Abstract
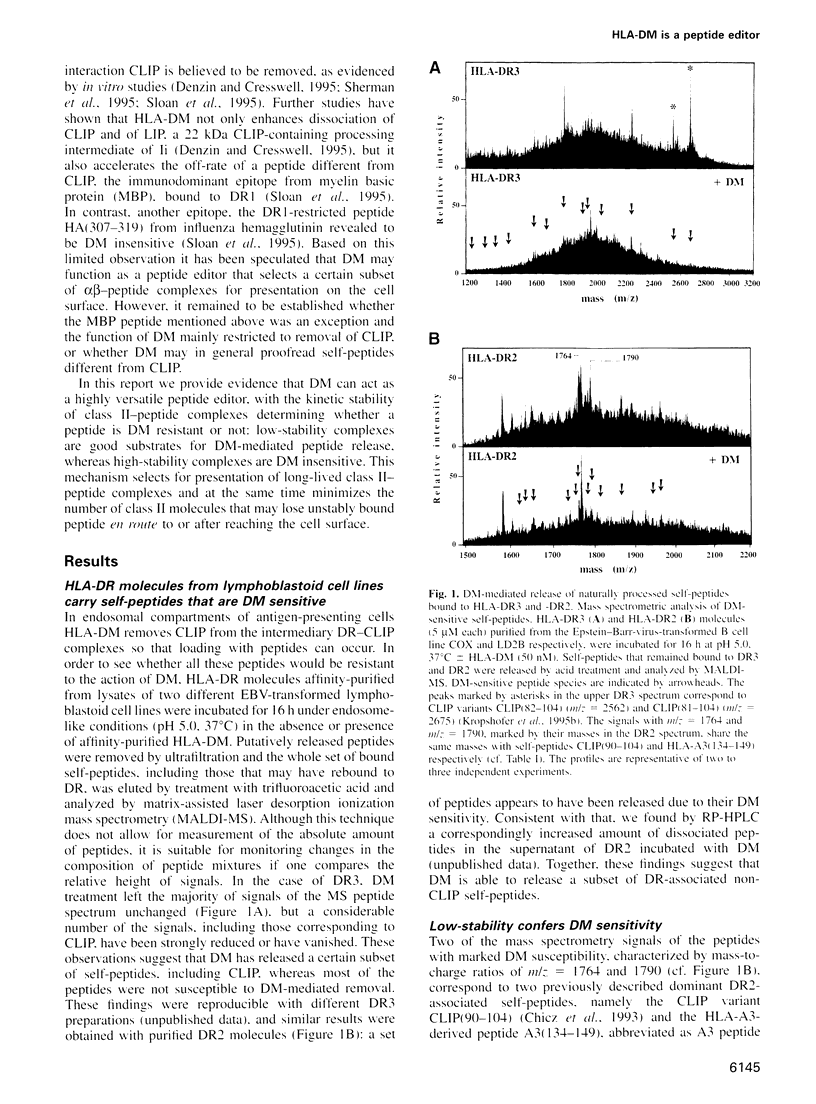
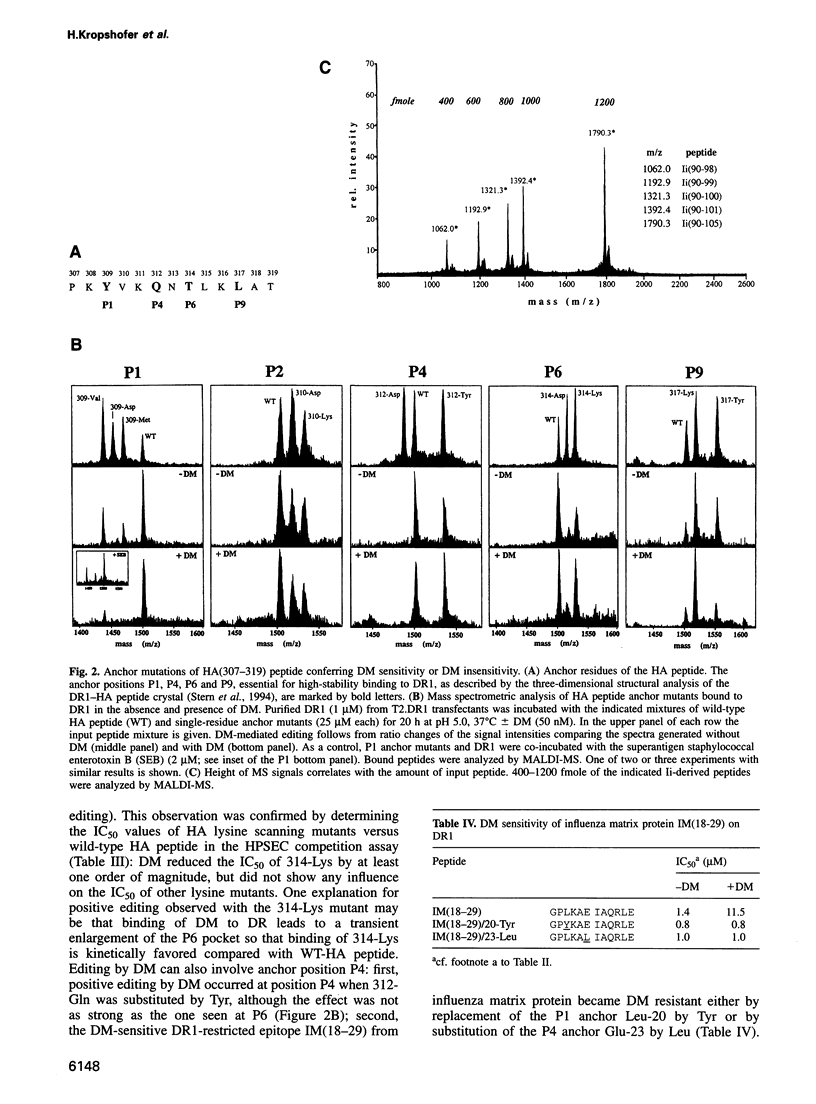
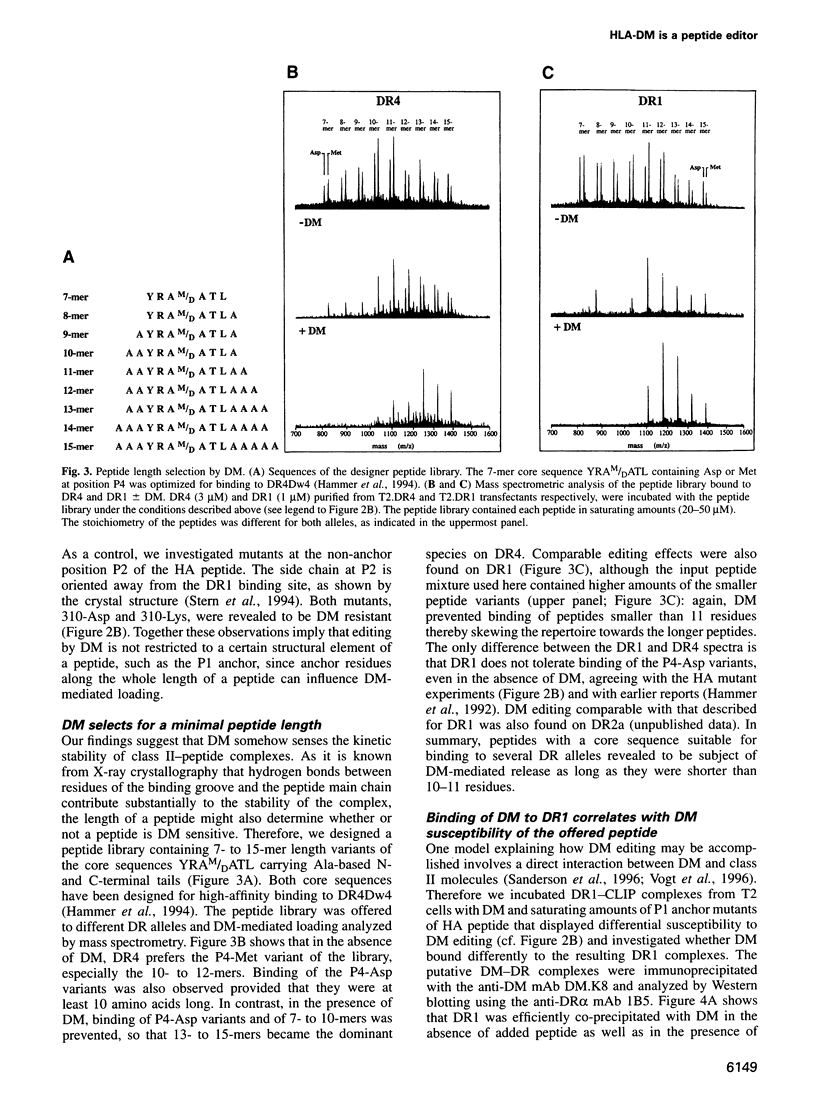
Antigenic peptide loading of classical major histocompatibility complex (MHC) class II molecules requires the exchange of the endogenous invariant chain fragment CLIP (class II associated Ii peptide) for peptides derived from antigenic proteins. This process is facilitated by the non-classical MHC class II molecule HLA-DM (DM) which catalyzes the removal of CLIP. Up to now it has been unclear whether DM releases self-peptides other than CLIP and thereby modifies the peptide repertoire presented to T cells. Here we report that DM can release a variety of peptides from HLA-DR molecules. DR molecules isolated from lymphoblastoid cell lines were found to carry a sizeable fraction of self-peptides that are sensitive to the action of DM. The structural basis for this DM sensitivity was elucidated by high-performance size exclusion chromatography and a novel mass spectrometry binding assay. The results demonstrate that the overall kinetic stability of a peptide bound to DR determines its sensitivity to removal by DM. We show that DM removes preferentially those peptides that contain at least one suboptimal side chain at one of their anchor positions or those that are shorter than 11 residues. These findings provide a rationale for the previously described ligand motifs and the minimal length requirements of naturally processed DR-associated self-peptides. Thus, in endosomal compartments, where peptide loading takes place, DM can function as a versatile peptide editor that selects for high-stability MHC class II-peptide complexes by kinetic proofreading before these complexes are presented to T cells.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams T. E., Bodmer J. G., Bodmer W. F. Production and characterization of monoclonal antibodies recognizing the alpha-chain subunits of human ia alloantigens. Immunology. 1983 Dec;50(4):613–624. [PMC free article] [PubMed] [Google Scholar]
- Avva R. R., Cresswell P. In vivo and in vitro formation and dissociation of HLA-DR complexes with invariant chain-derived peptides. Immunity. 1994 Dec;1(9):763–774. doi: 10.1016/s1074-7613(94)80018-9. [DOI] [PubMed] [Google Scholar]
- Bakke O., Dobberstein B. MHC class II-associated invariant chain contains a sorting signal for endosomal compartments. Cell. 1990 Nov 16;63(4):707–716. doi: 10.1016/0092-8674(90)90137-4. [DOI] [PubMed] [Google Scholar]
