Abstract
Background/Aim
Mitophagy is a cardinal process for maintaining healthy and functional mitochondria. A decline in mitophagy has been associated with age-related pathologies. We aimed to investigate mitophagy changes in age-related balance problems using an animal model.
Materials and Methods
C57BL/6J mice were divided into young (1 month old) and aged (12 months old) groups. Balance performance, mitochondrial DNA integrity, ATP content, mitophagic process, and mitophagy-related genes and proteins were investigated in both groups.
Results
Balance and motor performance were reduced in the aged group. Mitochondrial DNA integrity and ATP content, and mRNA levels of PINK1, Parkin, BNIP3, AMBRA1, MUL1, NIX, Bcl2-L-13, Atg3, Atg5, Atg12, and Atg13 in the vestibule were significantly lower in aged mice compared with those in young mice. The protein levels of PINK1, Parkin, BNIP3, LC3B, and OXPHOS subunits were significantly decreased in the aged vestibule. Mitophagosome and mitophagolysosome counts and the immunohistochemical expression of Parkin and BNIP3 were also decreased in the saccule, utricle, and crista ampullaris in the aged group.
Conclusion
A general decrease in mitophagy with aging might be attributed to a decrease in cellular function in the aged vestibule during the development of age-related balance problems.
Keywords: Mitophagy, aging, vestibular system, mice
The vestibular system is a sensory organ that provides information regarding head movement, position, and balance (1). The peripheral vestibular system consists of two otolith organs, the utricle and saccule, which sense linear acceleration and the crista ampullaris in three semicircular canals, for sensing angular acceleration. Vestibular organs degenerate with age and functional changes have been observed in vestibular cells; however, the underlying mechanisms remain unclear (2). Impairment of balance due to age-related degeneration of the vestibular system leads to falls and significant morbidity in older adults. Aging is also associated with mitochondrial dysfunction (3). Mitochondria play a cardinal role in supplying energy in the form of adenosine triphosphate (ATP) and influence all aspects of cellular function (4). The accumulation of damaged mitochondria compromises cell and tissue functions during aging (5). Autophagy maintains healthy cellular components by regulating the turnover of damaged cellular organelles (6). Damaged mitochondria are degraded by selective autophagy, mitophagy, which involves the degradation of damaged and dysfunctional mitochondria through the formation of mitophagolysosomes (7). The appropriate induction of autophagy prevents the age-dependent accumulation of dysfunctional cellular components and contributes to cellular signaling function (8). However, reduced mitophagy has been observed in dysfunctional aged tissues and organs, leading to various age-related pathologies (3,9). Compromised mitophagy may be a cause of cellular dysfunction in the peripheral vestibular systems of patients with age-related balance impairment. The present study aimed to investigate any changes in mitophagy in aged peripheral vestibular systems using a mouse model.
Materials and Methods
Mice and animal care. All animal experiments were approved by the Institutional Animal Care and Use Committee of Chosun University (approval no. CIACUC2022-A0006) and performed in accordance with the ARRIVE guidelines and strict regulations approved by the committee. C57BL/6J mice (Mus musculus) were obtained from the Animal Facility of Aging Science at the KBSI Gwangju Center (Gwangju, South Korea). Mice were randomly divided into two groups (young: 1 month of age, n=5; age: 12 months, n=5). All mice were euthanized by cervical dislocation under 5% isoflurane anesthesia after the rotarod test and the peripheral vestibular structures were isolated from the mice.
Rotarod test. The rotarod test was performed using a rotarod apparatus (Panlab/Harvard Apparatus, Barcelona, Spain) to evaluate sensorimotor coordination and balance in the rodents. C57BL/6J mice were first habituated to stay on the rod for 60 s at a constant speed of five rotations per minute (rpm). This habituation procedure was repeated for a total of three trials per day separated by 10 min intervals for three days. After training, mice underwent two test sessions. They were first placed in separate lanes on the rod and subjected to an accelerating speed (from 4 to 40 rpm) of over 300 s. Next, mice were tested on the rotarod at constant speeds (4, 16, 28, and 40 rpm). The latency to fall (s) and speed at which they fell (rpm) were recorded for statistical analysis. Five trials were conducted for each mouse.
Real-time PCR analysis. To test mitochondrial DNA integrity, 20 ng of total DNA was isolated from whole vestibular organ tissues, except for the cochlea of the mouse inner ear. The mtDNA long fragment (10.1 kb) was amplified using the following primers: 5’-GCCAGCCTGACCCATAGCCATAATAT-3’ and 5’-GAGAGATT TTATGGGTGTAATGCGG-3’. The short mtDNA fragment (117 bp) was amplified using the following primers: 5’- CCCAGCTACTA CCATCATTCAAGT-3’ and 5’-GATGGT TTGGGAGATTGGTT GATGT-3’. Real-time PCR was performed using the SYBR Premix Ex Taq kit (TaKaRa Bio, Shiga, Japan) according to the manufacturer’s instructions. The short mtDNA fragment was used as a normalization factor for mtDNA copies.
