Abstract
Bone marrow mesenchymal stem cells (MSCs) comprise a heterogeneous population of postnatal progenitor cells with profound immunomodulatory properties, such as upregulation of Foxp3+ regulatory T cells (Tregs) and downregulation of Th17 cells. However, it is unknown whether different MSC subpopulations possess the same range of immunomodulatory function. Here, we show that a subset of single colony-derived MSCs producing IL-17 is different from bulk MSC population in that it cannot upregulate Tregs, downregulate Th17 cells, or ameliorate disease phenotypes in a colitis mouse model. Mechanistically, we reveal that IL-17, produced by these MSCs, activates the NFκB pathway to downregulate TGF-β production in MSCs, resulting in abolishment of MSC-based immunomodulation. Furthermore, we show that NFκB is able to directly bind to TGF-β promoter region to regulate TGF-β expression in MSCs. Moreover, these IL-17+ MSCs possess anti-Candida albicans growth effects in vitro and therapeutic effect in C. albicans-infected mice. In summary, this study shows that MSCs contain an IL-17+ subset capable of inhibiting C. albicans growth, but attenuating MSC-based immunosuppression via NFκB-mediated downregulation of TGF-β.
Keywords: stem cell biology, development, immunity, molecular pathogenesis
Introduction
Mesenchymal stem cells (MSCs) are progenitor cells with capacity of self-renewal, multilineage differentiation, and immunomodulation. Emerging experimental and clinical evidence has shown that systemic infusion of MSCs results in significant immunosuppressive effects in terms of treating a variety of inflammatory and autoimmune diseases by inhibiting the proliferation and function of immune cells, including dendritic cells, T and B lymphocytes, and natural killer (NK) cells1,2,3. The immunomodulatory properties of MSCs are associated with direct cell-cell contact with T cells or/and production of a variety of cytokines and chemokines, such as interleukin 10 (IL-10), nitric oxide (NO), indoleamine 2, 3-dioxygenase (IDO), prostaglandin E2 (PGE2), transforming growth factor β (TGF-β), tumor necrosis factor-inducible gene 6 (TSG-6), and chemokine (C-C motif) ligand 2 (CCL2)1,2,4,5,6,7,8. These cytokines and chemokines contribute to the consequent regulation of CD4+CD25+Foxp3+ regulatory T (Treg) cells and proinflammatory T helper 17 (Th17) cells, balancing the host immune homeostasis8,9,10.
Although it is well known that MSCs possess immunosuppressive effect, it is unclear whether all MSC subsets have an equal degree of immunosuppression. Recently, several reports showed that engagement of different Toll-like receptors (TLRs) in MSCs either enhanced or inhibited their immunosuppressive properties11,12,13. For instance, TLR4-primed MSCs produced proinflammatory mediators, while TLR3-primed MSCs expressed mostly immunosuppressive ones12. Furthermore, NO and IDO were reported to act as a switch in determining MSC-based immunosuppression or enhancing immune response14. The accumulated evidence suggests that MSCs may possess variable immunoregulatory function, but the detailed mechanism is unknown.
Interleukin-17 (IL-17), a proinflammatory cytokine, has critical roles in the pathogenesis of autoimmune diseases, such as rheumatoid arthritis, psoriasis, inflammatory bowel diseases, diabetes, and multiple sclerosis15. IL-17 also contributes to host defense against extracellular and intracellular pathogens, including Aspergillus fumigatus, Cryptococcus neoformans and Candida albicans (C. albicans)16,17. In addition to CD4+ Th17 cells, a variety of T cells, including CD8+ T cells (Tc17), γδT cells, natural killer T (NKT) cells, innate immune cells, macrophages, and paneth cells, have the capacity to produce IL-1718,19,20,21. It was reported that Foxp3+/IL-17+ Tregs lose their immunosuppressive function due to production of IL-1722. In this study, we identify a new subset of IL-17+ MSCs capable of inhibiting C. albicans growth and attenuating cell-based immunosuppression.
Results
Identification of IL-17+ bone marrow MSCs
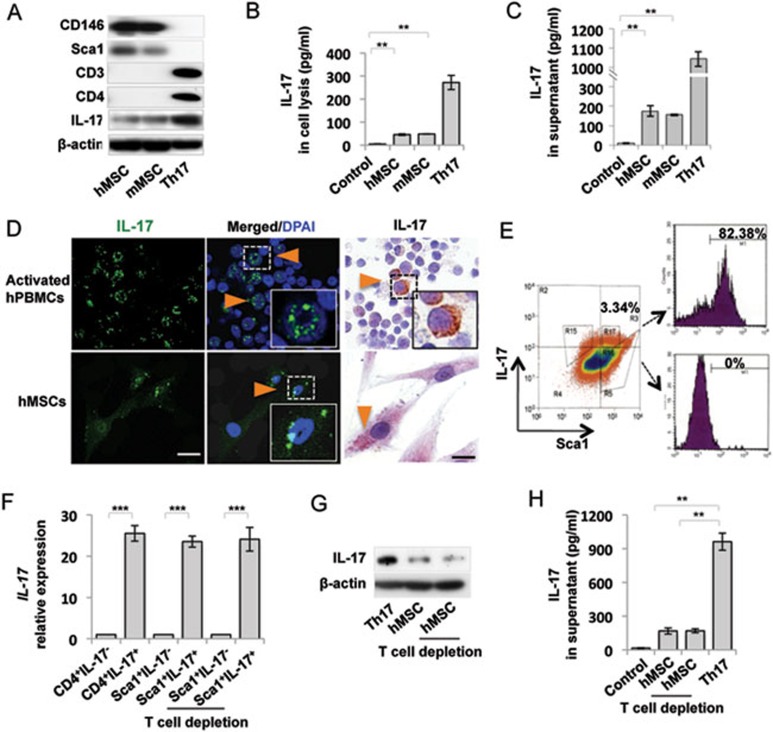
To examine whether MSCs contain a subset of cells expressing IL-17, we used western blot and ELISA analysis to show that culture expanded human (h) and mouse (m) MSCs expressed IL-17 (Figure 1A and 1B). hMSCs and mMSCs expressed CD146 and Sca1, but not T cell markers CD3, CD4, CD8a and hematopoietic stem cell marker CD34, as assessed by western blot (Figure 1A) and flow cytometric analysis (Supplementary information, Figure S1). The secretion of IL-17 to the culture medium was detectable by ELISA assay (Figure 1C). Immunofluorescent and immunocytochemical staining also showed that hMSCs expressed IL-17, but not T cell markers CD3, CD4 and CD8a. Activated human peripheral blood mononuclear cells (hPBMCs) were used as a positive control (Figure 1D and Supplementary information, Figure S2). To determine the percentage of IL-17+ cells in total MSCs, flow cytometric analysis was performed and showed that 3.34% MSCs were positive for both IL-17 and Sca-1 (Figure 1E). The Sca1+IL-17+ MSCs, but not Sca1+IL-17− MSCs, expressed IL-17 mRNA, as assessed by qPCR (Figure 1F). Recent study showed that MSCs promoted Th17 cell expansion through activating caspase 1 when co-culturing with activated T cells or PBMCs23. To exclude the possibility of IL-17 produced by T cell contamination in MSC population, CD3 antibody/complement treatment was used to deplete potential contaminated T cells in MSC cultures (Supplementary information, Figure S3). qPCR analysis showed that Sca1+IL-17+ MSCs expressed similar level of IL-17 mRNA after CD3 antibody/complement treatment. Western blot and ELISA analysis also showed that T cell depletion did not affect expression levels of IL-17 in hMSCs (Figure 1G and 1H). Since combinatorial cytokine treatment, such as IL-6, TGF-β1, IL-1β and IL-23, stimulated Th17 polarization and IL-17 secretion24,25, we examined whether such cytokine treatment would alter IL-17 expression in MSCs. After treating with a variety of combinatorial cytokines, we found that IL-1β and IL-6 treatment was capable of upregulating the number of IL-17+ MSCs and IL-17 secretion to the culture medium, as assessed by flow cytometric analysis and ELISA (Supplementary information, Figure S4A and S4B). Additionally, IL-1β and IL-6 treatment elevated IL-17 expression, as assessed by qPCR and western blot analysis (Supplementary information, Figure S4C and S4D). It was reported that transcription factor retinoid orphan receptor γ T (RORγt) regulated IL-17 expression via signal transducer and activator of transcription 3 (Stat3)26,27. Therefore, we measured the expression levels of Stat3 and RORγt in IL-1β and IL-6-treated MSCs and found that IL-1β and IL-6 treatment elevated IL-17 expression, along with increased p-Stat3 and RORγt, as assessed by western blotting analysis (Supplementary information, Figure S4D). When MSCs were pretreated with 5, 15-DPP, a selective Stat3 inhibitor, IL-1β and IL-6-induced upregulation of IL-17 and RORγt was blocked (Supplementary information, Figure S4E). Collectively, these data indicate that MSCs contain a unique subset of IL-17+ cells and that IL-1β and IL-6 treatment elevated the number of IL-17+ MSCs through Stat3 and RORγt signaling.
