Abstract
Infectious bronchitis virus, otherwise known as coronavirus, can cause mild upper respiratory tract illnesses in children and adults. Rarely has coronavirus been linked, either by serology or nasal wash, to pneumonia. We report a case of a young woman who, following treatment for stage IIIA breast cancer using a high-dose chemotherapy regimen followed by autologous bone marrow and stem cell transplantation, developed respiratory failure and was found to have coronavirus pneumonia as diagnosed by electron microscopy from BAL fluid. We propose that coronavirus should be considered in the differential diagnosis of acute respiratory failure in cancer patients who have undergone high-dose chemotherapy and autologous hematopoietic support.
Key words: bone marrow transplantation, breast cancer, coronavirus pneumonia, high-dose chemotherapy, idiopathic pneumonia syndrome
Abbreviations: BCNU, carmusitne; BMT, bonemarrow transplant; CAF, cyclophosphamide, doxorubicin, fluorouracil; CMV, cytomegalovirus; Dlco, diffusion of carbonmonoxide; DPTS, delayed pulmonary toxicity syndrome; EM, electronmicroscopy; HDC/ABMT, high-dose chemotherapy autologous bone marrowtransplant; IPS, idiopathic pneumonia syndrome; RSV, respiratorysyncytial virus
Interstitial pneumonitis is a frequent complication following high-dose chemotherapy and autologous bone marrow transplantation (HDC/ABMT).1 ,2 Infectious etiologies have been identified in about 50% of cases and include cytomegalovirus (CMV), herpes simplex virus, respiratory syncytical virus (RSV), Pneumocystis carinii, adenovirus, Aspergillus, and Candida.1 ,2 In the remaining cases in which no infectious agents could be identified, several distinct syndromes have been described. Idiopathic pneumonia syndrome (IPS) is the most severe form of posttransplant interstitial pneumonitis and is best characterized by severe hypoxemia, radiographic infiltrates, and is associated with high mortality.3 ,4 ,5However, we have recently described in detail a milder form of posttransplant pneumonitis occurring in autologous bone marrow transplant (BMT) patients that we have termed delayed pulmonary toxicity syndrome (DPTS).6 DPTS is characterized by its high incidence (69%), prompt response to steroids with resolution of pulmonary symptoms and improvement in diffusion of carbon monoxide (Dlco), and very low mortality.6 ,7Radiographic abnormalities, when present, typically appear as scattered, predominantly peripheral ill-defined opacities.8
Possible mechanisms for the pathogenesis of either IPS or DPTS include the following: (1) pulmonary drug toxic reactions caused by carmustine (BCNU) and/or cyclophosphamide;3 ,9 (2) imbalance of lung cytokines that promote a proinflammatory state;10 ,11 (3) occult latent viral infections (eg, CMV, RSV, Epstein-Barr virus, or human herpes 6 virus);12 ,13 or (4) an imbalance in pulmonary oxidants and antioxidants.14 Allogeneic transplant patients, in contrast to autologous, are also susceptible to graft-vs-host disease processes and in addition, are maintained in an immunodeficiency state for an extended time.
The coronaviruses are one of the more recently identified major family of viruses that have been associated with the development of the common cold. They are a family of positive-sense single-stranded RNA viruses with distinctive club-shape projections imparting a crown or corona-shaped appearance. OC43 and 229E are two major serologic coronavirus strains identified to be important in human disease.15 ,16 ,17 Only indirectly has coronavirus been linked to lower respiratory tract infections.18 ,19 ,20
In this report, we document the development of acute respiratory failure following BMT that was atypical of either IPS or DPTS and that, we believe, was caused or exacerbated by coronavirus pneumonia. To our knowledge, this represents the first reported case of coronavirus isolated from lower respiratory tract specimens in an adult with pneumonia.
Case Report
A 35-year-old Jamaican woman, 155 days post HDC/ABMT for stage IIIA breast carcinoma, was admitted to Duke University Hospital with complaints of fatigue, anorexia, and weight loss and for further workup of hyperbilirubinemia.