To analyze mitophagy-related genes, total RNA was extracted from whole vestibular organ tissues, except for the cochlea of the mouse inner ear. RNA was reverse-transcribed using an M-MLV cDNA Synthesis Kit (Enzynomics, Daejeon, Republic of Korea) according to manufacturer’s instructions. Real-time PCR was performed using the SYBR Premix Ex Taq kit (TaKaRa Bio) according to the manufacturer’s instructions. Primers used for real-time PCR were as follows: PINK1 forward, 5’-GCTTGCCAATCCCTTCTATG-3’, reverse, 5’-CTCTCGCTGGAGCAGTGAC-3’; Parkin forward, 5’-AAACCGGATGAGTGGTGAGT-3’, reverse, 5’-AGCTACCGACG TGTCCTTGT-3’; AMBRA1 forward, 5’-CTACTGGGACCAGCTAA GTGAAA-3’, reverse, 5’-ACGTGGCTCTGCTGGTTC-3’; MUL1 forward, 5’-GTCATCGAAGGAGCTGTGC-3’, reverse, 5’-GTAGTT CGGTTCCACACCATC-3’; NIX forward, 5’-GCAGGGACTAGCT CTCAG-3’, reverse, 5’-TGCTCAGTCGCTTTCCAATA-3’; BNIP3 forward, 5’-GCTCCCAGACACCACAAGAT-3’, reverse, 5’-TGA GAGTAGCTGTGCGCTTC-3’; BCL2-L-13 forward, 5’- AGCAGAA TTCATGGCGTCCTCTACGACTGC-3’, reverse, 5’-AGCAGAATTC TTACTTTCTTCTTAAAGCCAGTGCA-3’; Atg3 forward, 5’-CCA TTGAAAACCATCCTCATCTC-3’, reverse, 5’-GCCTTCTGCAAC TGTCTCAATAATT-3’; Atg5 forward, 5’-GGACAGCTGCACACA CTTGG-3’, reverse, 5’-TGGCTCTATCCCGTGAATCAT-3’; Atg12 forward, 5’-TGAATCAGTCCTTTGCCCCT-3’, reverse, 5’-CATGC CTGGGATTTGCAGT-3’; Atg13 forward, 5’-TGGCGGAAGATTT GGACTCC-3’, reverse, 5’-GGGTTTCCACAAAGGCATCG-3’; and GAPDH forward, 5’-GTATTGGGCGCCTGGTCACC-3’, reverse, 5’-CGCTCCTGGAAGATGGTGATGG-3’.
The threshold cycle number (Ct) for the examined genes was normalized to the Ct value for the housekeeping gene, GAPDH. The relative mRNA level of the gene of interest was determined and expressed as the ratio of the mRNA level in the aged group mouse tissues to that in the young group mouse tissues. The fold induction values for gene expression levels were calculated using the ΔΔCt method.
ATP contents. ATP concentration in the tissues was evaluated using an ATP Assay Kit (Biovision, Milpitas, CA, USA) following the manufacturer’s protocol. The amount of ATP was measured using a BioTek ELz800 Microplate reader (BioTek, Winooski, VT, USA) and quantified based on the amount of light produced (absorbance at 570 nm). ATP levels were determined based on a standard curve, calculated as μmol/g (wet weight), and expressed as a relative percentage of the control (young group).
Western blot analysis. Total protein was extracted from the whole vestibular organ tissues, except for the cochlea of the mouse inner ear. The expression of each protein was determined using western blotting. Primary polyclonal PINK1, Parkin, BNIP3 (1:1,000; Thermo Fisher Scientific, Waltham, MA, USA), COX4, LC3B (1:1,000; Cell Signaling Technology, Danvers, MA, USA), OXPHOS Rodent WB Antibody Cocktail (6 μg/ml; Thermo Fisher Scientific), and β-actin (1:4,000; Santa Cruz Biotechnology, Dallas, TX, USA) antibodies were used together with anti-mouse in sheep or anti-rabbit in donkeys secondary antibodies (1:4,000; Jackson Immuno Research, West Grove, PA, USA). The relative expression levels of all proteins were determined by densitometry and normalized to that of β-actin.