- Bijlmakers M. J., Benaroch P., Ploegh H. L. Mapping functional regions in the lumenal domain of the class II-associated invariant chain. J Exp Med. 1994 Aug 1;180(2):623–629. doi: 10.1084/jem.180.2.623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks A. G., Campbell P. L., Reynolds P., Gautam A. M., McCluskey J. Antigen presentation and assembly by mouse I-Ak class II molecules in human APC containing deleted or mutated HLA DM genes. J Immunol. 1994 Dec 15;153(12):5382–5392. [PubMed] [Google Scholar]
- Busch R., Cloutier I., Sékaly R. P., Hämmerling G. J. Invariant chain protects class II histocompatibility antigens from binding intact polypeptides in the endoplasmic reticulum. EMBO J. 1996 Jan 15;15(2):418–428. [PMC free article] [PubMed] [Google Scholar]
- Buus S., Sette A., Colon S. M., Jenis D. M., Grey H. M. Isolation and characterization of antigen-Ia complexes involved in T cell recognition. Cell. 1986 Dec 26;47(6):1071–1077. doi: 10.1016/0092-8674(86)90822-6. [DOI] [PubMed] [Google Scholar]
- Chicz R. M., Urban R. G., Gorga J. C., Vignali D. A., Lane W. S., Strominger J. L. Specificity and promiscuity among naturally processed peptides bound to HLA-DR alleles. J Exp Med. 1993 Jul 1;178(1):27–47. doi: 10.1084/jem.178.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chicz R. M., Urban R. G., Lane W. S., Gorga J. C., Stern L. J., Vignali D. A., Strominger J. L. Predominant naturally processed peptides bound to HLA-DR1 are derived from MHC-related molecules and are heterogeneous in size. Nature. 1992 Aug 27;358(6389):764–768. doi: 10.1038/358764a0. [DOI] [PubMed] [Google Scholar]
- Cho S. G., Attaya M., Monaco J. J. New class II-like genes in the murine MHC. Nature. 1991 Oct 10;353(6344):573–576. doi: 10.1038/353573a0. [DOI] [PubMed] [Google Scholar]
- Denzin L. K., Cresswell P. HLA-DM induces CLIP dissociation from MHC class II alpha beta dimers and facilitates peptide loading. Cell. 1995 Jul 14;82(1):155–165. doi: 10.1016/0092-8674(95)90061-6. [DOI] [PubMed] [Google Scholar]
- Fling S. P., Arp B., Pious D. HLA-DMA and -DMB genes are both required for MHC class II/peptide complex formation in antigen-presenting cells. Nature. 1994 Apr 7;368(6471):554–558. doi: 10.1038/368554a0. [DOI] [PubMed] [Google Scholar]
- Freisewinkel I. M., Schenck K., Koch N. The segment of invariant chain that is critical for association with major histocompatibility complex class II molecules contains the sequence of a peptide eluted from class II polypeptides. Proc Natl Acad Sci U S A. 1993 Oct 15;90(20):9703–9706. doi: 10.1073/pnas.90.20.9703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Germain R. N. MHC-dependent antigen processing and peptide presentation: providing ligands for T lymphocyte activation. Cell. 1994 Jan 28;76(2):287–299. doi: 10.1016/0092-8674(94)90336-0. [DOI] [PubMed] [Google Scholar]