The patient had been well until 13 months prior to hospital admission when a left breast mass was noted on self-examination. An incisional biopsy specimen with subsequent modified radical mastectomy revealed an infiltrating ductal carcinoma with 14/15 lymph nodes positive for metastatic tumor. A staging chest/abdomen/pelvis/head CT and bone scan revealed no metastatic disease. Results of initial laboratory studies, including liver function tests, were normal. Baseline pulmonary function testing demonstrated an FEV1 of 2.27 L/s (76%), FVC of 2.79 L (80%), total lung capacity of 3.46 L (71%), and a hemoglobin-corrected Dlco of 28.5 (mL/min/mm Hg) (133%).
The patient was enrolled on Cancer and Leukemia Group B protocol 9082. She received four cycles of CAF (cyclophosphamide, 600 mg/m2 on day 1, doxorubicin, 60 mg/m2 on day 1, and fluorouracil, 600 mg/m2 on days 1 and 8) given every 28 days (Fig 1 ) that was followed by high-dose CPA/CDDP/BCNU (cyclophosphamide [1,875 mg/m2], given IV over 1 h each of three consecutive days (day −6, −5, −4), cisplatin [55 mg/m2] given by continuous infusion over 24 h each day for days −6, −5, and −4, and BCNU [600 mg/m2] infused over 2 h on day −3). The total plasma area under the curve for cyclophosphamide was 93,280 μg/mL × min. Cisplatin levels were determined at the end of each 24-h dose and were 112.4, 62.8, and 121.6 ng/mL. The BCNU plasma area under the curve was 607.5 μg/mL × min. Two days following high-dose chemotherapy, peripheral blood progenitor cells were reinfused (days −1, 0, +1) and autologous bone marrow (0.7 ± 0.25 × 108 cells per kilogram of body weight) was reinfused on day +1 to provide cellular support. Engraftment was uneventful and occurred by 2 weeks.
Figure 1.
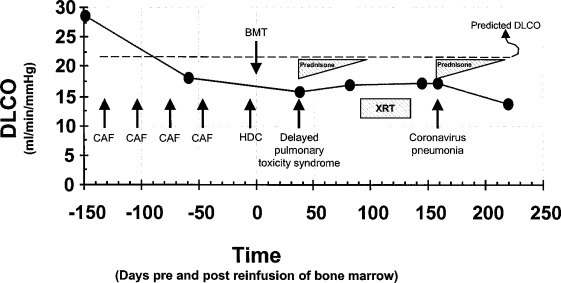
Chronologic series of events leading up to the coronavirus pneumonia. At the time points indicated (circles), the absolute Dlco, corrected for hemoglobin concentration, was measured.31 The patient's predicted Dlco is shown by a dashed line. The four cycles of adjuvant chemotherapy are indicated by CAF and occurred at days −132, −103, −75, and −47. XRT = external radiation therapy; HDC = high-dose chemotherapy.
Thirty-eight days post-BMT, the patient developed over the course of 2 days, a nonproductive cough and dyspnea. An arterial blood gas determination performed on room air showed a pH of 7.45, Pco 2 of 36 mm Hg, Po 2of 89 mm Hg, and O2 saturation of 98%. Pulmonary function testing at this time showed that her Dlco had fallen 54% from her baseline level (Fig 1). Based on these findings, she was diagnosed as having DPTS and was started on a regimen of oral prednisone at 60 mg/d. In addition, prophylactic oral trimethoprim/sulfamethoxazole was prescribed. She responded to prednisone treatment with prompt resolution of her pulmonary symptoms and a modest improvement of 11% in her Dlco.
At 83 days post-BMT, routine blood testing demonstrated mildly elevated results of liver function tests. At 91 days, her chest wall and regional node radiation therapy was begun and continued over 6 weeks for a total dose of 60.4 Gy.
At 157 days post-BMT, the patient was admitted to the hospital for workup of persistent hyperbilirubinemia. Percutaneous liver biopsy specimens demonstrated severe, centrilobular cholestasis with plugs in canaliculi consistent with centrilobular canalicular and hepatocellular cholestasis. This was believed to be consistent with an idiosyncratic drug reaction. Later that evening, she developed a brief episode of hypotension and complained of right shoulder pain. Abdominal CT demonstrated a small right subcapsular hematoma at the site of the liver biopsy. On hospital day 3, the patient spiked a fever with a temperature of 38.9°C. Blood and urine cultures were sent to the laboratory and the patient was started on a regimen of oral clarithromycin. At about this time, she complained of a sore throat and a new cough, slightly productive of sputum. A nasal swab was negative for RSV, parainfluenza A, B, and C, adenovirus, and influenza A and B. Results of pulmonary function testing were unchanged from 13 days previously (Fig 1).