Immunostaining. The inner ears of young and aged mice were fixed in 4% paraformaldehyde overnight at 4˚C. The fixed inner ear was decalcified using a Calci-Clear Rapid (National Diagnostics, Atlanta, GA, USA) for 7 days at 4˚C. After washing with PBS, tissues were dehydrated and embedded in paraffin. Paraffin-embedded tissues were sectioned into 4 μm sections at the level of the saccule, utricle, and crista ampullaris. Representative sections were used for experiments. For immunohistochemistry and immunofluorescence, previously characterized polyclonal antibodies against the following proteins were used: LC3B (1:100; Cell Signaling Technology), LAMP1, TOM20 (1:100; Santa Cruz Biotechnology, Dallas, TX, USA), Parkin, and BNIP3 (1:100; Thermo Fisher Scientific).
Sections for immunofluorescence were immunolabelled with Alexa Fluor 488 conjugated chicken anti-rabbit or Alexa Fluor 594 conjugated chicken anti-mouse antibody (1:200; Invitrogen, Carlsbad, CA, USA). Confocal laser microscopy was performed using a Carl Zeiss LSM800 laser confocal microscope (Carl Zeiss, Oberkochen, Germany), and imaged using the Zeiss microscope image software ZEN (Carl Zeiss). The colocalization coefficient levels were quantified using ImageJ software (NIH, Bethesda, MD, USA).
Immunohistochemical sections were visualized using a biotinylated anti-rabbit IgG and avidin-biotin-peroxidase (ABC) detection system (Vector Laboratories, Burlingame, CA, USA). Novocastra Hematoxylin (Leica Biosystems, Nussloch, Deutschland) was used to visualize the cell nucleus. The immunoreactivity of Parkin and BNIP3 was calculated by scoring the staining intensity (0, none; 1, weak; 2, moderate; 3, strong) and the percentage of positive cells (0: ≤5%, 1: 6-25%, 2: 26-50%, 3: 51-75%, 4: ≥76%). The results are expressed as the product of both scores.
Statistical analysis. The results were statistically analyzed using the SPSS 25.0 software (SPSS Inc., Chicago, IL, USA). The Mann-Whitney U-test was used to analyze the data. A p-value of <0.05 was considered statistically significant.
Results
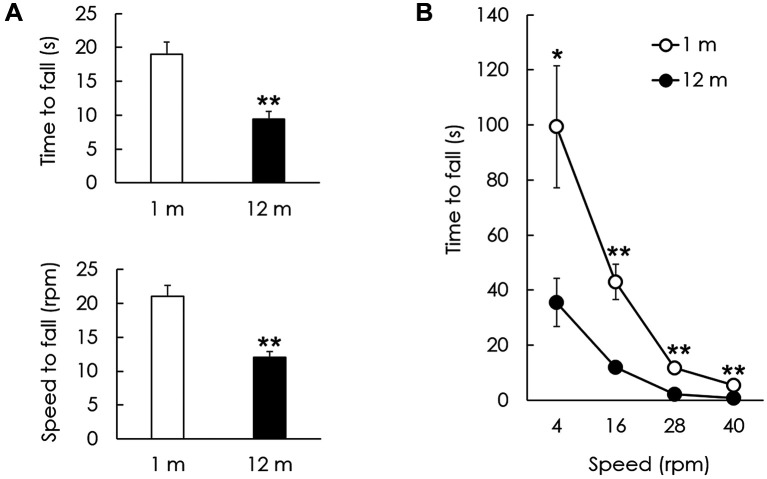
Balance and coordination decline in aged C57BL/6J mice. Balance and motor performance were assessed using a rotarod test. The latencies and speed of falling off the rod in the young (1 month) and aged (12 months) groups were compared using an accelerating rotarod. Time and speed to fall in the young group were 19.0±1.76 s and 21.0±1.67 rpm, respectively. Time and speed to fall in the aged group were 9.4±1.12 s and 12.0±0.89 rpm, respectively (p<0.01, Figure 1A). The latency to fall off the rod at a constant speed (4, 16, 28, and 40 rpm) was significantly lower in the aged group compared with that in the young group at all speeds (p<0.05, Figure 1B).
Figure 1. Locomotor performance of young and aged mice on rotarod test. (A) The latency and speed to fall on an acceleration rotarod in the aged group both significantly decreased in comparison to those of the young group. (B) The latency to fall on the rotarod at a constant speed was investigated between 4 and 40 rpm. The aged group showed a significant decrease in latency to fall at 4, 16, 28, and 40 rpm. The data are shown as the mean±SEM of five independent experimental results (1 m: 1 month; 12 m: 12 months; *p<0.05; **p<0.01).
