Abstract
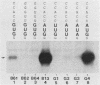
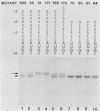
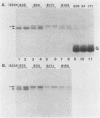
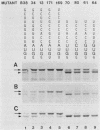
The context requirements for recognition of an initiator codon were evaluated in vitro by monitoring the relative use of two AUG codons that were strategically positioned to produce long (pre-chloramphenicol acetyl transferase [CAT]) and short versions of CAT protein. The yield of pre-CAT initiated from the 5'-proximal AUG codon increased, and synthesis of CAT from the second AUG codon decreased, as sequences flanking the first AUG codon increasingly resembled the eucaryotic consensus sequence. Thus, under prescribed conditions, the fidelity of initiation in extracts from animal as well as plant cells closely mimics what has been observed in vivo. Unexpectedly, recognition of an AUG codon in a suboptimal context was higher when the adjacent downstream sequence was capable of assuming a hairpin structure than when the downstream region was unstructured. This finding adds a new, positive dimension to regulation by mRNA secondary structure, which has been recognized previously as a negative regulator of initiation. Translation of pre-CAT from an AUG codon in a weak context was not preferentially inhibited under conditions of mRNA competition. That result is consistent with the scanning model, which predicts that recognition of the AUG codon is a late event that occurs after the competition-sensitive binding of a 40S ribosome-factor complex to the 5' end of mRNA. Initiation at non-AUG codons was evaluated in vitro and in vivo by introducing appropriate mutations in the CAT and preproinsulin genes. GUG was the most efficient of the six alternative initiator codons tested, but GUG in the optimal context for initiation functioned only 3 to 5% as efficiently as AUG. Initiation at non-AUG codons was artifactually enhanced in vitro at supraoptimal concentrations of magnesium.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahlquist P., Dasgupta R., Shih D. S., Zimmern D., Kaesberg P. Two-step binding of eukaryotic ribosomes to brome mosaic virus RNA3. Nature. 1979 Sep 27;281(5729):277–282. doi: 10.1038/281277a0. [DOI] [PubMed] [Google Scholar]
- Andersen R. D., Birren B. W., Taplitz S. J., Herschman H. R. Rat metallothionein-1 structural gene and three pseudogenes, one of which contains 5'-regulatory sequences. Mol Cell Biol. 1986 Jan;6(1):302–314. doi: 10.1128/mcb.6.1.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson C. W., Buzash-Pollert E. Can ACG serve as an initiation codon for protein synthesis in eucaryotic cells? Mol Cell Biol. 1985 Dec;5(12):3621–3624. doi: 10.1128/mcb.5.12.3621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachman N. J., Lomax M. I., Grossman L. I. Two bovine genes for cytochrome c oxidase subunit IV: a processed pseudogene and an expressed gene. Gene. 1987;55(2-3):219–229. doi: 10.1016/0378-1119(87)90282-4. [DOI] [PubMed] [Google Scholar]
- Baim S. B., Sherman F. mRNA structures influencing translation in the yeast Saccharomyces cerevisiae. Mol Cell Biol. 1988 Apr;8(4):1591–1601. doi: 10.1128/mcb.8.4.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becerra S. P., Rose J. A., Hardy M., Baroudy B. M., Anderson C. W. Direct mapping of adeno-associated virus capsid proteins B and C: a possible ACG initiation codon. Proc Natl Acad Sci U S A. 1985 Dec;82(23):7919–7923. doi: 10.1073/pnas.82.23.7919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattaneo R., Schmid A., Eschle D., Baczko K., ter Meulen V., Billeter M. A. Biased hypermutation and other genetic changes in defective measles viruses in human brain infections. Cell. 1988 Oct 21;55(2):255–265. doi: 10.1016/0092-8674(88)90048-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cigan A. M., Donahue T. F. Sequence and structural features associated with translational initiator regions in yeast--a review. Gene. 1987;59(1):1–18. doi: 10.1016/0378-1119(87)90261-7. [DOI] [PubMed] [Google Scholar]