On hospital day 4, the patient was noted to be tachypneic with a respiratory rate in the 40s. An arterial blood gas determination obtained on room air showed a pH of 7.46, Pco 2of 25, Po 2 of 36, and O2 saturation of 70%. An echocardiogram demonstrated normal ventricular function. Despite a 3-L diuresis, the patient's respiratory status continued to deteriorate and she was electively intubated. Because of concerns of recurrent DPTS, the patient was started on a regimen of high-dose methylprednisolone at 60 mg IV every 6 h. Erythromycin, sulfamethoxazole and trimethoprim (Bactrim), and imipenem were added empirically. A portable chest radiograph and chest CT scan demonstrated bilateral, predominantly lower lobe air space opacities (Fig 2 ).
Figure 2.
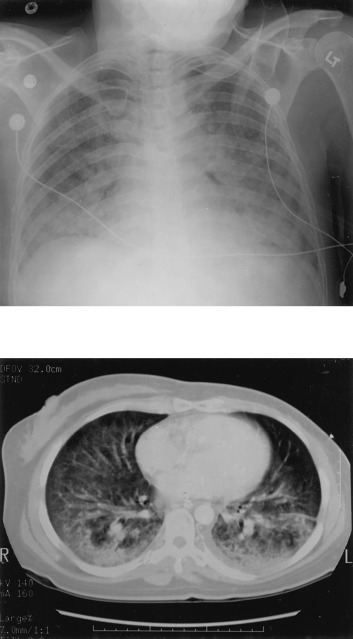
Top: a portable chest radiograph was performed on hospital day 4, demonstrating new, bilateral airspace opacities. Bottom: a chest CT radiograph on hospital day 5, demonstrating bilateral airspace disease and small left pleural effusion.
On hospital day 6, the patient underwent bronchoscopy and a BAL of both lower lobes was obtained via the endotracheal tube. Microbiological examination demonstrated the BAL sample to be negative for fungus, acid-fast bacilli, Gram stain and culture, cytology with special stains, respiratory viral battery (RSV, parainfluenza A, B, and C, adenovirus, and influenza A and B), CMV, and herpes simplex virus culture. Cytologic analysis showed rare atypical cells on a background of highly reactive pneumocytes. Electron microscopy (EM) of the BAL fluid demonstrated numerous viral particles with features that were diagnostic of coronavirus (Fig 3 ).
Figure 3.
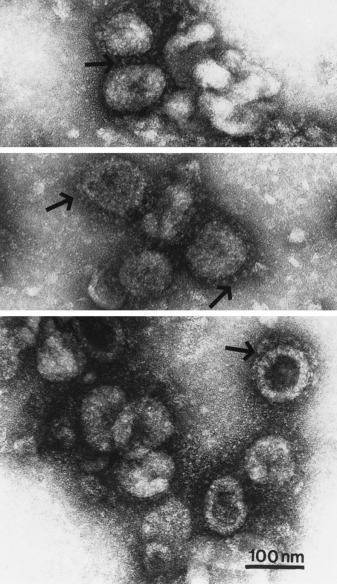
EMs obtained from bilateral lower lobe bronchoalveolar fluid revealing numerous pleomorphic (but mostly spherical) virions. Club-shaped surface projections that are distinctive for coronavirus, can be seen (arrows). Bar = 100 nm.
On hospital day 7, treatment with all antibiotics was stopped and the patient had completely defervesced. On hospital day 8, the patient was successfully extubated. By hospital day 10, she was breathing room air with on oxygen saturation of 96%. Her steroids were changed to prednisone at 60 mg to be tapered over 6 to 8 weeks. On hospital day 12, she was discharged to home. A follow-up visit at post-BMT day 220 showed her Dlco to be decreased approximately 21% from her prehospitalization value (Fig 1). However, her respiratory status remained asymptomatic.