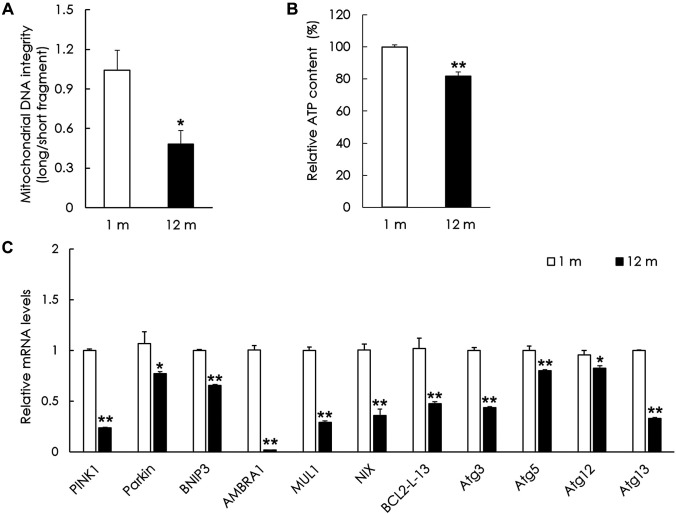
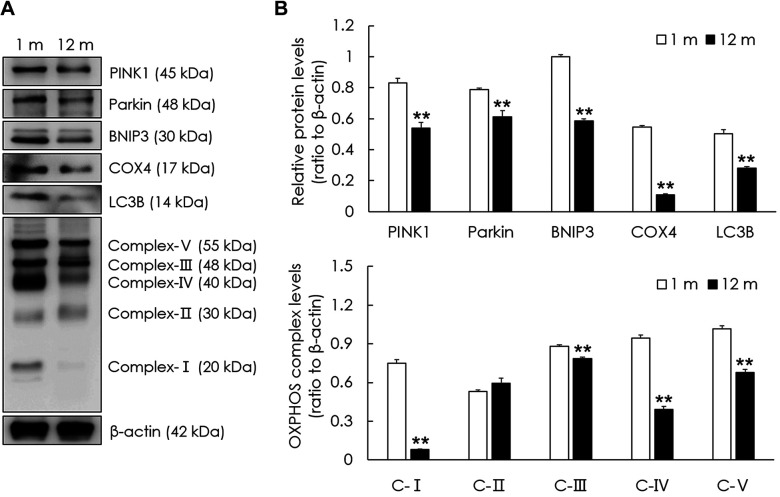
Aging results in increased mitochondrial damage and decrease in the expression of mitophagy-related genes and proteins in the peripheral vestibular system. To examine mitochondrial damage in the aged group, mitochondrial DNA integrity was investigated relative to the young group. Mitochondrial DNA integrity in the peripheral vestibular system of the aged group was significantly lower than that of the young group (p<0.05, Figure 2A). To examine mitochondrial function, the ATP content was investigated in both groups. The ATP content in the peripheral vestibular system of the aged group was significantly lower than that of the young group (p<0.01, Figure 2B). To examine the changes in mitophagy in the aged vestibular system, representative mitophagy genes were investigated and compared with those in the young group. The mRNA levels of PINK1, Parkin, BNIP3, AMBRA1, MUL1, NIX, Bcl2-L-13, Atg3, Atg5, Atg12, and Atg13 were all significantly decreased in the peripheral vestibular system of the aged group compared with those in the young group (p<0.05, Figure 2C). Representative mitophagy proteins were investigated in both groups. The protein levels of PINK1, Parkin, BNIP3, and LC3B were significantly decreased in the peripheral vestibular system of the aged group compared with those of the young group (p<0.01, Figure 3). The mitochondrial oxidative phosphorylation (OXPHOS) complex was examined to investigate changes in mitochondrial function in the aged vestibular system. The OXPHOS subunits (complexes I, III, IV, and V) were significantly decreased in the peripheral vestibular system of the aged group compared with those of the young group (p<0.01, Figure 3). These results indicate that mitophagy-related genes and proteins and mitochondrial function decreased in the aged vestibular system.
Figure 2. Changes in mitochondrial DNA integrity, ATP content, and the expression of mitophagy-related genes in the aged vestibular system. (A) Mitochondrial DNA integrity in the peripheral vestibular system of mice in the aged group was significantly lower in comparison to that of the young group. (B) ATP content in the peripheral vestibular system of aged mice was significantly lower than that of young mice. (C) Relative mRNA levels of PINK1, Parkin, BNIP3, AMBRA1, MUL1, NIX, BCL2-L-13, Atg3, Atg5, Atg12, and Atg13 were all significantly decreased in the peripheral vestibular system of mice in the aged group compared with those in the young group. The data are shown as the mean±SEM of five independent experimental results (1 m: 1 month; 12 m: 12 months; *p<0.05; **p<0.01).
Figure 3. Changes in the expression of mitophagy-related and oxidative phosphorylation (OXPHOS) proteins in the aged vestibular system. (A) Western blot analysis of PINK1, Parkin, BNIP3, COX4, LC3B, and OXPHOS subunits. β-actin was used as a loading control. (B) Relative protein levels of PINK1, Parkin, BNIP3, COX4, LC3B, and OXPHOS (I, III, IV, and V) all significantly decreased in the peripheral vestibular system of mice in the aged group compared with those in the young group. The data are shown as the mean±SEM of five independent experimental results (1 m: 1 month; 12 m: 12 months; **p<0.01).
