 and anti-inflammatory cytokines such as IL-10, IL-13, and transforming growth factor-β (TGF-β) is important for development and progression of inflammation in autoimmune diseases including G-EAT.25,34,39,40,41,42,43,44,45,46,47,48 To determine whether proinflammatory and anti-inflammatory cytokines were differentially expressed in thyroids of FLIP Tg+ and Tg− recipients, mRNA expression of particular cytokines in individual thyroids was determined by RT-PCR 20 (data not shown) and 40 (Figure 7) days after cell transfer. Because regulatory T cells can reduce inflammation in many autoimmune diseases including EAT,49,50,51,52 expression of Foxp3 as a marker of regulatory T cells49 was also investigated. Expression of cytokine and Foxp3 mRNA was undetectable in thyroids of normal FLIP Tg+ or Tg− mice (Figure 7, A–G). At day 20, IL-17 mRNA was lower in thyroids of FLIP Tg+ recipients compared with Tg− recipients, whereas expression of other cytokines and Foxp3 mRNA was similar in thyroids of both FLIP Tg+ and Tg− recipients with G-EAT (data not shown). By day 40, expression of proinflammatory cytokines such as IFN-γ, TNF-α, and IL-17 was lower (Figure 7, A–C), whereas expression of anti-inflammatory cytokines such as IL-10 and IL-13 was higher (Figure 7, D and E) in thyroids of FLIP Tg+ compared with Tg− recipients, even though G-EAT severity scores were similar (4 to 5+) for both groups. Expression of TGF-β and Foxp3 was similar in thyroids of FLIP Tg+ and Tg− recipients with G-EAT (Figure 7, F and G). IL-4 expression could not be assessed because of very low expression levels at days 20 and 40. These results suggested that expression of proinflammatory and anti-inflammatory cytokines in recipient thyroids correlated with differences in the outcome of G-EAT seen at day 60 (Figure 2) in FLIP Tg+ versus Tg− recipients, consistent with the increased apoptosis of inflammatory cells in thyroids of FLIP Tg+ recipients compared with Tg− recipients (Figure 5).
Figure 7.
Cytokine and Foxp3 mRNA expression in thyroids of FLIP Tg+ and Tg− recipients. A–G: mRNA was isolated from a single thyroid lobe of individual mice 40 days after cell transfer and amplified as described in Materials and Methods. All thyroids had comparable 4 to 5+ severity scores. IFN-γ, TNF-α, IL-17, IL-10, IL-13, TGF-β, and Foxp3 mRNA (A–G) was undetectable in thyroids of normal Tg+ or Tg− mice. At day 40, IFN-γ, TNF-α, and IL-17 mRNA expression (A–C) was lower, IL-10 and IL-13 mRNA expression (D and E) was higher, TGF-β and Foxp3 mRNA expression (F and G) was similar in thyroids of FLIP Tg+ compared with Tg− littermate recipients. Results are expressed as the mean ratio of cytokine densitometric U/hypoxanthine phosphoribosyltransferase ± SEM (×100) of five to six mice per group and are representative of three independent experiments. A significant difference between G-EAT FLIP Tg− recipients and Tg+ recipients is indicated by the asterisk (P < 0.05).
Discussion
The caspase-8 inhibitor FLIP is a well-defined inhibitor of Fas receptor signaling.8,28 Overexpression of FLIP inhibits Fas-mediated apoptosis of T lymphocytes28,30 and can exacerbate autoimmune diseases such as EAE and multiple sclerosis.13,53 In addition, TECs from patients with Grave’s disease highly express FLIP, and expression of cFLIP was shown to protect the TECs from Fas-mediated apoptosis.11 We are currently generating primary thyrocyte cultures from thyroids of FLIP Tg+ mice and Tg− littermates to directly determine whether FLIP expression on TECs in this model protects TECs from Fas-mediated and tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in the presence or absence of proinflammatory cytokines.