Discussion
The incidence of pulmonary toxic reactions following high doses of cyclophosphamide/cisplatin and BCNU with autologous bone marrow support in the Duke University Bone Marrow Transplant Program is approximately 39 to 69%.6 ,7 ,21 Most patients in whom infection is ruled out have a low mortality rate and a prompt response to prednisone therapy. Because of these features, we have termed this posttransplant complication DPTS.6 DPTS is to be contrasted to IPS, a syndrome characterized by severe hypoxemia, radiographic infiltrates, and high mortality.4
Our patient demonstrated three phases of pulmonary toxic reactions. Phase I is subclinical and asymptomatic and is best characterized by a decreased Dlco measurement following the first three cycles of the CAF induction chemotherapy (Fig 1). Little is known about the contribution that the conditioning regimen plays in the development of lung injury.
Phase II occurred following HDC/ABMT and was diagnosed as DPTS based on its timing, associated pulmonary symptoms, decrease in Dlco, and prompt response to corticosteroid therapy with subsequent improvement in Dlco.
Phase III occurred approximately 158 days following BMT when the patient developed acute respiratory failure. Was coronavirus pneumonia responsible for her respiratory failure? We believe so. The BAL sample was obtained 36 h after intubation and the bronchoscope was placed into the lumen of the endotracheal tube, thus minimizing possible nasal mucosa contamination. We have considered other possibilities. For example, the coronavirus could simply have been present in the BAL specimen, but was not pathogenic. In this scenario, the etiology of the respiratory failure could have been caused by several different factors, including radiation pneumonitis, drug-induced interstitial pneumonitis, or showering of the lung with inflammatory cytokines from an abdominal or septic source resulting in ARDS. Radiation pneumonitis seems less likely since the pattern of radiographic lung injury does not duplicate the radiation port,6 although case studies have shown that radiation pneumonitis can occur outside the radiation port.22 It would be unusual to see interstitial pneumonitis from the HDC/ABMT this far out after transplant and without a concomitant decrease in Dlco.6 The liver biopsy, which occurred 2 days before respiratory failure, was complicated by a stable subcapsular hematoma, a brief episode of hypotension, and fever. We cannot exclude the possibility that inflammatory cytokines, released from GI sources, showered the lung causing acute lung injury leading to respiratory failure. Also, we could not rule out bacterial pneumonia as a complicating factor since the patient was receiving antibiotics for >24 h prior to obtaining BAL fluid and this would be predicted to lower our diagnostic yield of bacteria.
Can coronavirus cause lower respiratory tract infection? Animal strains of coronavirus appear more virulent than their human counterparts and have been shown to cause pneumonia in mice and pigs.23 ,24In humans, experimental intranasal inoculation of coronavirus in volunteers results in a typical coryzal illness with nasal discharge and a notable absence of systemic complaints.25 ,26 ,27However, lower respiratory tract involvement has been suggested based on coronavirus positive nasal washes in two children with pneumonia18 and by coronavirus seroconversion in adults who had pneumonia.19 ,20 Treatment options for upper respiratory tract infection caused by coronavirus are limited, but studies using intranasal interferon α-2b28 ,29 and intranasal nedocromil30 improve symptoms.
In this report, we document coronavirus pneumonia complicating autologous BMT. Whether coronavirus pneumonia is capable by itself of causing respiratory failure or if it requires additional factors (eg, immunocompromised host) remains to be determined. Our patient's condition initially improved with institution of corticosteroids but longer-term follow-up showed some deterioration in lung function, leaving us to wonder if coronavirus causes direct cytopathologic effects in lower respiratory airway cells.
We discovered coronavirus in this patient because the BAL specimen was evaluated by EM. EM is not typically performed on all BAL specimens owing to uncertainties with specificity and sensitivity as well as cost/benefit analyses. Because idiopathic pneumonias in our patient population account for a large proportion of posttransplant complications, we have elected to use EM in evaluating our BAL samples. However, the routine use of EM should await results of clinical trials.
Although it is not routinely evaluated for, coronavirus should be considered in the differential diagnoses of respiratory failure in patients who have undergone high-dose chemotherapy and autologous BMT.
Footnotes
This work was supported by NIH grant HL55166 and the Duke Comprehensive Cancer Center. Dr. Folz is a Parker B. Francis Fellow in Pulmonary Research.