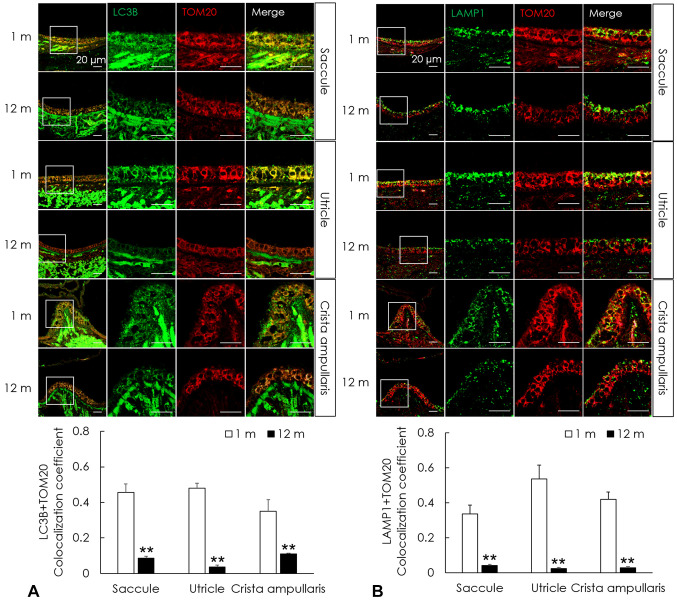
Mitophagy decreases in the peripheral vestibular system of aged C57BL/6J mice. To examine changes in the intracellular degradation process of mitophagy, immunofluorescence analysis of mitophagosomes and mitophagolysosomes was performed. Mitophagosomes in the saccule, utricle, and crista ampullaris were investigated using a reliable marker of autophagy, LC3B, and translocase of the outer membrane 20 (TOM20), a major mitochondrial receptor. Immunofluorescence analysis of LC3B and TOM20 showed that their expression was significantly decreased in the saccule, utricle, and crista ampullaris of the aged group compared with those in the young group (p<0.01, Figure 4A). Mitophagolysosomes in the saccule, utricle, and crista ampullaris were investigated using the lysosome markers LAMP1 and TOM20. Immunofluorescence analysis of LAMP1 and TOM20 showed significantly decreased expression in the saccule, utricle, and crista ampullaris in the aged group than in the young group (p<0.01, Figure 4B). These results indicate that mitophagy is decreased in the aged peripheral vestibular system.
Figure 4. Impaired mitophagy in the aged vestibular organs of C57BL/6J mice. (A) Autophagosomes and mitochondria were investigated using immunofluorescence for LC3B (green) and TOM20 (red), respectively. Mitophagosomes were analyzed by their colocalization (yellow, the overlap color) and the colocalization significantly decreased in the saccule, utricle, and crista ampullaris of mice in the aged group than in those of the young mice. (B) The fusion of lysosomes and mitophagosomes was investigated using immunofluorescence for LAMP1 (green) and TOM20 (red), respectively. Mitophagolysosomes were analyzed by their colocalization (yellow, the overlap color) and the colocalization significantly decreased in the saccule, utricle, and crista ampullaris of mice in the aged group than in those of the young group. The data are shown as the mean±SEM of five independent experimental results (1 m: 1 month; 12 m: 12 months; **p<0.01).
Parkin and BNIP3 decrease in the peripheral vestibular system of aged C57BL/6J mice. To examine the major mitophagy pathways, the expression levels of Parkin and BNIP3 were investigated using immunohistochemical staining. Immunohistochemistry scores of Parkin in the saccule, utricle, and crista ampullaris were 8.0±1.67, 10.8±1.2, and 8.2±1.11, respectively, in the young group; however, the scores significantly decreased to 2.0±0.84, 2.4±0.75, and 3.6±0.81, respectively, in the aged group. Immunohistochemistry scores of BNIP3 in the saccule, utricle, and crista ampullaris were 10.6±0.87, 6.6±0.87, and 4.2±0.49, respectively, in the young group; however, the scores significantly decreased to 1.6±0.757, 0.8±0.49, and 1.2±0.37, respectively, in the aged group (p<0.05, Figure 5). These results indicate that the expression of Parkin and BNIP3 decreased in the aged peripheral vestibular system.
Figure 5. Decreased Parkin and BNIP3 in the aged vestibular organs of C57BL/6J mice. Immunohistochemistry (IHC) scores of Parkin and BNIP3 in the saccule, utricle, and crista ampullaris of mice in the aged group were significantly lower than those of the young group. The data are shown as the mean±SEM of five independent experimental results (1 m: 1 month; 12 m: 12 months; *p<0.05; **p<0.01).