Our previous studies suggested that the site of expression of FLIP and FasL (TECs versus inflammatory cells) was predictive of whether thyroid lesions in mice with autoimmune thyroiditis would resolve or have chronic inflammation.23,24 When FLIP and FasL were both expressed by TECs, G-EAT lesions resolved earlier than when these molecules were expressed primarily by inflammatory cells, suggesting that expression of FLIP by TECs might promote G-EAT resolution by protecting FasL-expressing TECs from apoptosis.23,24 In this study, we asked whether constitutive expression of FLIP on TECs would promote earlier resolution of G-EAT in DBA/1 mice whose lesions normally do not resolve. As shown in Figure 1 and by others,54 expression of FLIP as a transgene in the thyroid has no effects on thyroid histology or function. Both FLIP Tg+ and FLIP Tg− recipients of WT splenocytes developed very severe G-EAT with comparable severity scores at day 20 (Figure 2). By day 60, thyroid lesions were beginning to resolve in most Tg+ recipients, whereas most Tg− littermates had continuing inflammation and fibrosis of the thyroid. When Trichrome staining was used to determine the extent of collagen deposition (fibrosis) in thyroids, there was more fibrosis at day 60 in thyroids of FLIP Tg− recipients compared with Tg+ littermates (Figures 2 and 3; Table 1). These results are consistent with our hypothesis that constitutive expression of FLIP on TECs results in less fibrosis and promotes earlier resolution of G-EAT.
CD8+ T cells can promote CD4+ T cell apoptosis in EAE,55,56,57 and CD8+ T cell and Fas/FasL interactions are required for early resolution of G-EAT.6,23,24,25,26 In the current study, effector CD4+ T cells always outnumbered CD8+ T cells in thyroids of FLIP Tg− recipients in which thyroid lesions did not resolve and progressed to fibrosis, whereas CD8+ T cells outnumbered CD4+ T cells in thyroids of FLIP Tg+ recipients in which lesions began to resolve earlier. CD4+ T cells were also predominant in G-EAT thyroids when anti-CD8 or anti-FasL was used to inhibit resolution.23,25 The fact that CD8+ T cells always outnumbered CD4+ T cells when G-EAT would resolve is consistent with the increased detection of apoptotic CD3+ T cells and stronger expression of active caspase-8 and active caspase-3 in CD4+ T cells in thyroids of FLIP Tg+ recipients compared with FLIP Tg− recipients. These results are consistent with the idea that CD8+ T cells may control the expansion of activated CD4+ T cells by inducing apoptosis of CD4+ effector T cells.23,39
Resolution of inflammation requires the removal of inflammatory cells that were recruited and expanded during the response.58 Our previous studies demonstrated a role for Fas-mediated apoptosis in G-EAT resolution.25 In this study, the two different outcomes of G-EAT in FLIP Tg+ and FLIP Tg− recipients were shown to be at least in part due to modulation of Fas-mediated apoptosis by expressing the anti-apoptotic molecule FLIP on TECs. The site of expression of FLIP and FasL proteins differed in thyroids of FLIP Tg+ and FLIP Tg− recipients. In FLIP Tg− recipients with chronic inflammation and fibrosis, FasL and FLIP were mainly expressed by inflammatory cells, whereas in FLIP Tg+ recipients with resolving G-EAT, FLIP and FasL were primarily expressed by TECs (Figure 4). Consistent with the FLIP and FasL distribution patterns, active caspase-8 and active caspase-3 were also mainly expressed by CD4+ inflammatory cells in FLIP Tg+ recipients with resolving G-EAT but were primarily expressed by TECs, resulting in apoptosis of TECs in FLIP Tg− recipients with chronic inflammation and fibrosis. These results are consistent with our previous results indicating that the site of expression of FLIP and FasL can predict whether G-EAT lesions will resolve or have chronic inflammation.24 The results presented here suggest that up-regulation of FLIP by inflammatory cells might confer resistance to Fas-mediated apoptosis contributing to chronic inflammation in FLIP Tg− recipients. We are currently generating transgenic mice expressing FLIP on CD2+ lymphocytes to directly test the hypothesis that overexpression of FLIP on inflammatory cells will result in chronic inflammation. On the other hand, TECs in FLIP Tg+ recipients with resolving G-EAT constitutively expressed FLIP, and this may function to protect TECs from apoptosis. TECs in FLIP Tg+ recipients also expressed FasL (Figure 4), which may contribute to the increased apoptosis of CD4+ effector T cells and earlier resolution of G-EAT in FLIP Tg+ recipients.23,24,26