References
- 1.Weiner RS, Boymer MM, Gale RP. Interstitial pneumonitis after bone marrow transplantation. Ann Intern Med. 1986;104:168–175. doi: 10.7326/0003-4819-104-2-168. [DOI] [PubMed] [Google Scholar]
- 2.Wingard JR, Mellits ED, Sostrin MB. Interstitial pneumonitis after allogeneic bone marrow transplantation. Medicine. 1988;67:175–186. doi: 10.1097/00005792-198805000-00004. [DOI] [PubMed] [Google Scholar]
- 3.Jones RB, Matthes S, Shpall EJ. Acute lung injury following treatment with high-dose cyclophosphamide, cisplatin, and carmustine: pharmacodynamic evaluation of carmustine. J Natl Cancer Inst. 1993;85:640–647. doi: 10.1093/jnci/85.8.640. [DOI] [PubMed] [Google Scholar]
- 4.Clark JG, Hansen JA, Hertz MI. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Respir Dis. 1993;147:1601–1606. doi: 10.1164/ajrccm/147.6_Pt_1.1601. [DOI] [PubMed] [Google Scholar]
- 5.Cherniack RM, Abrams J, Kalica AR. Pulmonary disease associated with breast cancer therapy. Am J Respir Crit Care Med. 1994;150:1169–1173. doi: 10.1164/ajrccm.150.4.7921454. [DOI] [PubMed] [Google Scholar]
- 6.Wilczynski SW, Erasmus JJ, Petros WP. Delayed pulmonary toxicity syndrome following high-dose chemotherapy and bone marrow transplantation for breast cancer. Am J Respir Crit Care Med. 1998;157:565–573. doi: 10.1164/ajrccm.157.2.9705072. [DOI] [PubMed] [Google Scholar]
- 7.Wilczynski SW, Petros WP, Hussein AM. Increased incidence of pulmonary toxicity in breast cancer patients undergoing high dose chemotherapy. Am J Respir Crit Care Med. 1996;153:A273. [Google Scholar]
- 8.Patz EF, Jr, Peters WP, Goodman PC. Pulmonary drug toxicity following high-dose chemotherapy with autologous bone marrow transplantation: CT findings in 20 cases. J Thorac Imaging. 1994;9:129–134. doi: 10.1097/00005382-199421000-00017. [DOI] [PubMed] [Google Scholar]
- 9.Aronin PA, Mahaley MS, Jr, Rudnick SA. Prediction of BCNU pulmonary toxicity in patients with malignant gliomas. N Engl J Med. 1980;303:183–188. doi: 10.1056/NEJM198007243030403. [DOI] [PubMed] [Google Scholar]
- 10.Rabinowitz J, Petros WP, Stuart AR. Characterization of endogenous cytokine concentrations after high-dose chemotherapy with autologous bone marrow support. Blood. 1993;81:2452–2459. [PubMed] [Google Scholar]
- 11.Murase T, Anscher MS, Petros WP. Changes in plasma transforming growth factor beta in response to high-dose chemotherapy for stage II breast cancer: possible implications for the prevention of hepatic veno-occlusive disease and pulmonary drug toxicity. Bone Marrow Transplant. 1994;15:173–178. [PubMed] [Google Scholar]
- 12.Englund JA, Sullivan CJ, Jordan MC. Respiratory syncytial virus infection in immunocompromised adults. Ann Int Med. 1988;109:203–208. doi: 10.7326/0003-4819-109-3-203. [DOI] [PubMed] [Google Scholar]
- 13.Fouillard L, Mouthon L, Laporte JP. Sever respiratory syncytial virus pneumonia after autologous bone marrow transplantation: a report of three cases and review. Bone Marrow Transplant. 1992;9:97–100. [PubMed] [Google Scholar]
- 14.Smith AC, Boyd MR. Preferential effects of 1,3-bis(2-chloroethyl)-1-nitrosurea (BCNU) on pulmonary glutathione reductase and glutathione/glutathione disulfide ratios: possible implications for lung toxicity. J Pharmacol Exp Ther. 1984;229:658–663. [PubMed] [Google Scholar]
- 15.Kaye HS, Marsh HB, Dowdle WR. Seroepidemiologic survey of coronavirus (strain OC43) related infections in a children's population. Am J Epidemiol. 1971;94:43–49. doi: 10.1093/oxfordjournals.aje.a121293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hendley JO, Fishburne HB, Gwaltney JM. Coronavirus infections in working adults: eight year study with 229E and OC43. Am Rev Respir Dis. 1972;105:805–811. doi: 10.1164/arrd.1972.105.5.805. [DOI] [PubMed] [Google Scholar]