Discussion
Age-related degeneration has been demonstrated in the peripheral vestibular system; however, the underlying molecular mechanisms are poorly understood (10). Mitochondria supply cellular energy through the production of ATP and play a crucial role in maintaining cellular fitness and function. However, aging results in the impairment of mitochondrial homeostasis in the sensory system (11). Mitochondrial DNA (mtDNA) deletions and dysfunction have been reported in age-related hearing loss and in the cochlea during aging (12). The vestibular neuroepithelium requires more energy and ATP production from OXPHOS because the information for balance is delivered through exceptionally fast neurotransmission at the vestibular hair cell synapses (13,14). Since mtDNA encodes the mitochondrial respiratory chain, OXPHOS capacity decreases due to aging-induced mitochondrial damage (15). Therefore, the accumulation of damaged mitochondria results in lower ATP production and higher concentrations of reactive oxygen species, which can trigger cell death (16). The present study revealed significant losses in the integrity of mtDNA, OXPHOS subunits, and ATP production in peripheral vestibular structures with aging.
Constant turnover of damaged mitochondria is important for maintaining a healthy mitochondrial population. Mitophagy is an autophagic process for the selective removal of mitochondria and is a key mechanism in the clearance of damaged and dysfunctional mitochondria in neurons (17,18). The present study showed the down-regulation of mitophagy-related genes and proteins in aged vestibular organs: the saccule, utricle, and crista ampullaris. Mitophagy involves the sequestration of mitochondria through the formation of mitophagosomes, double-membraned phagophores, and the fusion with lysosomes which leads to degradation of mitochondrial material (19). The present study showed a decrease in the formation of mitophagosomes and mitophagolysosomes in the aged vestibule, indicating the down-regulation of mitophagy with aging. The mechanisms of mitophagy can be divided into two categories: ubiquitin-dependent and ubiquitin-independent pathways (20). The PINK1/Parkin pathway is the most widely studied ubiquitin-dependent pathway in mammals (21,22). Mitochondrial damage decreases mitochondrial membrane potential and leads to the accumulation of PINK1 in the outer membrane of the mitochondria. Accumulation of PINK1 activates the E3-ubiquitin ligase Parkin, which promotes the selective degradation of damaged mitochondria (23). BNIP3 and NIX are the most studied mitophagy receptors localized to the mitochondrial outer membrane and are involved in ubiquitin-independent pathways (24). BNIP3 and NIX facilitate the programmed clearance of damaged mitochondria by triggering mitochondrial depolarization or through direct interaction with LC3 (25,26). The present study found a reduction in two major mitophagy pathways in the aged vestibule, which can hinder the elimination of dysfunctional mitochondria and mitochondrial homeostasis.
The general machinery for mitophagy involves Atg proteins, which regulate mitophagosome formation by forming multi-molecular complexes (27). Atg13 initiates autophagy and Atg3 converts LC3-I to LC3-II (28,29). The Atg5-Atg12 complex facilitates LC3 lipidation (30). Our results showed a general decrease in Atg mRNA levels in the aged vestibules.
Increasing evidence supports the role of mitophagy in the progression of aging and neurodegenerative diseases, such as Alzheimer’s, Parkinson’s, and Huntington’s disease (31-33). The induction of mitophagy can effectively prevent the accumulation of dysfunctional mitochondria and mitigate the production of reactive oxygen species from damaged mitochondria, both of which lead to cell death during aging (34,35). Up-regulation of the PINK1/Parkin pathway showed a neuroprotective effect in a model of Parkinson’s disease (36) and attenuated cardiomyocyte senescence (37). Parkin overexpression alleviated behavioral abnormalities in a mouse model of Alzheimer’s disease (38).
Taken together, our results showed a general decrease in mitophagy in the aged vestibule. Enhancing mitophagy in the vestibular system may be a novel approach to prevent age-related balance problems.
Conclusion
This study revealed that mitophagy in the peripheral vestibular system is impaired with aging, suggesting that reduced mitophagy with aging may be a major etiology of cellular dysfunction in the aged vestibule. Up-regulation of mitophagy may be a preventive strategy for patients with age-related balance problems.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2022R1A2C1005091).
Conflicts of Interest
The Authors have no conflicts of interest to declare in relation to this study.
Authors’ Contributions
Conceptualization: SIC, ERJ; Data curation: ERJ, SL; Formal analysis: ERJ, SL; Funding acquisition: SIC; Investigation: ERJ, SL; Methodology: SIC, ERJ; Project administration: ERJ; Resources: SIC; Software: ERJ; Supervision: SIC; Validation: ERJ, SL; Visualization: ERJ, SL; Roles/Writing - original draft: SIC; Writing - review & editing: SIC. All Authors reviewed and approved the final manuscript.