Inflammation is associated with production of proinflammatory cytokines that recruit and activate inflammatory cells to amplify the response. IFN-γ, TNF-α, and IL-17 are major proinflammatory cytokines important for development and progression of most autoimmune diseases.40,41,42,43,44,46,47,48 Although the mRNA levels of IFN-γ and TNF-α were up-regulated to a similar extent at day 20 in both FLIP Tg+ and Tg− recipients with G-EAT (data not shown), IFN-γ, IL-17, and TNF-α mRNA (Figure 7, A–C) were all decreased in thyroids of FLIP Tg+ compared with Tg− recipients at day 40. We have shown that neutralization of TNF-α or deficiency of IFN-γ promotes G-EAT resolution in this model,39,46 and proinflammatory cytokines can promote destruction of TECs by promoting TEC apoptosis.11,47,48 Thus, expression of proinflammatory cytokines such as IFN-γ, TNF-α, and IL-17 in recipient thyroids correlates with and might contribute to the different outcome of G-EAT in FLIP Tg+ versus FLIP Tg− recipients. The reduced proinflammatory cytokines in FLIP Tg+ recipients might, at least in part, occur as a result of increased apoptosis of CD4+ T cells secreting these cytokines in FLIP Tg+ recipients. Cytokines such as IL-10, IL-13, and TGF-β are can function as immunoregulatory or anti-inflammatory molecules that control the proinflammatory cytokine response.11,45,48,51,58 Under physiological conditions, they can function to limit potentially injurious effects of sustained or excessive inflammatory reactions. Under pathological conditions, anti-inflammatory cytokines are unable to control the proinflammatory activities that predominate in immune-mediated diseases.45,48,51 In our G-EAT model, although the mRNA levels of IL-10, IL-13, and TGF-β were up-regulated to a similar extent at day 20, when there was strong inflammation in both FLIP Tg+ and Tg− recipients (data not shown), expression of both IL-10 and IL-13 mRNA was higher in thyroids of FLIP Tg+ compared with Tg− recipients at day 40 (Figure 7, D and E). Therefore, increased expression of anti-inflammatory cytokines in recipient thyroids might contribute to the different outcomes of G-EAT in FLIP Tg+ versus FLIP Tg− recipients by inhibiting the function of proinflammatory cytokines. Because most active caspase-8+ and active caspase-3+ cells in FLIP Tg+ recipients are CD4+ T cells (Figure 5 B and E, right panels) and Th1 cells are more sensitive than Th2 cells to caspase-mediated cell death,28,59,60 it is perhaps not surprising that there is a relatively higher expression of Th2 cytokines in FLIP Tg+ recipients. Thus, the equilibrium between proinflammatory and anti-inflammatory cytokines might direct the progression of inflammation in G-EAT. One anti-inflammatory cytokine that was not increased in thyroids of FLIP Tg+ mice is TGF-β (Figure 7F). Because TGF-β is profibrotic45 and fibrosis was reduced in FLIP Tg+ recipients (Figure 3 and Table 1), the interplay between the anti-inflammatory and profibrotic functions of TGF-β might lead to a net result of similar TGF-β expression in thyroids of Tg+ (predominantly anti-inflammatory) versus Tg− (predominantly profibrotic) mice. In this regard, we have shown previously that neutralization of TGF-β decreases fibrosis and promotes G-EAT resolution in this model.34 Regulatory T cells, identifiable by expression of the transcription factor Foxp3, can inhibit several autoimmune diseases including EAT.49,50,51,52 Assessment of Foxp3 mRNA (Figure 7G) and protein (data not shown) expression was similar in thyroids of FLIP Tg+ compared with Tg− recipients at days 20 and 40, suggesting that regulatory T cells do not play a primary role in G-EAT resolution in FLIP Tg+ mice.