- 17.Hamre D, Beem M. Virologic studies of acute respiratory disease in young adults: V. Coronavirus 229E infections during 6 years of surveillance. Am J Epidemiol. 1972;96:94–106. doi: 10.1093/oxfordjournals.aje.a121445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McIntosh K, Chao RK, Krause HE. Coronavirus infection in acute lower respiratory tract disease of infants. J Infect Dis. 1974;130:502–507. doi: 10.1093/infdis/130.5.502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wenzel RP, Hendley JO, Davies JA. Coronavirus infections in military recruits. Am Rev Respir Dis. 1974;109:621–624. doi: 10.1164/arrd.1974.109.6.621. [DOI] [PubMed] [Google Scholar]
- 20.Riski H, Hovi T. Coronavirus infections of man associated with diseases other than the common cold. J Med Virol. 1980;6:259–265. doi: 10.1002/jmv.1890060309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Todd NW, Peters WP, Ost AH. Pulmonary drug toxicity in patients with primary breast cancer treated with high-dose combination chemotherapy and autologous bone marrow transplantation. Am Rev Respir Dis. 1993;147:1264–1270. doi: 10.1164/ajrccm/147.5.1264. [DOI] [PubMed] [Google Scholar]
- 22.Fulkerson WJ, McLendon RE, Prosnitz LR. Adult respiratory distress syndrome after limited thoracic radiotherapy. Cancer. 1986;57:1941–1946. doi: 10.1002/1097-0142(19860515)57:10<1941::aid-cncr2820571009>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 23.Bhatt PN, Jacoby R, Jonas AM. Respiratory infection in mice with sialodacryoadenitis virus, a coronavirus of rats. Infect Immun. 1977;18:823–827. doi: 10.1128/iai.18.3.823-827.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jabrane A, Girard C, Elazhary Y. Pathogenicity of porcine respiratory coronavirus isolated in Quebec. Can Vet J. 1994;35:86–92. [PMC free article] [PubMed] [Google Scholar]
- 25.Bradburne AF, Bynoe ML, Tyrrell DAJ. Effects of a ‘new’ human respiratory virus in volunteers. BMJ. 1967;3:767–769. doi: 10.1136/bmj.3.5568.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Reed SE. The behavior of recent isolates of human respiratory coronavirus in vitro and in volunteers: evidence of heterogeneity among 229E-related strains. J Med Virol. 1984;13:179–192. doi: 10.1002/jmv.1890130208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bende M, Barrow I, Heptonstall J. Changes in human nasal mucosa during experimental coronavirus common colds. Acta Otolaryngol (Stockh) 1989;107:262–269. doi: 10.3109/00016488909127507. [DOI] [PubMed] [Google Scholar]
- 28.Higgins PG, Phillpotts RJ, Scott GM. Intranasal interferon as protection against experimental respiratory coronavirus infection in volunteers. Antimicrob Agents Chemother. 1983;24:713–715. doi: 10.1128/aac.24.5.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Turner RB, Felton A, Kosak K. Prevention of experimental coronavirus colds with intranasal α-2b interferon. J Infect Dis. 1986;154:443–447. doi: 10.1093/infdis/154.3.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Barrow GI, Higgins PG, Al-Nakib W. The effect of intranasal nedocromil sodium on viral upper respiratory tract infections in human volunteers. Clin Exp Allergy. 1990;20:45–51. doi: 10.1111/j.1365-2222.1990.tb02774.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.American Thoracic Society Single-breath carbon monoxide diffusing capacity (transfer factor) Am J Respir Crit Care Med. 1995;152:2185–2198. doi: 10.1164/ajrccm.152.6.8520796. [DOI] [PubMed] [Google Scholar]