References
- 1.Holstein GR. The vestibular system. In: The Human Nervous System. Amsterdam, the Netherlands, Elsevier. 2012:pp. 1239–1269. [Google Scholar]
- 2.Ishiyama G. Imbalance and vertigo: the aging human vestibular periphery. Semin Neurol. 2009;29(05):491–499. doi: 10.1055/s-0029-1241039. [DOI] [PubMed] [Google Scholar]
- 3.Rappe A, McWilliams TG. Mitophagy in the aging nervous system. Front Cell Dev Biol. 2022;10:978142. doi: 10.3389/fcell.2022.978142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Spinelli JB, Haigis MC. The multifaceted contributions of mitochondria to cellular metabolism. Nat Cell Biol. 2018;20(7):745–754. doi: 10.1038/s41556-018-0124-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Linnane AW, Ozawa T, Marzuki S, Tanaka M. Mitochondrial DNA mutations as an important contributor to ageing and degenerative diseases. Lancet. 1989;333(8639):642–645. doi: 10.1016/s0140-6736(89)92145-4. [DOI] [PubMed] [Google Scholar]
- 6.Cuervo AM, Macian F. Autophagy and the immune function in aging. Curr Opin Immunol. 2014;29:97–104. doi: 10.1016/j.coi.2014.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fivenson EM, Lautrup S, Sun N, Scheibye-Knudsen M, Stevnsner T, Nilsen H, Bohr VA, Fang EF. Mitophagy in neurodegeneration and aging. Neurochem Int. 2017;109:202–209. doi: 10.1016/j.neuint.2017.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Collier JJ, Suomi F, Oláhová M, McWilliams TG, Taylor RW. Emerging roles of ATG7 in human health and disease. EMBO Mol Med. 2021;13(12):e14824. doi: 10.15252/emmm.202114824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Palikaras K, Lionaki E, Tavernarakis N. Coordination of mitophagy and mitochondrial biogenesis during ageing in C. elegans. Nature. 2015;521(7553):525–528. doi: 10.1038/nature14300. [DOI] [PubMed] [Google Scholar]
- 10.Brosel S, Laub C, Averdam A, Bender A, Elstner M. Molecular aging of the mammalian vestibular system. Ageing Res Rev. 2016;26:72–80. doi: 10.1016/j.arr.2015.12.007. [DOI] [PubMed] [Google Scholar]
- 11.Palikaras K, Tavernarakis N. Mitophagy in neurodegeneration and aging. Front Genet. 2012;3:297. doi: 10.3389/fgene.2012.00297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Keithley EM. Pathology and mechanisms of cochlear aging. J Neurosci Res. 2020;98(9):1674–1684. doi: 10.1002/jnr.24439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McCue MP, Guinan JJ Jr. Spontaneous activity and frequency selectivity of acoustically responsive vestibular afferents in the cat. J Neurophysiol. 1995;74(4):1563–1572. doi: 10.1152/jn.1995.74.4.1563. [DOI] [PubMed] [Google Scholar]
- 14.Eatock RA. Specializations for fast signaling in the amniote vestibular inner ear. Integr Comp Biol. 2018;58(2):341–350. doi: 10.1093/icb/icy069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bigland MJ, Brichta AM, Smith DW. Effects of ageing on the mitochondrial genome in rat vestibular organs. Curr Aging Sci. 2018;11(2):108–117. doi: 10.2174/1874609811666180830143358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rugarli EI, Langer T. Mitochondrial quality control: a matter of life and death for neurons. EMBO J. 2012;31(6):1336–1349. doi: 10.1038/emboj.2012.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ding WX, Yin XM. Mitophagy: mechanisms, pathophysiological roles, and analysis. Biol Chem. 2012;393(7):547–564. doi: 10.1515/hsz-2012-0119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kobro-Flatmoen A, Lagartos-Donate MJ, Aman Y, Edison P, Witter MP, Fang EF. Re-emphasizing early Alzheimer’s disease pathology starting in select entorhinal neurons, with a special focus on mitophagy. Ageing Res Rev. 2021;67:101307. doi: 10.1016/j.arr.2021.101307. [DOI] [PubMed] [Google Scholar]