In conclusion, as predicted by our hypothesis, transgenic expression of FLIP in the thyroid partially protects TECs from apoptosis and promotes earlier resolution of G-EAT, directly demonstrating that FLIP plays an important role in G-EAT resolution. This G-EAT model provides a well-characterized animal model that can be useful to clarify the mechanisms by which tissues can be induced to up-regulate FLIP and FasL to protect themselves from apoptosis or induce other cells to be destroyed. Such studies may contribute to the development of strategies for treatment of human autoimmune diseases.
Acknowledgments
We thank Patti Mierzwa for excellent technical assistance, Dr. Motoyasu Saji (The Ohio State University, Columbus, OH) for providing the RTg promoter, Dr. Susanne Lens (Netherlands Cancer Institute) for providing the cFLIP plasmid, Dr. Daniel Altschuler (University of Pittsburgh) for providing the PCCL3 thyroid cell line, and Dr. George Smith and Mark Foecking (University of Missouri) for assistance with FLAG-tagged protein detection. We also thank Dr. Lixing Reneker for helpful discussions.
Footnotes
Address reprint requests to Dr. Helen Braley-Mullen, Division of Immunology and Rheumatology, Department of Medicine, University of Missouri, NE307 Medical Sciences, Columbia, MO 65212. E-mail: mullenh@health.missouri.edu.
Supported by National Institutes of Health grant DK35527.
References
- Lira SA, Martin AP, Marinkovic T, Furtado GC. Mechanisms regulating lymphocytic infiltration of the thyroid in murine models of thyroiditis. Crit Rev Immunol. 2005;25:251–262. doi: 10.1615/critrevimmunol.v25.i4.10. [DOI] [PubMed] [Google Scholar]
- Braley-Mullen H, Johnson M, Sharp GC, Kyriakos M. Induction of experimental autoimmune thyroiditis in mice with in vitro activated splenic T cells. Cell Immunol. 1985;93:132–143. doi: 10.1016/0008-8749(85)90394-6. [DOI] [PubMed] [Google Scholar]
- Conaway DH, Giraldo AA, David CS, Kong YC. In situ analysis of T cell subset composition in experimental autoimmune thyroiditis after adoptive transfer of activated spleen cells. Cell Immunol. 1990;125:247–253. doi: 10.1016/0008-8749(90)90078-6. [DOI] [PubMed] [Google Scholar]
- Braley-Mullen H, Sharp GC, Bickel JT, Kyriakos M. Induction of severe granulomatous experimental autoimmune thyroiditis in mice by effector cells activated in the presence of anti-interleukin 2 receptor antibody. J Exp Med. 1991;173:899–912. doi: 10.1084/jem.173.4.899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braley-Mullen H, Sharp GC, Tang H, Chen K, Kyriakos M, Bickel JT. Interleukin-12 promotes activation of effector cells that induce a severe destructive granulomatous form of murine experimental autoimmune thyroiditis. Am J Pathol. 1998;152:1347–1358. [PMC free article] [PubMed] [Google Scholar]
- Braley-Mullen H, McMurray RW, Sharp GC, Kyriakos M. Regulation of the induction and resolution of granulomatous experimental autoimmune thyroiditis in mice by CD8+ T cells. Cell Immunol. 1994;153:492–504. doi: 10.1006/cimm.1994.1045. [DOI] [PubMed] [Google Scholar]
- Braley-Mullen H, Sharp GC. Adoptive transfer murine model of granulomatous experimental autoimmune thyroiditis. Int Rev Immunol. 2000;19:535–555. doi: 10.3109/08830180009088511. [DOI] [PubMed] [Google Scholar]