- Germain R. N., Rinker A. G., Jr Peptide binding inhibits protein aggregation of invariant-chain free class II dimers and promotes surface expression of occupied molecules. Nature. 1993 Jun 24;363(6431):725–728. doi: 10.1038/363725a0. [DOI] [PubMed] [Google Scholar]
- Ghosh P., Amaya M., Mellins E., Wiley D. C. The structure of an intermediate in class II MHC maturation: CLIP bound to HLA-DR3. Nature. 1995 Nov 30;378(6556):457–462. doi: 10.1038/378457a0. [DOI] [PubMed] [Google Scholar]
- Hammer J., Belunis C., Bolin D., Papadopoulos J., Walsky R., Higelin J., Danho W., Sinigaglia F., Nagy Z. A. High-affinity binding of short peptides to major histocompatibility complex class II molecules by anchor combinations. Proc Natl Acad Sci U S A. 1994 May 10;91(10):4456–4460. doi: 10.1073/pnas.91.10.4456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammer J., Takacs B., Sinigaglia F. Identification of a motif for HLA-DR1 binding peptides using M13 display libraries. J Exp Med. 1992 Oct 1;176(4):1007–1013. doi: 10.1084/jem.176.4.1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammer J., Valsasnini P., Tolba K., Bolin D., Higelin J., Takacs B., Sinigaglia F. Promiscuous and allele-specific anchors in HLA-DR-binding peptides. Cell. 1993 Jul 16;74(1):197–203. doi: 10.1016/0092-8674(93)90306-b. [DOI] [PubMed] [Google Scholar]
- Kelly A. P., Monaco J. J., Cho S. G., Trowsdale J. A new human HLA class II-related locus, DM. Nature. 1991 Oct 10;353(6344):571–573. doi: 10.1038/353571a0. [DOI] [PubMed] [Google Scholar]
- Kropshofer H., Bohlinger I., Max H., Kalbacher H. Self and foreign peptides interact with intact and disassembled MHC class II antigen HLA-DR via tryptophan pockets. Biochemistry. 1991 Sep 24;30(38):9177–9187. doi: 10.1021/bi00102a008. [DOI] [PubMed] [Google Scholar]
- Kropshofer H., Max H., Halder T., Kalbus M., Muller C. A., Kalbacher H. Self-peptides from four HLA-DR alleles share hydrophobic anchor residues near the NH2-terminal including proline as a stop signal for trimming. J Immunol. 1993 Nov 1;151(9):4732–4742. [PubMed] [Google Scholar]