- 19.Ganley IG, Simonsen A. Diversity of mitophagy pathways at a glance. J Cell Sci. 2022;135(23):jcs259748. doi: 10.1242/jcs.259748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lu Y, Li Z, Zhang S, Zhang T, Liu Y, Zhang L. Cellular mitophagy: Mechanism, roles in diseases and small molecule pharmacological regulation. Theranostics. 2023;13(2):736–766. doi: 10.7150/thno.79876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ashrafi G, Schwarz TL. The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ. 2013;20(1):31–42. doi: 10.1038/cdd.2012.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pickrell AM, Youle RJ. The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron. 2015;85(2):257–273. doi: 10.1016/j.neuron.2014.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lazarou M, Sliter DA, Kane LA, Sarraf SA, Wang C, Burman JL, Sideris DP, Fogel AI, Youle RJ. The ubiquitin kinase PINK1 recruits autophagy receptors to induce mitophagy. Nature. 2015;524(7565):309–314. doi: 10.1038/nature14893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang Q, Xue H, Yue Y, Hao S, Huang SH, Zhang Z. Role of mitophagy in the neurodegenerative diseases and its pharmacological advances: A review. Front Mol Neurosci. 2022;15:1014251. doi: 10.3389/fnmol.2022.1014251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ney PA. Mitochondrial autophagy: Origins, significance, and role of BNIP3 and NIX. Biochim Biophys Acta 1853(10 Pt. 2015;B):2775–2783. doi: 10.1016/j.bbamcr.2015.02.022. [DOI] [PubMed] [Google Scholar]
- 26.Novak I. Mitophagy: a complex mechanism of mitochondrial removal. Antioxid Redox Signal. 2012;17(5):794–802. doi: 10.1089/ars.2011.4407. [DOI] [PubMed] [Google Scholar]
- 27.Mizushima N, Komatsu M. Autophagy: Renovation of cells and tissues. Cell. 2011;147(4):728–741. doi: 10.1016/j.cell.2011.10.026. [DOI] [PubMed] [Google Scholar]
- 28.Mizushima N. The role of the Atg1/ULK1 complex in autophagy regulation. Curr Opin Cell Biol. 2010;22(2):132–139. doi: 10.1016/j.ceb.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 29.Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 2000;19(21):5720–5728. doi: 10.1093/emboj/19.21.5720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hanada T, Noda NN, Satomi Y, Ichimura Y, Fujioka Y, Takao T, Inagaki F, Ohsumi Y. The Atg12-Atg5 conjugate has a novel E3-like activity for protein lipidation in autophagy. J Biol Chem. 2007;282(52):37298–37302. doi: 10.1074/jbc.C700195200. [DOI] [PubMed] [Google Scholar]
- 31.Chen H, Chan DC. Mitochondrial dynamics—fusion, fission, movement, and mitophagy—in neurodegenerative diseases. Hum Mol Genet. 2009;18(R2):R169–R176. doi: 10.1093/hmg/ddp326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Burté F, Carelli V, Chinnery PF, Yu-Wai-Man P. Disturbed mitochondrial dynamics and neurodegenerative disorders. Nat Rev Neurol. 2015;11(1):11–24. doi: 10.1038/nrneurol.2014.228. [DOI] [PubMed] [Google Scholar]
- 33.Martinez-Vicente M, Talloczy Z, Wong E, Tang G, Koga H, Kaushik S, de Vries R, Arias E, Harris S, Sulzer D, Cuervo AM. Cargo recognition failure is responsible for inefficient autophagy in Huntington’s disease. Nat Neurosci. 2010;13(5):567–576. doi: 10.1038/nn.2528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang H, Bosch-Marce M, Shimoda LA, Tan YS, Baek JH, Wesley JB, Gonzalez FJ, Semenza GL. Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J Biol Chem. 2008;283(16):10892–10903. doi: 10.1074/jbc.M800102200. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 35.Ryu D, Mouchiroud L, Andreux PA, Katsyuba E, Moullan N, Nicolet-Dit-Félix AA, Williams EG, Jha P, Lo Sasso G, Huzard D, Aebischer P, Sandi C, Rinsch C, Auwerx J. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat Med. 2016;22(8):879–888. doi: 10.1038/nm.4132. [DOI] [PubMed] [Google Scholar]
- 36.Li R, Chen J. Salidroside protects dopaminergic neurons by enhancing PINK1/parkin-mediated mitophagy. Oxid Med Cell Longev. 2019;2019:9341018. doi: 10.1155/2019/9341018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Soh JEC, Shimizu A, Molla MR, Zankov DP, Nguyen LKC, Khan MR, Tesega WW, Chen S, Tojo M, Ito Y, Sato A, Hitosugi M, Miyagawa S, Ogita H. RhoA rescues cardiac senescence by regulating Parkin-mediated mitophagy. J Biol Chem. 2023;299(3):102993. doi: 10.1016/j.jbc.2023.102993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hong X, Liu J, Zhu G, Zhuang Y, Suo H, Wang P, Huang D, Xu J, Huang Y, Yu M, Bian M, Sheng Z, Fei J, Song H, Behnisch T, Huang F. Parkin overexpression ameliorates hippocampal long-term potentiation and -amyloid load in an Alzheimer’s disease mouse model. Hum Mol Genet. 2014;23(4):1056–1072. doi: 10.1093/hmg/ddt501. [DOI] [PubMed] [Google Scholar]