Figure 1.
Identification of IL-17+ MSCs. (A) Western blot analysis showed that human (h) and mouse (m) MSCs expressed IL-17, CD146 and Sca1, but not CD3 and CD4. Th17 cells were used as a positive control. (B) ELISA assay showed that hMSCs and mMSCs produced IL-17. Th17 cells were used as a positive control. Protein lysis buffer was used as a negative control. (C) IL-17 was detected in 5× concentrated MSC culture medium, as assessed by ELISA analysis. Th17 cells were used as a positive control. Regular culture medium was used as a negative control. (D) Immunofluorescent and immunocytochemical staining showed that hMSCs expressed IL-17 (Orange triangles). Activated hPBMCs were used as a positive control. (E) Flow cytometry analysis showed that 3.34% of MSCs were positive for IL-17 and Sca1. Sca1+ cells were gated and sorted for IL-17+ (82.38% purity) and IL-17− (100% purity) populations for the following experiments. (F) qPCR analysis showed that Sca1+IL-17+ MSCs, but not Sca1+IL-17− MSCs, expressed IL-17 mRNA. CD3 antibody/complement treatment could not deplete IL-17 expression in Sca1+IL-17+ MSCs. CD4+IL-17+ and CD4+IL-17− T cells were used as positive and negative controls, respectively. (G, H) Western blot and ELISA analysis showed that CD3 antibody/complement treatment did not affect IL-17 expression in hMSCs. **P < 0.01 and ***P < 0.001 (mean ± SD). Scale bar, 25 μm. The data are representative of three independent experiments.
Single colony-derived IL-17+ MSCs showed compromised immunosuppressive capacity
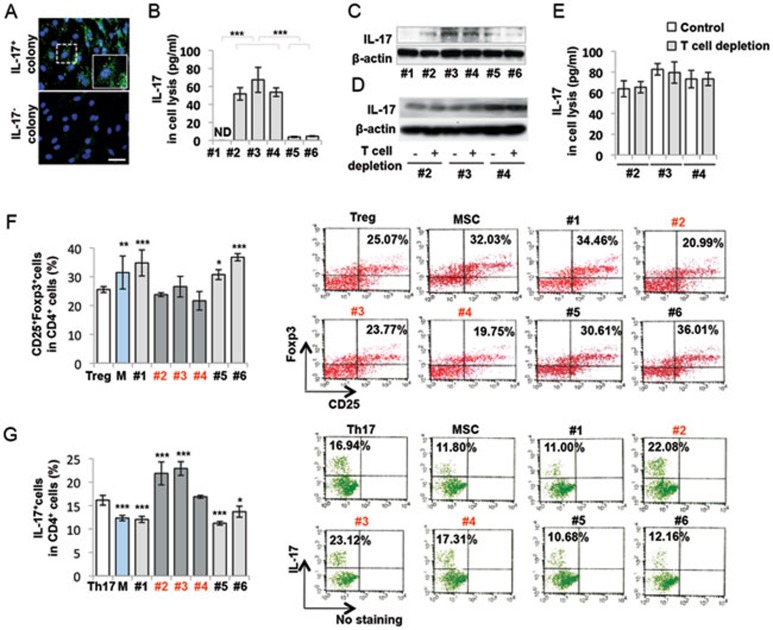
Since MSCs are a heterogeneous population containing multiple colony clusters, we confirmed that part of single colony-derived MSCs express IL-17. Fluorescent immunocytostaining showed that a small fraction (3.33%) of single colonies was positive for IL-17 antibody staining (Figure 2A). Next, we performed ELISA assay and selected three IL-17+ (#2, #3 and #4) and three IL-17− (#1, #5 and #6) single colony-derived MSCs (Figure 2B). Western blot analysis confirmed that the IL-17+ colonies expressed a significantly higher level of IL-17 than IL-17− colonies (Figure 2C). IL-17+ (#2, #3 and #4) MSCs were positive for CD146 and Sca1, but negative for CD3, CD4, CD8a and CD34, as assessed by flow cytometric analysis (Supplementary information, Figure S5). T cell depletion with CD3 antibody/complement treatment did not alter IL-17 expression in IL-17+ MSCs (#2, #3, #4), as assessed by western blot (Figure 2D) and ELISA analysis (Figure 2E). Single colony-derived MSCs may show variable stem cell properties, including proliferation and multipotent differentiation28. We showed that all selected single colony-derived MSCs were capable of maintaining proliferation as assayed by BrdU incorporation (Supplementary information, Figure S6A), in vitro osteogenic differentiation by mineralized nodule formation and expression of ALP (Supplementary information, Figure S6B), and adipogenic differentiation by staining of Oil Red O-positive adipocytes and expression of PPARγ and LPL (Supplementary information, Figure S6C). These results confirmed the stem cell properties and heterogeneity of these single colony-derived MSCs28. IL-17 is a proinflammatory cytokine capable of promoting inflammation by inducing various proinflammatory cytokines and chemokines29. Since MSC-based immunomodulation is associated with upregulation of Tregs and downregualtion of Th17 cells8,9,10, we examined whether IL-17+ MSCs were able to regulate Tregs and Th17 cells in a T cell/MSC co-culture system. When hMSCs were co-cultured with TGF-β1 and IL-2-treated or TGF-β1 and IL-6-treated human T cells, IL-17+MSCs (#2, #3 and #4) showed a significantly reduced capacity to upregulate the levels of Tregs and IL-10 and downregulate the levels of Th17 cells and IL-17 when compared to bulk and IL-17− MSCs (Figure 2F and 2G). These data indicate that IL-17+ MSCs exhibit compromised immunosuppressive function.