- Cigan A. M., Pabich E. K., Donahue T. F. Mutational analysis of the HIS4 translational initiator region in Saccharomyces cerevisiae. Mol Cell Biol. 1988 Jul;8(7):2964–2975. doi: 10.1128/mcb.8.7.2964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clements J. M., Laz T. M., Sherman F. Efficiency of translation initiation by non-AUG codons in Saccharomyces cerevisiae. Mol Cell Biol. 1988 Oct;8(10):4533–4536. doi: 10.1128/mcb.8.10.4533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran J., Kolakofsky D. Ribosomal initiation from an ACG codon in the Sendai virus P/C mRNA. EMBO J. 1988 Jan;7(1):245–251. doi: 10.1002/j.1460-2075.1988.tb02806.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dixon L., Jiricny J., Hohn T. Oligonucleotide directed mutagenesis of cauliflower mosaic virus DNA using a repair-resistant nucleoside analogue: identification of an agnogene initiation codon. Gene. 1986;41(2-3):225–231. doi: 10.1016/0378-1119(86)90102-2. [DOI] [PubMed] [Google Scholar]
- Donahue T. F., Cigan A. M. Genetic selection for mutations that reduce or abolish ribosomal recognition of the HIS4 translational initiator region. Mol Cell Biol. 1988 Jul;8(7):2955–2963. doi: 10.1128/mcb.8.7.2955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Florkiewicz R. Z., Sommer A. Human basic fibroblast growth factor gene encodes four polypeptides: three initiate translation from non-AUG codons. Proc Natl Acad Sci U S A. 1989 Jun;86(11):3978–3981. doi: 10.1073/pnas.86.11.3978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallie D. R., Sleat D. E., Watts J. W., Turner P. C., Wilson T. M. A comparison of eukaryotic viral 5'-leader sequences as enhancers of mRNA expression in vivo. Nucleic Acids Res. 1987 Nov 11;15(21):8693–8711. doi: 10.1093/nar/15.21.8693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grass D. S., Manley J. L. Selective translation initiation on bicistronic simian virus 40 late mRNA. J Virol. 1987 Jul;61(7):2331–2335. doi: 10.1128/jvi.61.7.2331-2335.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta K. C., Patwardhan S. ACG, the initiator codon for a Sendai virus protein. J Biol Chem. 1988 Jun 25;263(18):8553–8556. [PubMed] [Google Scholar]
- Hann S. R., King M. W., Bentley D. L., Anderson C. W., Eisenman R. N. A non-AUG translational initiation in c-myc exon 1 generates an N-terminally distinct protein whose synthesis is disrupted in Burkitt's lymphomas. Cell. 1988 Jan 29;52(2):185–195. doi: 10.1016/0092-8674(88)90507-7. [DOI] [PubMed] [Google Scholar]
- Hunter A. R., Farrell P. J., Jackson R. J., Hunt T. The role of polyamines in cell-free protein synthesis in the wheat-germ system. Eur J Biochem. 1977 May 2;75(1):149–157. doi: 10.1111/j.1432-1033.1977.tb11512.x. [DOI] [PubMed] [Google Scholar]
- Jacks T., Madhani H. D., Masiarz F. R., Varmus H. E. Signals for ribosomal frameshifting in the Rous sarcoma virus gag-pol region. Cell. 1988 Nov 4;55(3):447–458. doi: 10.1016/0092-8674(88)90031-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kodaira M., Kinashi T., Umemura I., Matsuda F., Noma T., Ono Y., Honjo T. Organization and evolution of variable region genes of the human immunoglobulin heavy chain. J Mol Biol. 1986 Aug 20;190(4):529–541. doi: 10.1016/0022-2836(86)90239-1. [DOI] [PubMed] [Google Scholar]
- Kozak M. An analysis of 5'-noncoding sequences from 699 vertebrate messenger RNAs. Nucleic Acids Res. 1987 Oct 26;15(20):8125–8148. doi: 10.1093/nar/15.20.8125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Analysis of ribosome binding sites from the s1 message of reovirus. Initiation at the first and second AUG codons. J Mol Biol. 1982 Apr 25;156(4):807–820. doi: 10.1016/0022-2836(82)90143-7. [DOI] [PubMed] [Google Scholar]
- Kozak M. At least six nucleotides preceding the AUG initiator codon enhance translation in mammalian cells. J Mol Biol. 1987 Aug 20;196(4):947–950. doi: 10.1016/0022-2836(87)90418-9. [DOI] [PubMed] [Google Scholar]
- Kozak M. Bifunctional messenger RNAs in eukaryotes. Cell. 1986 Nov 21;47(4):481–483. doi: 10.1016/0092-8674(86)90609-4. [DOI] [PubMed] [Google Scholar]
- Kozak M. Circumstances and mechanisms of inhibition of translation by secondary structure in eucaryotic mRNAs. Mol Cell Biol. 1989 Nov;9(11):5134–5142. doi: 10.1128/mcb.9.11.5134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Influence of mRNA secondary structure on binding and migration of 40S ribosomal subunits. Cell. 1980 Jan;19(1):79–90. doi: 10.1016/0092-8674(80)90390-6. [DOI] [PubMed] [Google Scholar]