- Kropshofer H., Max H., Müller C. A., Hesse F., Stevanovic S., Jung G., Kalbacher H. Self-peptide released from class II HLA-DR1 exhibits a hydrophobic two-residue contact motif. J Exp Med. 1992 Jun 1;175(6):1799–1803. doi: 10.1084/jem.175.6.1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kropshofer H., Vogt A. B., Hämmerling G. J. Structural features of the invariant chain fragment CLIP controlling rapid release from HLA-DR molecules and inhibition of peptide binding. Proc Natl Acad Sci U S A. 1995 Aug 29;92(18):8313–8317. doi: 10.1073/pnas.92.18.8313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kropshofer H., Vogt A. B., Stern L. J., Hämmerling G. J. Self-release of CLIP in peptide loading of HLA-DR molecules. Science. 1995 Nov 24;270(5240):1357–1359. doi: 10.1126/science.270.5240.1357. [DOI] [PubMed] [Google Scholar]
- Lampson L. A., Levy R. Two populations of Ia-like molecules on a human B cell line. J Immunol. 1980 Jul;125(1):293–299. [PubMed] [Google Scholar]
- Lindstedt R., Liljedahl M., Péléraux A., Peterson P. A., Karlsson L. The MHC class II molecule H2-M is targeted to an endosomal compartment by a tyrosine-based targeting motif. Immunity. 1995 Nov;3(5):561–572. doi: 10.1016/1074-7613(95)90127-2. [DOI] [PubMed] [Google Scholar]
- Lotteau V., Teyton L., Peleraux A., Nilsson T., Karlsson L., Schmid S. L., Quaranta V., Peterson P. A. Intracellular transport of class II MHC molecules directed by invariant chain. Nature. 1990 Dec 13;348(6302):600–605. doi: 10.1038/348600a0. [DOI] [PubMed] [Google Scholar]
- Marks M. S., Roche P. A., van Donselaar E., Woodruff L., Peters P. J., Bonifacino J. S. A lysosomal targeting signal in the cytoplasmic tail of the beta chain directs HLA-DM to MHC class II compartments. J Cell Biol. 1995 Oct;131(2):351–369. doi: 10.1083/jcb.131.2.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris P., Shaman J., Attaya M., Amaya M., Goodman S., Bergman C., Monaco J. J., Mellins E. An essential role for HLA-DM in antigen presentation by class II major histocompatibility molecules. Nature. 1994 Apr 7;368(6471):551–554. doi: 10.1038/368551a0. [DOI] [PubMed] [Google Scholar]
- O'Sullivan D., Arrhenius T., Sidney J., Del Guercio M. F., Albertson M., Wall M., Oseroff C., Southwood S., Colón S. M., Gaeta F. C. On the interaction of promiscuous antigenic peptides with different DR alleles. Identification of common structural motifs. J Immunol. 1991 Oct 15;147(8):2663–2669. [PubMed] [Google Scholar]
- Rammensee H. G., Friede T., Stevanoviíc S. MHC ligands and peptide motifs: first listing. Immunogenetics. 1995;41(4):178–228. doi: 10.1007/BF00172063. [DOI] [PubMed] [Google Scholar]