Figure 2.
IL-17+ MSCs showed compromised immunosuppressive capacity. (A) Fluorescent immunocytostaining showed that a small population of single colony cluster (3.33%) of hMSCs expressed IL-17. Upper panel, IL-17+ mMSC single colony. Lower panel, IL-17− mMSC single colony. (B, C) Single colony-derived hMSCs exhibited different levels of IL-17 expression, as assessed by ELISA (B) and western blot (C). Colonies #1, #5 and #6 were denoted IL-17− MSCs, and colonies #2, #3 and #4 were denoted IL-17+ MSCs. (D, E) CD3 antibody/complement treatment did not deplete IL-17 expression in IL-17+ MSCs, as assessed by western blot (D) and ELISA (E). (F) After co-cultured with TGF-β1/IL-2-treated T cells, bulk MSCs and IL-17− MSCs (#1, #5 and #6), but not IL-17+ MSCs (#2, #3 and #4), upregulated the level of Tregs. Induced Treg without co-culture with MSCs was used as untreated control (Treg). (G) After co-cultured with TGF-β1/IL-6-treated T cells, bulk MSCs and IL-17− MSCs (#1, #5 and #6) significantly inhibited Th17 cells. However, IL-17+ MSCs (#2, #3 and #4) generally showed no significant effect on inhibiting Th17 cells. Clones #2 and #3 IL-17+ MSCs even elevated the level of Th17 cells. Th17: induced Th17 without co-cultured MSCs was used as untreated control. M: MSC. *P < 0.05; **P < 0.01 and ***P < 0.001 (mean ± SD). (F) *compared to Treg group (G) *compared to Th17 group. Scale bar, 50 μm. The data are representative of three independent experiments.
IL-17+ MSCs showed impaired immunotherapeutic effect in colitis mice
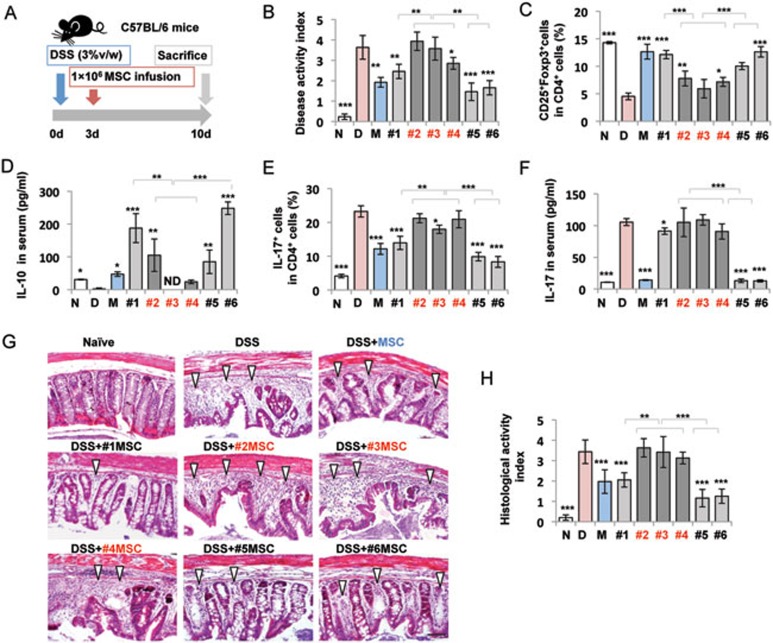
To further examine the immunomodulatory property of IL-17+ MSCs, we compared the therapeutic effect of IL-17+ MSCs and bulk MSCs in dextran sulfate sodium (DSS)-induced colitis mice (Figure 3A)30,31. Oral administration of 3% DSS for 10 days induced acute colitis in C57BL/6 mice, as characterized by an overall elevation of disease activity index (DAI) based on the presence of sustained weight loss and bloody diarrhea/loose feces9,31. Bulk and single colony-derived MSCs (#1-#6) were systemically infused into experimental colitis mice on day 3 post 3% DSS induction (Figure 3A). Bulk and IL-17− MSC infusions (#1, #5 and #6), but not IL-17+ MSC (#2, #3 and #4) infusion, significantly reduced DAI and levels of Th17 cells and IL-17, along with elevated levels of Tregs and IL-10 (Figure 3B-3F). In addition, bulk and IL-17− MSC (#1, #5 and #6) infusion significantly ameliorated colonic transmural inflammation and suppressed mucosal ulceration, thus restoring normal intestinal structure and resulting in a reduced histological index, but IL-17+MSC (#2, #3 and #4) infusion failed to restore normal intestinal structure (Figure 3G and 3H). These data together confirm that IL-17+ MSCs have compromised immunomodulatory function.
Figure 3.
IL-17+ MSCs showed impaired immunotherapeutic effect in colitis mice. (A) Schema of MSC infusion in DSS-induced experimental colitis mice. (B) Bulk MSC and IL-17− MSC (#1, #5 and #6) infusions significantly reduced the disease activity index (DAI), as determined by the presence of sustained weight loss and bloody diarrhea/loose feces. IL-17+ MSC (#2 and #3) infusion did not reduce the DAI. (C, D) Treg number and IL-10 level were significantly decreased in colitis mice compared to control C57BL/6 mice. IL-17− MSC (#1, #5 and #6) infusion significantly elevated the level of Tregs and serum IL-10 in colitis mice compared to IL-17+ MSC (#2, #3 and #4) treatment groups. (E, F) Th17 cell number and IL-17 level were significantly elevated in colitis mice compared to control C57/BL6 mice. Bulk MSC and IL-17− MSC (#1, #5 and #6) infusions, but not IL-17+ MSC (#2, #3 and #4) infusion, significantly reduced the levels of Th17 cells and serum IL-17 in colitis mice. (G, H) Hematoxylin and eosin staining showed infiltration of inflammatory cells (white triangles) in colitis mice. Bulk MSC and IL-17− MSC infusions, but not IL-17+ MSC infusion, significantly rescued disease phenotypes and reduced histological index in colitis mice. N: Naive; D: DSS; M: MSC. *P < 0.05; **P < 0.01 and ***P < 0.001 (mean ± SD). (B-F, H) *compared to DSS group. Scale bars, 100 μm. The data are representative of three independent experiments.