- Thome M, Tschopp J. Regulation of lymphocyte proliferation and death by FLIP. Nat Rev Immunol. 2001;1:50–58. doi: 10.1038/35095508. [DOI] [PubMed] [Google Scholar]
- Nagata S, Golstein P. The Fas death factor. Science. 1995;267:1449–1456. doi: 10.1126/science.7533326. [DOI] [PubMed] [Google Scholar]
- Stassi G, De Maria R. Autoimmune thyroid disease: new models of cell death in autoimmunity. Nat Rev Immunol. 2002;2:195–204. doi: 10.1038/nri750. [DOI] [PubMed] [Google Scholar]
- Stassi G, Di Liberto D, Todaro M, Zeuner A, Ricci-Vitiani L, Stoppacciaro A, Ruco L, Farina F, Zummo G, De Maria R. Control of target cell survival in thyroid autoimmunity by T helper cytokines via regulation of apoptotic proteins. Nat Immunol. 2000;1:483–488. doi: 10.1038/82725. [DOI] [PubMed] [Google Scholar]
- Giordano C, Richiusa P, Bagnasco M, Pizzolanti G, Di Blasi F, Sbriglia MS, Mattina A, Pesce G, Montagna P, Capone F, Misiano G, Scorsone A, Pugliese A, Galluzzo A. Differential regulation of Fas-mediated apoptosis in both thyrocyte and lymphocyte cellular compartments correlates with opposite phenotypic manifestations of autoimmune thyroid disease. Thyroid. 2001;11:233–244. doi: 10.1089/105072501750159615. [DOI] [PubMed] [Google Scholar]
- Semra YK, Seidi OA, Sharief MK. Overexpression of the apoptosis inhibitor FLIP in T cells correlates with disease activity in multiple sclerosis. J Neuroimmunol. 2001;113:268–274. doi: 10.1016/s0165-5728(00)00443-4. [DOI] [PubMed] [Google Scholar]
- Tisch R, McDevitt H. Insulin-dependent diabetes mellitus. Cell. 1996;85:291–297. doi: 10.1016/s0092-8674(00)81106-x. [DOI] [PubMed] [Google Scholar]
- Suvannavejh GC, Dal Canto MC, Matis LA, Miller SD. Fas-mediated apoptosis in clinical remissions of relapsing experimental autoimmune encephalomyelitis. J Clin Invest. 2000;105:223–231. doi: 10.1172/JCI8561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabelko-Downes KA, Cross AH, Russell JH. Dual role for Fas ligand in the initiation of and recovery from experimental allergic encephalomyelitis. J Exp Med. 1999;189:1195–1205. doi: 10.1084/jem.189.8.1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su X, Hu Q, Kristan JM, Costa C, Shen Y, Gero D, Matis LA, Wang Y. Significant role for Fas in the pathogenesis of autoimmune diabetes. J Immunol. 2000;164:2523–2532. doi: 10.4049/jimmunol.164.5.2523. [DOI] [PubMed] [Google Scholar]
- Itoh N, Imagawa A, Hanafusa T, Waguri M, Yamamoto K, Iwahashi H, Moriwaki M, Nakajima H, Miyagawa J, Namba M, Makino S, Nagata S, Kono N, Matsuzawa Y. Requirement of Fas for the development of autoimmune diabetes in nonobese diabetic mice. J Exp Med. 1997;186:613–618. doi: 10.1084/jem.186.4.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang SM, Schneider DB, Lin Z, Hanahan D, Dichek DA, Stock PG, Baekkeskov S. Fas ligand expression in islets of Langerhans does not confer immune privilege and instead targets them for rapid destruction. Nat Med. 1997;3:738–743. doi: 10.1038/nm0797-738. [DOI] [PubMed] [Google Scholar]
- Mahiou J, Walter U, Lepault F, Godeau F, Bach JF, Chatenoud L. In vivo blockade of the Fas-Fas ligand pathway inhibits cyclophosphamide-induced diabetes in NOD mice. J Autoimmun. 2001;16:431–440. doi: 10.1006/jaut.2000.0476. [DOI] [PubMed] [Google Scholar]
- Batteux F, Lores P, Bucchini D, Chiocchia G. Transgenic expression of Fas ligand on thyroid follicular cells prevents autoimmune thyroiditis. J Immunol. 2000;164:1681–1688. doi: 10.4049/jimmunol.164.4.1681. [DOI] [PubMed] [Google Scholar]