- Kozak M. Influences of mRNA secondary structure on initiation by eukaryotic ribosomes. Proc Natl Acad Sci U S A. 1986 May;83(9):2850–2854. doi: 10.1073/pnas.83.9.2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Migration of 40 S ribosomal subunits on messenger RNA when initiation is perturbed by lowering magnesium or adding drugs. J Biol Chem. 1979 Jun 10;254(11):4731–4738. [PubMed] [Google Scholar]
- Kozak M. Point mutations close to the AUG initiator codon affect the efficiency of translation of rat preproinsulin in vivo. Nature. 1984 Mar 15;308(5956):241–246. doi: 10.1038/308241a0. [DOI] [PubMed] [Google Scholar]
- Kozak M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell. 1986 Jan 31;44(2):283–292. doi: 10.1016/0092-8674(86)90762-2. [DOI] [PubMed] [Google Scholar]
- Kozak M. The scanning model for translation: an update. J Cell Biol. 1989 Feb;108(2):229–241. doi: 10.1083/jcb.108.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Translation of insulin-related polypeptides from messenger RNAs with tandemly reiterated copies of the ribosome binding site. Cell. 1983 Oct;34(3):971–978. doi: 10.1016/0092-8674(83)90554-8. [DOI] [PubMed] [Google Scholar]
- Kwoh T. J., Kwoh D. Y., McCue A. W., Davis G. R., Patrick D., Gingeras T. R. Introduction and expression of the bacterial PaeR7 methylase gene in mammalian cells. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7713–7717. doi: 10.1073/pnas.83.20.7713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Landsman D., McBride O. W., Soares N., Crippa M. P., Srikantha T., Bustin M. Chromosomal protein HMG-14. Identification, characterization, and chromosome localization of a functional gene from the large human multigene family. J Biol Chem. 1989 Feb 25;264(6):3421–3427. [PubMed] [Google Scholar]
- Landsman D., Srikantha T., Westermann R., Bustin M. Chromosomal protein HMG-14. Complete human cDNA sequence and evidence for a multigene family. J Biol Chem. 1986 Dec 5;261(34):16082–16086. [PubMed] [Google Scholar]
- Lodish H. F., Rose J. K. Relative importance of 7-methylguanosine in ribosome binding and translation of vesicular stomatitis virus mRNA in wheat germ and reticulocyte cell-free systems. J Biol Chem. 1977 Feb 25;252(4):1181–1188. [PubMed] [Google Scholar]
- Lütcke H. A., Chow K. C., Mickel F. S., Moss K. A., Kern H. F., Scheele G. A. Selection of AUG initiation codons differs in plants and animals. EMBO J. 1987 Jan;6(1):43–48. doi: 10.1002/j.1460-2075.1987.tb04716.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcu K., Dudock B. Characterization of a highly efficient protein synthesizing system derived from commercial wheat germ. Nucleic Acids Res. 1974 Nov;1(11):1385–1397. doi: 10.1093/nar/1.11.1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Rear J. J., Temin H. M. Spontaneous changes in nucleotide sequence in proviruses of spleen necrosis virus, an avian retrovirus. Proc Natl Acad Sci U S A. 1982 Feb;79(4):1230–1234. doi: 10.1073/pnas.79.4.1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peabody D. S. Translation initiation at an ACG triplet in mammalian cells. J Biol Chem. 1987 Aug 25;262(24):11847–11851. [PubMed] [Google Scholar]
- Peabody D. S. Translation initiation at non-AUG triplets in mammalian cells. J Biol Chem. 1989 Mar 25;264(9):5031–5035. [PubMed] [Google Scholar]
- Pelletier J., Flynn M. E., Kaplan G., Racaniello V., Sonenberg N. Mutational analysis of upstream AUG codons of poliovirus RNA. J Virol. 1988 Dec;62(12):4486–4492. doi: 10.1128/jvi.62.12.4486-4492.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinck M., Fritsch C., Ravelonandro M., Thivent C., Pinck L. Binding of ribosomes to the 5' leader sequence (N = 258) of RNA 3 from alfalfa mosaic virus. Nucleic Acids Res. 1981 Mar 11;9(5):1087–1100. doi: 10.1093/nar/9.5.1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pirastu M., Saglio G., Chang J. C., Cao A., Kan Y. W. Initiation codon mutation as a cause of alpha thalassemia. J Biol Chem. 1984 Oct 25;259(20):12315–12317. [PubMed] [Google Scholar]
- Prats A. C., De Billy G., Wang P., Darlix J. L. CUG initiation codon used for the synthesis of a cell surface antigen coded by the murine leukemia virus. J Mol Biol. 1989 Jan 20;205(2):363–372. doi: 10.1016/0022-2836(89)90347-1. [DOI] [PubMed] [Google Scholar]