IL-17+ MSCs showed reduced TGF-β1 expression
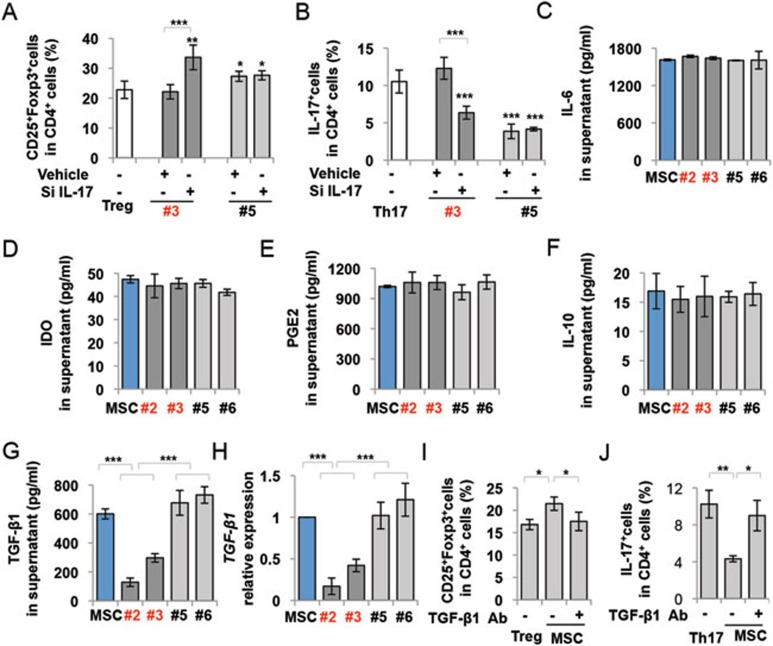
To examine whether IL-17 directly contributes to compromised immunoregulation in IL-17+ MSCs, we used small interfering (si) RNA to knockdown IL-17 expression in IL-17+ MSCs (Supplementary information, Figure S7) and found that reduced IL-17 expression in IL-17+ MSCs rescued their capacity to upregulate Tregs and downregulate Th17 cells (Figure 4A and 4B), suggesting that IL-17 production may be involved in the impairment of MSC-mediated immuomodulation. Next, we compared expression levels of MSC-associated immunomodulatory molecules, including IDO, IL-6, IL-10, PGE2, TGF-β1, TNF-α and IFN-γ in IL-17+ and IL-17− MSCs. No significant difference was detected in terms of expression levels of IL-6, IDO, PGE2 or IL-10 between IL-17+ and IL-17− MSCs (Figure 4C-4F). Expression levels of TNF-α and IFN-γ were too low to be detected in MSCs (data not shown). However, the levels of TGF-β1 secretion and expression were significantly reduced in IL-17+ MSCs compared to bulk and IL-17− MSCs, as assessed by ELISA and qPCR, respectively (Figure 4G and 4H). Using neutralizing antibody treatment, we next verified that the inhibition of TGF-β1 in MSCs reduced the capacity of MSCs to upregulate Tregs (Figure 4I) and downregulate Th17 cells (Figure 4J). These findings suggest that reduction in TGF-β1 production may play a critical role in reduced immunosuppression of IL-17+ MSCs.
Figure 4.
IL-17+ MSCs showed reduced TGF-β1 expression. (A, B) When IL-17 was knocked down by siRNA, the capacity to upregulate Tregs (A) and downregulate Th17 cells (B) in IL-17+ MSCs (#3), but not in IL-17− MSCs (#5), was increased. (C-G) ELISA analysis showed that expression levels of IL-6 (C), IDO (D), PGE2 (E), and IL-10 (F) were not significantly different among different single colony-derived MSCs and bulk MSCs. However, the expression level of TGF-β1 was significantly reduced in IL-17+ MSCs (#2, #3) compared to IL-17− MSCs (#5, #6) and bulk MSCs, as assessed by ELISA (G). (H) qPCR analysis showed that IL-17+ MSCs (#2, #3) expressed reduced TGF-β1 mRNA level compared to IL-17− MSCs (#5, #6) and bulk MSCs. (I, J) Blocking TGF-β1 with neutralizing antibody in MSC/T cell co-culture system attenuated MSC-mediated upregulation of Tregs (I) and downregulation of Th17 cells (J). *P < 0.05; **P < 0.01 and ***P < 0.001. (mean ± SD). (A) *compared to Treg group, (B) *compared to Th17 group. The data are representative of three independent experiments.
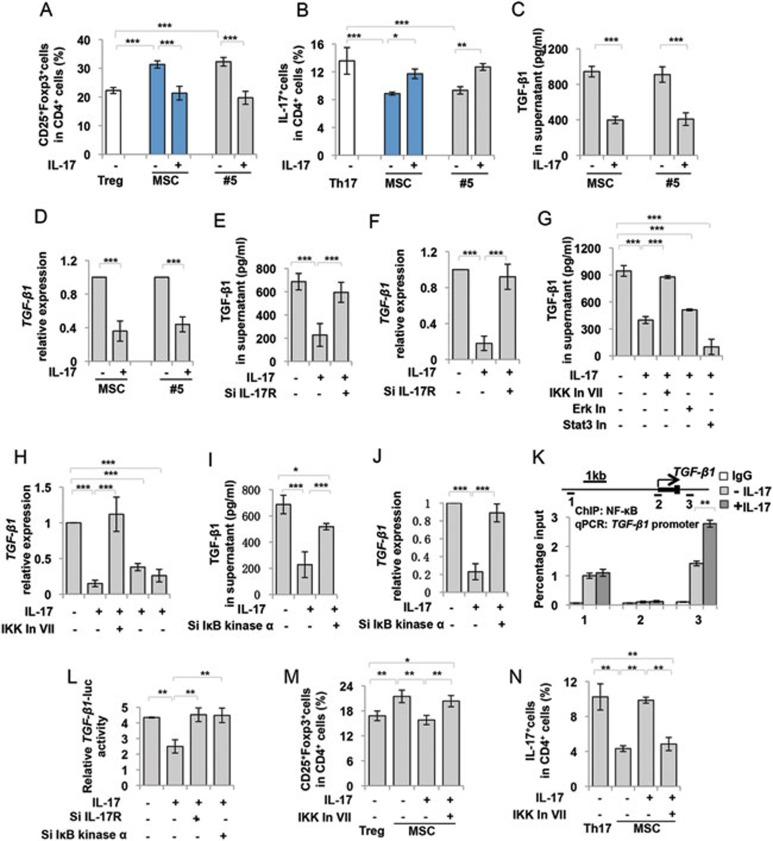
IL-17 inhibited TGF-β1 expression via activation of NFκB pathway
To verify the functional role of IL-17 in MSC-based immunomodulation, we used IL-17 to treat bulk MSCs and IL-17− MSCs (#5), and found that IL-17 treatment induced a significant reduction in MSC-mediated upregulation of Tregs and downregulation of Th17 cells compared to untreated control group (Figure 5A and 5B). Next, we examined whether IL-17 contributed to the decreased TGF-β1 expression in MSCs. ELISA and qPCR analysis showed that IL-17 treatment downregulated TGF-β1 expression in bulk and IL-17− MSCs (#5) (Figure 5C and 5D). This downregulated TGF-β expression could be reversed by knockdown of IL-17 receptor (IL-17R) expression by siRNA (Figure 5E, 5F and Supplementary information, Figure S7). Since IL-17 activates several signaling pathways, including nuclear factor (NF) κB pathway, extracellular signal-regulated kinase (ERK), JNK, Stat3 and p38 pathways, to induce proinflammatory cytokine and chemokine production21, we performed western blot analysis and showed that IL-17 treatment induced phosphorylation of p65, IκBα, ERK and Stat3, but not p38 and JNK, in MSCs (Supplementary information, Figure S8). To identify the specific pathway that may contribute to IL-17-mediated reduction of TGF-β1 expression, we used specific inhibitors to effectively block IL-17 downstream pathways (Supplementary information, Figure S8). Subsequently, ELISA and qPCR analysis showed that IκB kinase (IKK) inhibitor VII (inhibition of NFκB-mediated gene expression), but not Stat3 (5, 15-DP) or Erk (PD325901) inhibitor rescued TGF-β1 expression in IL-17-treated MSCs (Figure 5G and 5H). Knockdown of IKKα by siRNA to block IκBα degradation (inhibition of NFκB-mediated gene expression; IKKα siRNA: sc-29365, Santa Cruz Biotechnology, Santa Cruz, CA, USA) also blocked IL-17-induced downregulation of TGF-β1 in MSCs (Figure 5I, 5J and Supplementary information, Figure S7), suggesting that the NFκB pathway may play a critical role in IL-17-mediated downregulation of TGF-β1. To determine how NFκB regulates TGF-β1 expression in MSCs, NFκB-binding consensus sequence in TGF-β1 promoter region was examined by chromatin immunoprecipitation (ChIP) with NFκB-specific antibody, followed by qPCR assay using a primer pair designed near the predicted site. Of the three putative binding sites, one NFκB-binding DNA sequence was significantly enriched in IL-17-treated MSCs (Figure 5K). Luciferase assay showed that TGF-β1 promoter activity was inhibited by IL-17 treatment, which could be rescued by IL-17R and IKKα siRNAs (Figure 5L). To further verify the role of IL-17 in inhibiting MSC-based immunomodulation via the NFκB pathway, we used IKK inhibitor VII to block IL-17-activated NFκB signaling and found that IKK inhibitor VII treatment enabled IL-17-treated MSCs to upregulate Tregs and downregulate Th17 cells (Figure 5M and 5N). These findings indicate that IL-17 blocks MSC immunosuppressive function via IL-17R/ NFκB/TGF-β1 signaling (Supplementary information, Figure S9).