- Riberdy J. M., Newcomb J. R., Surman M. J., Barbosa J. A., Cresswell P. HLA-DR molecules from an antigen-processing mutant cell line are associated with invariant chain peptides. Nature. 1992 Dec 3;360(6403):474–477. doi: 10.1038/360474a0. [DOI] [PubMed] [Google Scholar]
- Roche P. A., Cresswell P. Invariant chain association with HLA-DR molecules inhibits immunogenic peptide binding. Nature. 1990 Jun 14;345(6276):615–618. doi: 10.1038/345615a0. [DOI] [PubMed] [Google Scholar]
- Roche P. A., Cresswell P. Proteolysis of the class II-associated invariant chain generates a peptide binding site in intracellular HLA-DR molecules. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3150–3154. doi: 10.1073/pnas.88.8.3150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roche P. A., Marks M. S., Cresswell P. Formation of a nine-subunit complex by HLA class II glycoproteins and the invariant chain. Nature. 1991 Dec 5;354(6352):392–394. doi: 10.1038/354392a0. [DOI] [PubMed] [Google Scholar]
- Romagnoli P., Germain R. N. The CLIP region of invariant chain plays a critical role in regulating major histocompatibility complex class II folding, transport, and peptide occupancy. J Exp Med. 1994 Sep 1;180(3):1107–1113. doi: 10.1084/jem.180.3.1107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadegh-Nasseri S., Stern L. J., Wiley D. C., Germain R. N. MHC class II function preserved by low-affinity peptide interactions preceding stable binding. Nature. 1994 Aug 25;370(6491):647–650. doi: 10.1038/370647a0. [DOI] [PubMed] [Google Scholar]
- Sanderson F., Kleijmeer M. J., Kelly A., Verwoerd D., Tulp A., Neefjes J. J., Geuze H. J., Trowsdale J. Accumulation of HLA-DM, a regulator of antigen presentation, in MHC class II compartments. Science. 1994 Dec 2;266(5190):1566–1569. doi: 10.1126/science.7985027. [DOI] [PubMed] [Google Scholar]
- Sanderson F., Thomas C., Neefjes J., Trowsdale J. Association between HLA-DM and HLA-DR in vivo. Immunity. 1996 Jan;4(1):87–96. doi: 10.1016/s1074-7613(00)80301-5. [DOI] [PubMed] [Google Scholar]
- Sette A., Ceman S., Kubo R. T., Sakaguchi K., Appella E., Hunt D. F., Davis T. A., Michel H., Shabanowitz J., Rudersdorf R. Invariant chain peptides in most HLA-DR molecules of an antigen-processing mutant. Science. 1992 Dec 11;258(5089):1801–1804. doi: 10.1126/science.1465617. [DOI] [PubMed] [Google Scholar]