- Giordano C, Stassi G, De Maria R, Todaro M, Richiusa P, Papoff G, Ruberti G, Bagnasco M, Testi R, Galluzzo A. Potential involvement of Fas and its ligand in the pathogenesis of Hashimoto’s thyroiditis. Science. 1997;275:960–963. doi: 10.1126/science.275.5302.960. [DOI] [PubMed] [Google Scholar]
- Wei Y, Chen K, Sharp GC, Yagita H, Braley-Mullen H. Expression and regulation of Fas and Fas ligand on thyrocytes and infiltrating cells during induction and resolution of granulomatous experimental autoimmune thyroiditis. J Immunol. 2001;167:6678–6686. doi: 10.4049/jimmunol.167.11.6678. [DOI] [PubMed] [Google Scholar]
- Wei Y, Chen K, Sharp GC, Braley-Mullen H. FLIP and FasL expression by inflammatory cells vs. thyrocytes can be predictive of chronic inflammation or resolution of autoimmune thyroiditis. Clin Immunol. 2003;108:221–233. doi: 10.1016/s1521-6616(03)00146-3. [DOI] [PubMed] [Google Scholar]
- Wei Y, Chen K, Sharp GC, Braley-Mullen H. Fas ligand is required for resolution of granulomatous experimental autoimmune thyroiditis. J Immunol. 2004;173:7615–7621. doi: 10.4049/jimmunol.173.12.7615. [DOI] [PubMed] [Google Scholar]
- Tang H, Chen K, Sharp GC, McKee L, Braley-Mullen H. Apoptosis of thyrocytes and effector cells during induction and resolution of granulomatous experimental autoimmune thyroiditis. Int Immunol. 2000;12:1629–1639. doi: 10.1093/intimm/12.12.1629. [DOI] [PubMed] [Google Scholar]
- White CA, McCombe PA, Pender MP. The role of Fas, Fas ligand and bcl-2 in T cell apoptosis in the central nervous system in experimental autoimmune encephalomyelitis. J Neuroimmunol. 1998;82:47–55. doi: 10.1016/S0165-5728(97)00187-2. [DOI] [PubMed] [Google Scholar]
- Budd RC, Yeh WC, Tschopp J. cFLIP regulation of lymphocyte activation and development. Nat Rev Immunol. 2006;6:196–204. doi: 10.1038/nri1787. [DOI] [PubMed] [Google Scholar]
- Rasper DM, Vaillancourt JP, Hadano S, Houtzager VM, Seiden I, Keen SL, Tawa P, Xanthoudakis S, Nasir J, Martindale D, Koop BF, Peterson EP, Thornberry NA, Huang J, MacPherson DP, Black SC, Hornung F, Lenardo MJ, Hayden MR, Roy S, Nicholson DW. Cell death attenuation by ‘Usurpin’, a mammalian DED-caspase homologue that precludes caspase-8 recruitment and activation by the CD-95 (Fas, APO-1) receptor complex. Cell Death Differ. 1998;5:271–288. doi: 10.1038/sj.cdd.4400370. [DOI] [PubMed] [Google Scholar]
- Tschopp J, Irmler M, Thome M. Inhibition of fas death signals by FLIPs. Curr Opin Immunol. 1998;10:552–558. doi: 10.1016/s0952-7915(98)80223-9. [DOI] [PubMed] [Google Scholar]
- Golks A, Brenner D, Fritsch C, Krammer PH, Lavrik IN. c-FLIPR, a new regulator of death receptor-induced apoptosis. J Biol Chem. 2005;280:14507–14513. doi: 10.1074/jbc.M414425200. [DOI] [PubMed] [Google Scholar]
- Mitsiades N, Poulaki V, Mitsiades CS, Koutras DA, Chrousos GP. Apoptosis induced by FasL and TRAIL/Apo2L in the pathogenesis of thyroid diseases. Trend Endocrinol Metab. 2001;12:384–390. doi: 10.1016/s1043-2760(01)00441-6. [DOI] [PubMed] [Google Scholar]