Figure 5.
IL-17 downregulated TGF-β1 expression via activation of NF-κB pathway. (A, B) Recombinant IL-17 (20 ng/ml) treatment attenuated bulk and IL-17– MSC (#5)-mediated upregulation of Tregs (A) and downregulation of Th17 cells (B). (C, D) Recombinant IL-17 treatment downregulated TGF-β1 expression in bulk and IL-17– MSCs (#5), as assessed by ELISA (C) and qPCR analysis (D). (E, F) Knockdown of IL-17R by siRNA rescued the reduced TGF-β1 expression by recombinant IL-17 treatment, as assessed by ELISA (E) and qPCR analysis (F). (G, H) IKK inhibitor VII, but not Stat3 (5, 15-DPP) and ERK (PD325901) inhibitors, rescued TGF-β1 expression in IL-17-treated MSCs as assessed by ELISA (G) and qPCR analysis (H). (I, J) Knockdown of IKKα by siRNA, which blocks IκBα degradation to inhibit NFκB, blocked IL-17-induced downregulation of TGF-β1 in MSCs, as assessed by ELISA (I) and qPCR (J). (K) ChIP-PCR assay of NF-κB binding on TGF-β1 promoter. Cell lysate from control MSCs and IL-17-treated MSCs was immunoprecipitated by NF-κB antibody, and relative enrichment of TGF-β1 promoter was quantified by qPCR with primers targeted at the indicated regions. IL-17 treatment could enhance NF-κB binding on TGF-β1 promoter at promoter region 3. (L) Luciferase assay showed that relative TGF-β1 promoter activity decreased in the presence of IL-17, which was rescued by IL-17R and IKKα siRNAs. (M, N) Recombinant IL-17 (20 ng/ml) treatment attenuated MSC-mediated upregulation of Tregs (M) and downregulation of Th17 cells (N), while the effect of recombinant IL-17 on MSCs was blocked by IKK inhibitor VII treatment. In: Inhibitor. *P < 0.05; **P < 0.01 and ***P < 0.001. (mean ± SD). The data are representative of three independent experiments.
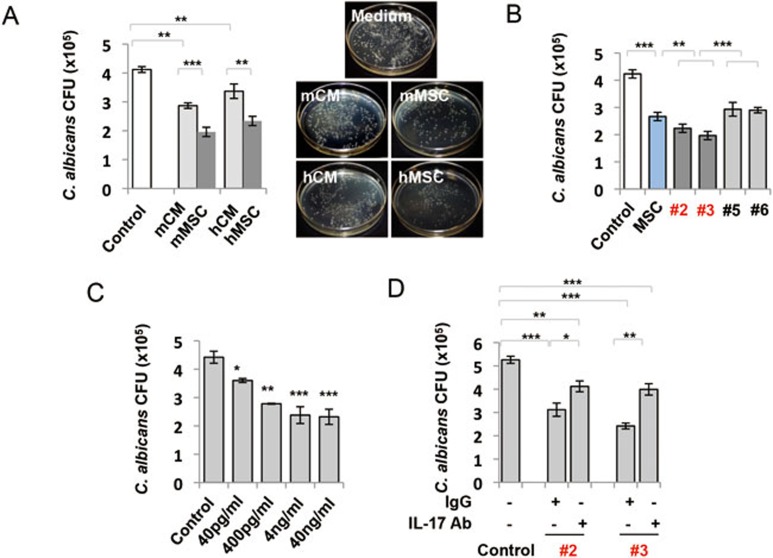
IL-17+ MSCs inhibited C. albicans growth
Previous studies indicated that IL-17 possessed anti-C. albicans effect32. To examine whether MSCs, especially IL-17+ MSCs, have anti-C. albicans effect, we co-cultured MSCs with C. albicans for 6 h and showed that MSCs and conditional medium (CM) significantly inhibited C. albicans growth compared to the control medium. It appeared that MSCs showed more significant inhibition of C. albicans growth than CM (Figure 6A). These data suggest that anti-C. albicans effect of MSCs is partially associated with soluble secreted factors. Next, we compared anti-C. albicans activity of IL-17+ and IL-17− MSCs and found that IL-17+ MSCs (#2 and #3) showed increased anti-C. albicans effect compared to IL-17− and bulk MSCs (#5 and #6; Figure 6B). To confirm that IL-17 directly inhibited C. albicans, we added recombinant IL-17 to cultured C. albicans and found that IL-17 inhibited C. albicans growth in a dose-dependent manner (Figure 6C). Anti-IL-17 neutralizing antibody could partially reduce MSC-mediated anti-C. albicans activity in comparison with the isotype control IgG group (Figure 6D).
Figure 6.
IL-17+ MSCs inhibited C. albicans growth. (A) MSCs and their conditioned medium (CM) inhibited C. albicans growth after 6 h co-incubation in vitro. MSCs showed significantly increased anti-C. albicans growth effect over their CM (n = 8). (B) Single colony-derived MSCs exhibited variable anti-C. albicans activity in vitro. Compared with bulk MSCs and IL-17− MSCs (#5 and #6), IL-17+ MSCs (#2 and #3) showed significantly increased anti-C. albicans activity (n = 8). (C) Recombinant IL-17 treatment had anti-C. albicans growth effect in a dose-dependent manner (n = 4). (D) Preincubation of IL-17+MSCs (#2, #3) with anti-IL-17 neutralizing antibody (IL-17 Ab, 5 μg/ml), but not with the control isotype IgG (5 μg/ml), reduced their anti-C. albicans activity (n = 8). *P < 0.05; **P < 0.01 and ***P < 0.001. (mean ± SD). (D) *compared to control group. The data are representative of three independent experiments.