- Sette A., Southwood S., Miller J., Appella E. Binding of major histocompatibility complex class II to the invariant chain-derived peptide, CLIP, is regulated by allelic polymorphism in class II. J Exp Med. 1995 Feb 1;181(2):677–683. doi: 10.1084/jem.181.2.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherman M. A., Weber D. A., Jensen P. E. DM enhances peptide binding to class II MHC by release of invariant chain-derived peptide. Immunity. 1995 Aug;3(2):197–205. doi: 10.1016/1074-7613(95)90089-6. [DOI] [PubMed] [Google Scholar]
- Sloan V. S., Cameron P., Porter G., Gammon M., Amaya M., Mellins E., Zaller D. M. Mediation by HLA-DM of dissociation of peptides from HLA-DR. Nature. 1995 Jun 29;375(6534):802–806. doi: 10.1038/375802a0. [DOI] [PubMed] [Google Scholar]
- Stebbins C. C., Loss G. E., Jr, Elias C. G., Chervonsky A., Sant A. J. The requirement for DM in class II-restricted antigen presentation and SDS-stable dimer formation is allele and species dependent. J Exp Med. 1995 Jan 1;181(1):223–234. doi: 10.1084/jem.181.1.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern L. J., Brown J. H., Jardetzky T. S., Gorga J. C., Urban R. G., Strominger J. L., Wiley D. C. Crystal structure of the human class II MHC protein HLA-DR1 complexed with an influenza virus peptide. Nature. 1994 Mar 17;368(6468):215–221. doi: 10.1038/368215a0. [DOI] [PubMed] [Google Scholar]
- Urban R. G., Chicz R. M., Strominger J. L. Selective release of some invariant chain-derived peptides from HLA-DR1 molecules at endosomal pH. J Exp Med. 1994 Aug 1;180(2):751–755. doi: 10.1084/jem.180.2.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verreck F. A., Vermeulen C., Poel A. V., Jorritsma P., Amons R., Coligan J. E., Drijfhout J. W., Koning F. The generation of SDS-stable HLA DR dimers is independent of efficient peptide binding. Int Immunol. 1996 Mar;8(3):397–404. doi: 10.1093/intimm/8.3.397. [DOI] [PubMed] [Google Scholar]
- Vogt A. B., Kropshofer H., Kalbacher H., Kalbus M., Rammensee H. G., Coligan J. E., Martin R. Ligand motifs of HLA-DRB5*0101 and DRB1*1501 molecules delineated from self-peptides. J Immunol. 1994 Aug 15;153(4):1665–1673. [PubMed] [Google Scholar]
- Vogt A. B., Kropshofer H., Moldenhauer G., Hämmerling G. J. Kinetic analysis of peptide loading onto HLA-DR molecules mediated by HLA-DM. Proc Natl Acad Sci U S A. 1996 Sep 3;93(18):9724–9729. doi: 10.1073/pnas.93.18.9724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogt A. B., Stern L. J., Amshoff C., Dobberstein B., Hämmerling G. J., Kropshofer H. Interference of distinct invariant chain regions with superantigen contact area and antigenic peptide binding groove of HLA-DR. J Immunol. 1995 Nov 15;155(10):4757–4765. [PubMed] [Google Scholar]