- Lens SM, Kataoka T, Fortner KA, Tinel A, Ferrero I, MacDonald RH, Hahne M, Beermann F, Attinger A, Orbea HA, Budd RC, Tschopp J. The caspase 8 inhibitor c-FLIP modulates T-cell receptor-induced proliferation but not activation-induced cell death of lymphocytes. Mol Cell Biol. 2002;22:5419–5433. doi: 10.1128/MCB.22.15.5419-5433.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen K, Wei Y, Sharp GC, Braley-Mullen H. Inhibition of TGFβ1 by anti-TGFβ1 antibody or lisinopril reduced thyroid fibrosis in granulomatous experimental autoimmune thyroiditis. J Immunol. 2002;169:6530–6538. doi: 10.4049/jimmunol.169.11.6530. [DOI] [PubMed] [Google Scholar]
- Chen K, Wei Y, Sharp GC, Braley-Mullen H. Characterization of thyroid fibrosis in a murine model of granulomatous experimental autoimmune thyroiditis. J Leukocyte Biol. 2000;68:828–835. [PubMed] [Google Scholar]
- Chen K, Wei Y, Sharp GC, Braley-Mullen H. Balance of proliferation and cell death between thyrocytes and myofibroblasts regulates thyroid fibrosis in granulomatous experimental autoimmune thyroiditis (G-EAT). J Leukocyte Biol. 2005;77:166–172. doi: 10.1189/jlb.0904538. [DOI] [PubMed] [Google Scholar]
- Tang H, Sharp GC, Chen K, Braley-Mullen H. The kinetics of cytokine gene expression in the thyroids of mice developing granulomatous experimental autoimmune thyroiditis. J Autoimmunity. 1998;11:581–589. doi: 10.1006/jaut.1998.0247. [DOI] [PubMed] [Google Scholar]
- Tang H, Sharp GC, Peterson K, Braley-Mullen H. IFN-γ-deficient mice develop severe granulomatous experimental autoimmune thyroiditis with eosinophil infiltration in thyroids. J Immunol. 1998;160:5105–5112. [PubMed] [Google Scholar]
- Chen K, Wei Y, Sharp GC, Braley-Mullen H. Mechanisms of spontaneous resolution versus fibrosis in granulomatous experimental autoimmune thyroiditis. J Immunol. 2003;171:6236–6243. doi: 10.4049/jimmunol.171.11.6236. [DOI] [PubMed] [Google Scholar]
- Kassiotis G, Kollias G. TNF and receptors in organ-specific autoimmune disease: multi-layered functioning mirrored in animal models. J Clin Invest. 2001;107:1507–1508. doi: 10.1172/JCI13362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrington L, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol. 2005;6:1123–1132. doi: 10.1038/ni1254. [DOI] [PubMed] [Google Scholar]
- Dong C. Diversification of T-helper-cell lineages: finding the family root of IL-17-producing cells. Nat Rev Immunol. 2006;6:329–333. doi: 10.1038/nri1807. [DOI] [PubMed] [Google Scholar]
- Murphy KM, Reiner SL. The lineage decisions of helper T cells. Nat Rev Immunol. 2002;2:933–944. doi: 10.1038/nri954. [DOI] [PubMed] [Google Scholar]
- Seder RA, Paul WE. Acquisition of lymphokine-producing phenotype by CD4+ T cells. Annu Rev Immunol. 1994;12:635–673. doi: 10.1146/annurev.iy.12.040194.003223. [DOI] [PubMed] [Google Scholar]
- Li MO, Wan YW, Sanjabi S, Robertson A, Flavell RA. Transforming growth factor-β regulation of immune responses. Annu Rev Immunol. 2006;24:99–147. doi: 10.1146/annurev.immunol.24.021605.090737. [DOI] [PubMed] [Google Scholar]
- Chen K, Wei Y, Sharp GC, Braley-Mullen H. Decreasing TNFα results in less fibrosis and earlier resolution of granulomatous experimental autoimmune thyroiditis. J Leuk Biol. 2007;81:306–314. doi: 10.1189/jlb.0606402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang SH, Bretz JD, Phelps E, Mezosi E, Arscott PL, Utsugi S, Baker JR., Jr A unique combination of inflammatory cytokines enhances apoptosis of thyroid follicular cells and transforms nondestructive to destructive thyroiditis in experimental autoimmune thyroiditis. J Immunol. 2002;168:2470–2474. doi: 10.4049/jimmunol.168.5.2470. [DOI] [PubMed] [Google Scholar]