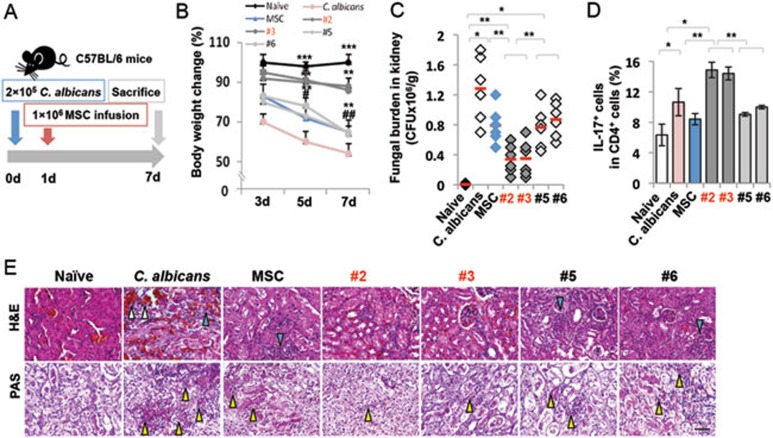
IL-17+ MSCs showed increased therapeutic effects in mice infected with C. albicans
To further determine the anti-C. albicans effect of IL-17+ MSCs, we infected C57BL/6 mice with 2 × 105 CFU of C. albicans (Figure 7A). One day after C. albicans injection, mice were treated with 1 × 106 single colony-derived MSCs. The extent of candidiasis was examined on day 7 post-MSC infusion by evaluating body weight alteration, kidney fungal burden, ratio of CD4+IL-17+ cells and histology. The body weight of mice infected with C. albicans was significantly reduced compared to the control C57BL/6 mice on days 3 to 7 post-infection. After MSC infusion, body weight was more significantly increased in IL-17+MSC treatment group than that of bulk and IL-17− MSC groups (Figure 7B). Although bulk and IL-17− MSC infusion groups showed some degree of reduction of total C. albicans counts in kidney, the IL-17+ MSC infusion group showed a more significant fungal clearance (Figure 7C). IL-17+ MSC infusion, but not bulk or IL-17− MSC infusion, elevated the level of Th17 cells, which may play an additional role in anti-C. albicans growth (Figure 7D). As shown by hematoxylin and eosin staining, IL-17+ MSC infusion significantly recovered histological structure of kidney compared to bulk and IL-17− MSCs infusions (Figure 7E). Infected mice showed the infiltration of C. albicans clusters in kidney, while mice treated with IL-17+ MSCs could more effectively clear C. albicans infiltration than groups treated with either bulk MSCs or IL-17− MSCs (Figure 7E). These findings suggest that IL-17+ MSCs possess a marked anti-fungal effect, but show compromised immunomodulatory property (Supplementary information, Figure S9).
Figure 7.
IL-17+ MSC infusion showed therapeutic effect in mice infected with C. albicans. (A) Schema of MSC infusion in C. albicans-infected mice. (B) Infected mice (n = 6) showed significantly reduced body weight from 3 to 7 days after C. albicans infection. IL-17+ MSC (#2, #3, n = 6)-infused group showed more effective inhibition of body weight loss compared to bulk MSC (n = 6) and IL-17− MSC (#5, #6, n = 6) groups. *versus C. albicans, #versus IL-17+MSC. (C) Fungal burden was measured on day 7 post-infection, as assessed by CFU of C. albicans in the kidneys. IL-17+MSCs (#2, #3) showed significantly higher fungal clearance compared to bulk and IL17−-MSCs (#5, #6). (D) The number of Th17 cells was elevated in infected mice compared to control C57BL/6 mice. IL-17+ MSC (#2, #3) infusion, but not bulk and IL-17− MSC (#5, #6) infusions, significantly elevated Th17 cells in infected mice. (E) Histological evaluation of C. albicans infection was performed on day 7 post-infection. Histological sections of kidney from the indicated mice were stained with H&E (upper panel) or PAS (lower panel). Kidney structure of C. albicans-infected mice was disturbed by infiltrating inflammatory cells (blue triangle) and red blood cells (white triangle). IL-17+ MSC infusion showed significant rescue of the kidney structures compared to bulk MSC and IL-17− MSC groups, as assessed by H&E staining. Infected mice showed the infiltration of C. albicans clusters (yellow triangle) in kidney, while the IL-17+ MSC infusion group showed a significantly more effective clearance of C. albicans infiltration compared to bulk and IL-17− MSC groups, as viewed by PAS staining. *P < 0.05; ** P < 0.01; and ***P < 0.001 (mean ± SD). Scale bars, 50 μm. The data are representative of three independent experiments.
Discussion
Previous studies indicate that MSC-based immunotherapy is a promising therapeutic approach for a variety of autoimmune diseases, including systemic sclerosis and systemic lupus erythematosus1,6,8,33. However, the detailed mechanism of MSC-based immune therapy is not fully elucidated34. Although many types of MSC surface molecules were identified and some subpopulations could be isolated according to the surface markers35, no specific functional MSC subpopulation has ever been successfully characterized, presenting a significant obstacle to the development of an effective MSC-based therapy. Thus, it is necessary to study the heterogeneity of MSCs to identify their different functional subsets. The aim of this study is to examine whether MSCs contain a novel subset of MSCs that produce functional IL-17. IL-17 is mainly produced by immune cells, such as Th17 and γδ T cells21, and here we identified a subset of IL-17+MSCs with the capacity to inhibit C. albicans growth. Distinct from bulk MSC population, IL-17+ MSCs are unable to upregulate Tregs or downregulate Th17 cells, a phenotype that can be rescued by knockdown of IL-17 expression, suggesting that IL-17 production in MSCs directly impairs MSC-based immunomodulatory function. Since MSC infusion can ameliorate DSS-induced colitis30,31,33, we infused IL-17+ MSCs to colitis mice to verify their in vivo immunoregulatory property and found that IL-17+ MSCs showed significantly reduced capacity to ameliorate disease phenotype in DSS-induced colitis mice compared to bulk or IL-17− MSCs. Knockdown of IL-17 expression in IL-17+ MSCs can, however, rescue their capacity to upregulate Tregs and downregulate Th17 cells. The downstream signaling of IL-17 involves several pathways, including NFκB, ERK, Stat3 and JNK21. We identified that IL-17 is capable of inhibiting TGF-β1 production in MSCs via the NFκB pathway. Since TGF-β1 plays a critical role in MSC-based immunomodulation by governing the levels of Tregs and Th17 cells36,37,38, reduced TGF-β1 expression in IL-17+MSCs results in abolishment of MSC-mediated immunosuppression. The decreased TGF-β1 production in IL-17+ MSCs can also be rescued by knockdown of IL-17 expression. Moreover, inhibition of the NFκB pathway by a chemical inhibitor or IKKα siRNA could recover TGF-β1 production, confirming that IL-17 regulates TGF-β1 production via the NFκB pathway. Additionally, we reveal that NFκB directly binds to the TGF-β1 promoter region to regulate TGF-β1 expression. These data imply that IL-17/NFκB/TGF-β1 signaling contributes to the impaired immunosuppressive function in IL-17+MSCs.