- Prats H., Kaghad M., Prats A. C., Klagsbrun M., Lélias J. M., Liauzun P., Chalon P., Tauber J. P., Amalric F., Smith J. A. High molecular mass forms of basic fibroblast growth factor are initiated by alternative CUG codons. Proc Natl Acad Sci U S A. 1989 Mar;86(6):1836–1840. doi: 10.1073/pnas.86.6.1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt G., Galpine A., Sharp N., Palmer S., Clemens M. J. Regulation of in vitro translation by double-stranded RNA in mammalian cell mRNA preparations. Nucleic Acids Res. 1988 Apr 25;16(8):3497–3510. doi: 10.1093/nar/16.8.3497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Resnick J., Shenk T. Simian virus 40 agnoprotein facilitates normal nuclear location of the major capsid polypeptide and cell-to-cell spread of virus. J Virol. 1986 Dec;60(3):1098–1106. doi: 10.1128/jvi.60.3.1098-1106.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts B. E., Paterson B. M. Efficient translation of tobacco mosaic virus RNA and rabbit globin 9S RNA in a cell-free system from commercial wheat germ. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2330–2334. doi: 10.1073/pnas.70.8.2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosa F., Berissi H., Weissenbach J., Maroteaux L., Fellous M., Revel M. The beta2-microglobulin mRNA in human Daudi cells has a mutated initiation codon but is still inducible by interferon. EMBO J. 1983;2(2):239–243. doi: 10.1002/j.1460-2075.1983.tb01412.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sangar D. V., Newton S. E., Rowlands D. J., Clarke B. E. All foot and mouth disease virus serotypes initiate protein synthesis at two separate AUGs. Nucleic Acids Res. 1987 Apr 24;15(8):3305–3315. doi: 10.1093/nar/15.8.3305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlicht H. J., Radziwill G., Schaller H. Synthesis and encapsidation of duck hepatitis B virus reverse transcriptase do not require formation of core-polymerase fusion proteins. Cell. 1989 Jan 13;56(1):85–92. doi: 10.1016/0092-8674(89)90986-0. [DOI] [PubMed] [Google Scholar]
- Shih D. S., Kaesberg P. Translation of the RNAs of brome mosaic virus: the monocistronic nature of RNA1 and RNA2. J Mol Biol. 1976 May 5;103(1):77–88. doi: 10.1016/0022-2836(76)90053-x. [DOI] [PubMed] [Google Scholar]
- Shpaer E. G. The secondary structure of mRNAs from Escherichia coli: its possible role in increasing the accuracy of translation. Nucleic Acids Res. 1985 Jan 11;13(1):275–288. doi: 10.1093/nar/13.1.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simonsen C. S., Walter M., Levinson A. D. Expression of the plasmid-encoded type I dihydrofolate reductase gene in cultured mammalian cells: a novel selectable marker. Nucleic Acids Res. 1988 Mar 25;16(5):2235–2246. doi: 10.1093/nar/16.5.2235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terwilliger E., Burghoff R., Sia R., Sodroski J., Haseltine W., Rosen C. The art gene product of human immunodeficiency virus is required for replication. J Virol. 1988 Feb;62(2):655–658. doi: 10.1128/jvi.62.2.655-658.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trempe J. P., Carter B. J. Alternate mRNA splicing is required for synthesis of adeno-associated virus VP1 capsid protein. J Virol. 1988 Sep;62(9):3356–3363. doi: 10.1128/jvi.62.9.3356-3363.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyc K., Konarska M., Gross H. J., Filipowicz W. Multiple ribosome binding to the 5'-terminal leader sequence of tobacco mosaic virus RNA. Assembly of an 80S ribosome X mRNA complex at the AUU codon. Eur J Biochem. 1984 May 2;140(3):503–511. doi: 10.1111/j.1432-1033.1984.tb08131.x. [DOI] [PubMed] [Google Scholar]
- Walker G. M. Magnesium and cell cycle control: an update. Magnesium. 1986;5(1):9–23. [PubMed] [Google Scholar]
- Wawrousek E. F., Piatigorsky J. Expression of the delta 1-crystallin gene: salt-dependent alteration in the cell-free synthesis of delta-crystallin polypeptides. Gene. 1987;58(2-3):155–165. doi: 10.1016/0378-1119(87)90372-6. [DOI] [PubMed] [Google Scholar]
- Weyens G., Charlier D., Roovers M., Piérard A., Glansdorff N. On the role of the Shine-Dalgarno sequence in determining the efficiency of translation initiation at a weak start codon in the car operon of Escherichia coli K12. J Mol Biol. 1988 Dec 20;204(4):1045–1048. doi: 10.1016/0022-2836(88)90061-7. [DOI] [PubMed] [Google Scholar]
- Zhang H., Scholl R., Browse J., Somerville C. Double stranded DNA sequencing as a choice for DNA sequencing. Nucleic Acids Res. 1988 Feb 11;16(3):1220–1220. doi: 10.1093/nar/16.3.1220. [DOI] [PMC free article] [PubMed] [Google Scholar]