- Bretz JD, Arscott PL, Myc A, Baker JR., Jr Inflammatory cytokine regulation of Fas-mediated apoptosis in thyroid follicular cells. J Biol Chem. 1999;274:25433–25438. doi: 10.1074/jbc.274.36.25433. [DOI] [PubMed] [Google Scholar]
- Shevach EM. Regulatory T cells in autoimmunity. Annu Rev Immunol. 2000;18:423–429. doi: 10.1146/annurev.immunol.18.1.423. [DOI] [PubMed] [Google Scholar]
- Morris GP, Chen L, Kong YM. CD137 signaling interferes with activation and function of CD4+CD25+ regulatory T cells in induced tolerance to experimental autoimmune thyroiditis. Cell Immunol. 2003;226:20–29. doi: 10.1016/j.cellimm.2003.11.002. [DOI] [PubMed] [Google Scholar]
- Gangi E, Vasu C, Cheatem D, Prabhakhar B. IL-10 producing CD4+CD25+ regulatory T cells play a critical role in granulocyte macrophage colony stimulatory factor-induced suppression of experimental autoimmune thyroiditis. J Immunol. 2005;174:7006–7013. doi: 10.4049/jimmunol.174.11.7006. [DOI] [PubMed] [Google Scholar]
- Yu S, Maiti P, Dyson M, Jain R, Braley-Mullen H. B cell-deficient NOD.H-2h4 mice have CD4+CD25+ T regulatory cells that inhibit the development of spontaneous autoimmune thyroiditis. J Exp Med. 2006;203:349–358. doi: 10.1084/jem.20051438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Djerbi M, Abdul-Majid KB, Abedi-Valugerdi M, Olsson T, Harris RA, Grandien A. Expression of the long form of human FLIP by retroviral gene transfer of hemopoietic stem cells exacerbates experimental autoimmune encephalomyelitis. J Immunol. 2003;170:2064–2073. doi: 10.4049/jimmunol.170.4.2064. [DOI] [PubMed] [Google Scholar]
- Wang SH, Arscott P, Wu P, Baker JR., Jr No apparent damage in the thyroid of transgenic mice expressing antiapoptotic FLIP. Thyroid. 2006;16:1–8. doi: 10.1089/thy.2006.16.1. [DOI] [PubMed] [Google Scholar]
- Sun D, Qin Y, Chluba J, Epplen JT, Wekerle H. Suppression of experimentally induced autoimmune encephalomyelitis by cytolytic T-T cell interactions. Nature. 1988;332:843–845. doi: 10.1038/332843a0. [DOI] [PubMed] [Google Scholar]
- Jiang H, Chess L. The specific regulation of immune responses by CD8+ T cells restricted by the MHC class Ib molecule, Qa-1. Annu Rev Immunol. 2000;18:185–216. doi: 10.1146/annurev.immunol.18.1.185. [DOI] [PubMed] [Google Scholar]
- Jiang H, Braunstein NS, Yu B, Winchester R, Chess L. CD8+ T cells control the TH phenotype of MBP-reactive CD4+ T cells in EAE mice. Proc Natl Acad Sci USA. 2001;98:6301–6306. doi: 10.1073/pnas.101123098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serhan C, Savill J. Resolution of inflammation: the beginning programs the end. Nat Immunol. 2005;6:1191–1197. doi: 10.1038/ni1276. [DOI] [PubMed] [Google Scholar]
- Zhang X, Brunner T, Carter L, Dutton RW, Rogers P, Bradley L, Sato T, Reed JC, Green D, Swain SL. Unequal death in T helper cell (Th)1 and Th2 effectors: Th1, but not Th2, effectors undergo rapid Fas/FasL-mediated apoptosis. J Exp Med. 1997;185:1837–1849. doi: 10.1084/jem.185.10.1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang XR, Zhang LY, Devadas S, Li L, Keegan AD, Shi YF. Reciprocal expression of TRAIL and CD95L in Th1 and Th2 cells: role of apoptosis in T helper subset differentiation. Cell Death Differ. 2003;10:203–210. doi: 10.1038/sj.cdd.4401138. [DOI] [PubMed] [Google Scholar]