Finally, fungus infection is frequently associated with reduced host immune activity. However, it appears that MSC-mediated immunosuppression may not increase the risk of fungus infection in the recipients39. It is well known that IL-17 provides host defense against extracellular pathogens and possesses anti-fungal effect40. Interestingly, similar to the IL-17 produced by Th17 cells, we found that MSC-produced IL-17 possesses antifungal effect. IL-17+ MSC infusion further increased Th17 levels in C. albicans-infected mice. This may explain why MSC infusion does not result in fungal infection. In this study, we showed that sorted Sca1+IL-17+ MSCs expressed similar levels of IL-17 mRNA to CD4+IL-17+T cells, which was not affected by depletion of CD3+ T cells. In addition, we used western blot and ELISA to show that MSCs expressed IL-17, which did not change after CD3+ T cell depletion. At functional level, we showed that IL-17 produced by MSCs is capable of inhibiting fungus both in vitro and in vivo. Thus, a subset of IL-17+ MSCs fails to upregulate Tregs and downregulate Th17 cells, but can inhibit C. albicans growth, suggesting that a different subset of MSCs may play a unique functional role in MSC-based immune therapies.
Although the mechanism by which IL-17+ MSCs upregulate Th17 cells is unknown, the interplay between donor MSCs and recipient microenvironment may determine MSC-based immunomodulatory response33,41. Our findings imply that MSC-based immunomodulation is regulated by different subsets of MSCs, in which they may exert either immunosuppressive or immunostimulatory functions. Previous studies showed that MSCs possess anti-bacterial effects via secretion of anti-microbial peptide LL-3742. Our results indicate that MSCs are able to antagonize fungus infection, suggesting that MSCs may serve as a defense barrier against microbe invasion. Existence of IL-17+ MSCs may counterbalance the immunosuppressive role of MSCs under infection situation. Furthermore, understanding functional roles of different MSC subpopulations will facilitate to maximize the therapeutic effects for inflammatory and autoimmune diseases. As an alternative strategy, purified subsets of IL-17− MSCs may offer improved therapeutic effects for immune diseases.
In conclusion, we identify a novel subpopulation of IL-17+ MSCs that inhibit C. albicans growth, but fail to execute MSC-based immunosuppression as a result of reduced TGF-β1 expression. These findings suggest that MSCs contain different functional subpopulations with correspondingly unique therapeutic functions.
Materials and Methods
Mice
Female C57BL/6J mice were purchased from the Jackson Lab (Bar Harbor, ME, USA). All animal experiments were performed under the institutionally approved protocols for the use of animal research (USC #10941 and 11327, and Peking University # LA2012-65).
Antibodies
Anti-CD4-PerCP, CD25-APC, Sca1-PE, CD146-PE, CD3ɛ, PE-CD34, APC-CD3, PE-CD8a and CD28 antibodies were purchased from BD Biosciences (Franklin Lakes, NJ, USA). Anti-Foxp3-PE, IL-17-APC, and IL-17-PE antibodies were purchased from eBioscience (San Diego, CA, USA). Unconjugated antibodies to IL-17 and RORγt were purchased from Millipore Corporation (Bedford, MA, USA) and Abcam (Cambridge, MA, USA). Unconjugated antibodies to p-Stat3, Stat3, p-ERK, ERK, p-p38, p38, p-JNK, JNK, p-p65 and p65 were purchased from Cell Signaling Technology (Danvers, MA, USA). LEAF™ Purified human specific IL-17 and TGF-β1 antibodies were purchased from BioLegend (San Diego, CA, USA). Unconjugated antibody to ALP and goat isotype control antibody were purchased from Santa Cruz Biotechnology. Anti-rat IgG-rhodamine antibody was purchased from Southern Biotech (Birmingham, AL, USA). Hybridoma (38.1) for human CD3 mAb was purchased from ATCC (Manassas, VA, USA). Anti-rabbit IgG-AlexaFluor 488 antibody was purchased from Invitrogen Corporation (Carlsbad, CA, USA). Anti-β-actin antibody, inhibitors for Stat3 (5, 15-DPP), ERK (PD325901), JNK (SP600125), IKK inhibitor VII and p38 inhibitor (SB203580) were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Isolation of mouse bone marrow MSCs
Bone marrow cells were flashed out from bone cavity of femurs and tibias with 2% heat-inactivated fetal bovine serum (FBS; Equitech-Bio, Kerrville, TX, USA) and cultured with α minimum essential medium (α-MEM, Invitrogen) supplemented with 20% FBS, 2 mM L-glutamine (Invitrogen), 55 μM 2-mercaptoethanol (Invitrogen), 100 U/ml penicillin, and 100 μg/ml streptomycin (Invitrogen), as previously reported43.
Human bone marrow MSCs culture
Human bone marrow aspirates from healthy adult donors (20-35 years of age) were purchased from AllCells (Emeryville, CA, USA). Human bone marrow progenitor cells were enriched using fresh human bone marrow by RosetteSep® Human Bone Marrow Progenitor Cell Pre-Enrichment Cocktail (STEMCELL Technologies, Vancouver, BC, Canada) following the manufacturer's instructions. The enriched single-cell suspensions were cultured with α-MEM supplemented with 15% FBS, 100 mM L-ascorbic acid-2-phosphate, 2 mM L-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin and passaged, as previously reported44.
Isolation of single colony derived-human MSCs from single cell
For isolation of single colony-derived MSCs, the enriched single-cell suspensions were plated at about 50-100 cells per 10 cm dish under 37 °C at 5% CO2 condition for 1-2 weeks and examined with an inverted microscope twice a day. The colonies were picked up using a Pyrex® cloning cylinder (Sigma-Aldrich) when they reached optimal size (50-100 cells in each colony). Single colony was derived from one cell.
Cell proliferation assay
Single colony-derived MSCs were passaged to 4-well chamber slides (Nunc, 1 × 104 per well) and incubated for 12 h under 37 °C at 5% CO2 condition. Then the cells were assayed for bromodeoxyuridine (BrdU) incorporation, as described previously45.
Osteogenic differentiation assay
Single colony-derived MSCs were cultured under osteogenic culture condition medium containing 1.8 mM KH2PO4 and 10 nM dexamethasone (Sigma-Aldrich). After 28 days of osteogenic induction, the cultures were stained with Alizarin Red. Expression of the osteogenic marker ALP was assayed by western blot analysis.
Adipogenic differentiation assay
Single colony-derived MSCs were cultured under adipogenic culture condition medium containing 0.5 mM Isobutylmethylxanthin (Sigma-Aldrich), 60 μM Indomethacin (Sigma-Aldrich), 0.5 μM Hydrocortisone (Sigma-Aldrich), and 10 μg/ml insulin (Sigma-Aldrich). After 14 days of adipogenic induction, the cultures were stained with Oil Red O. Expression of the adipogenic lineage-specific genes LPL and PPARγ was assayed by semi-quantitative PCR.
Immunofluorescent staining
MSCs were seeded on a 4-well chamber slide and incubated for 12 h under 37 °C at 5% CO2 condition. Then the slides were fixed in 4% paraformaldehyde, followed by 0.01% Triton-100 treatment for 10 min. The slides were blocked with normal serum matched to secondary antibodies for 1 h, followed by incubation with the specific or isotype-matched antibodies (1:100) overnight at 4 °C. Then the slides were treated with Rhodamin/FITC-conjugated secondary antibodies (1:200, Jackson ImmunoResearch, West Grove, PA, USA) for 1 h under 20 °C -25 °C in dark and mounted by means of a VECTASHIELD® Mounting Medium containing 4'6-diamidino-2-phenylindole (DAPI) (Vector Laboratories, Burlingame, CA, USA).
T lymphocyte isolation
Human PBMCs were purchased from AllCells. Human naïve T lymphocytes were purified by negative selection from PBMCs using a Pan T cell isolation kit (Miltenyi Biotec, Auburn, CA, USA), according to the manufacturer's instructions.
Tregs and Th17 cells co-cultured with MSCs
Human naïve T lymphocytes (1 × 106 per well) were precultured on 24-well multiplates under Dulbecco's Modified Eagle's Medium (DMEM, Lonza) with 10% heat-inactivated FBS, 50 μM 2-mercaptoethanol, 10 mM HEPES (Sigma-Aldrich), 1 mM sodium pyruvate (Sigma-Aldrich), 1% nonessential amino acid (Lonza Inc, Allendale, NJ, USA), 2 mM L-glutamine, 100 U/ml penicillin and 100 mg/ml streptomycin in the presence of plate-bound anti-CD3ɛ antibody (2 μg/ml) and soluble anti-CD28 antibody (2 μg/ml) for 2-3 days. For Treg differentiation, recombinant human TGF-β1 (2 ng/ml) (R&D Systems, Minneapolis, MN, USA) and IL-2 (2 ng/ml) (R&D Systems) were added. For Th17 induction, recombinant human TGF-β1 (2 ng/ml) and IL-6 (50 ng/ml) (R&D Systems) were added. IL-17+, IL-17− or IL-17-treated hMSCs and bulk MSCs were seeded on 24-well multiplates and incubated for 12 h. Then the activated T lymphocytes (0.5 × 106 per well) were loaded on the MSCs and co-cultured for three days. After 3 days, floating cells and culture supernatant were harvested. The cells were used to examine Treg and Th17 cell numbers by flow cytometry, and the supernatant was collected to analyze IL-17 and IL-10 levels by enzyme-linked immunosorbent assay (ELISA), following the manufacturer's instructions.
CD3+ T cell depletion by antibody/complement treatment
To deplete CD3+ T cells, 1 ml predetermined proper concentration of CD3 mAb (Hybridoma: 38.1, ATCC) was added to hPBMCs and hMSCs (1 × 107) and incubated for 30 min at 4 °C. The cells were centrifuged for 5 min to remove unbound antibody and discard supernatant. Then the pellets were resuspended in 1 ml (1:6) diluted rabbit complement (Pel-Freez Biologicals, Rogers, AR) and incubated for 30 min at 37 °C. The cells were centrifuged for 5 min to remove unbound complement and discard supernatant. One round of treatment with cytotoxic antibody/complement was repeated. Isotype IgM was used as a negative control. The efficacy of T cell depletion was determined by flow cytometric analysis. hMSCs were cultured for 3 days for IL-17 expression analysis46.
Flow cytometry analysis
The following antibodies were used for surface staining: anti-mouse Sca1-PE, mouse CD4-Percp, mouse CD25-APC, human CD4-Percp, human CD3-APC, human CD8a-PE, human CD34-PE and human CD25-APC. For intracellular cytokine or transcription factor expression, staining was performed using Foxp3 staining buffer according to the manufacturer's instructions (eBioscience). Anti-mouse Foxp3-PE, mouse IL-17-APC or -PE, human Foxp3-PE, and human IL-17-APC or -PE were used for intracellular staining. Data were acquired using FACScalibur (BD Bioscience).
Western blot
Cells were lysed in M-PER® mammalian protein extraction reagent (Pierce). 20 μg of total proteins were applied and separated on 4%-12% NuPAGE® gel (Invitrogen) and transferred to Immobilon™-P membranes (Millipore Corporation). The membranes were blocked with 5% non-fat dry milk and 0.1% Tween 20 for 1 h, followed by incubation with the primary antibodies (1:100-1 000 dilution) at 4 oC overnight. Then they were treated with horseradish peroxidase-conjugated rabbit or mouse IgG (Santa Cruz) (1:10 000) for 1 h, enhanced with a SuperSignal® West Pico Chemiluminescent Substrate (Pierce, Rockford, IL, USA), and exposed to BIOMAX MR films (Kodak, Rochester, NY, USA).
Semi-quantitative PCR and real-time PCR
For semi-quantitative PCR and real-time PCR (qPCR) analyses, total RNA was extracted with RNeasy Mini kit (Qiagen, Valencia, CA, USA), and cDNA was prepared using a SuperScript® III Reverse Transcriptase (RT) kit (Invitrogen). Semi-quantitative PCR was performed using gene-specific primers (Supplementary information, Table S1) and Platinum® PCR SuperMix (Invitrogen). The amplified PCR products were separated by 2% agarose gels with ethidium bromide and visualized by UV light. qPCR was carried out using SYBR® Green Supermix (Bio-Rad) on a Bio-Rad CFX96 Real Time system, as indicated by the manufacturer's protocol. The level of mRNA expression for each gene was normalized to GAPDH mRNA expression.
ELISA
Peripheral blood was collected from the retro-orbital plexus of mice and centrifuged to obtain the blood serum. Culture supernatants from MSCs were collected. MSCs were extracted using M-PER® mammalian protein extraction reagent. The samples were centrifuged and used for IL-17 (BioLegend), IL-10, IL-6, TNF-α, IFN-γ, PGE2 and TGF-β1 (R&D Systems) ELISA analysis.
Chromatin immunoprecipitation assay
Cells were fixed, nuclei were lysed, and chromatin was sheared with a Branson sonicator. Immunoprecipitation was performed with the Millipore ChIP kit according to the manufacturer's protocol. To precipitate NFκB complexes, NFκB antibody was used and goat IgG was used as an isotype control. Percentage input was determined by removing an aliquot of sheared chromatin prior to immunoprecipitation and comparing amplification of this DNA with amplification of the precipitated chromatin. Putative NFκB-binding sites in TGF-β1 promoter regions were investigated to assay the relative enrichment by qPCR. Primers are listed in Supplementary information, Table S1.
Luciferase assay
TGF-β1 promoter region was PCR amplified from MSC genomic DNA and inserted into pGL3 vector by HindIII and XhoII to construct the reporter plasmid. MSCs on a 24-well plate were transiently transfected with TGF-β1-luciferase plasmid and renilla luciferase plasmid at the ratio of 10:1. After 24 h of transfection, cells were collected, and luciferase activity was measured with a dual-luciferase reporter assay kit (Promega). All data were normalized to renilla luciferase activity.
MSC infusion into acute colitis mice
Acute colitis was induced by administering 3% (w/v) dextran sulfate sodium (DSS, molecular mass 36 000-50 000 Da; MP Bio-chemicals, France) through drinking water, which was fed ad libitum for 10 days. Bulk MSCs and single colony-derived MSCs were infused (1 × 106 cells) into colitis mice (n = 6) intravenously on day 3 after feeding DSS water. In the control group, mice received PBS (n = 6). Samples from all mice were harvested on day 10 after feeding DSS water. Induced colitis was evaluated as previously described30,31.
C. albicans culture and anti-fungal activity assays
C. albicans (ATCC®MYA-2876™) was grown overnight at 37 °C in liquid Luria-Bertani (LB) medium (Difco BD, Sparks, MD, USA) with slight agitation and harvested by centrifugation. The pellet was resuspended and centrifuged. The density of inoculum was adjusted to 1.0 at A600 nm, corresponding to 5 × 106- 1 × 107 CFU/ml, as determined by serial dilution and colony counts. Inhibition of C. albicans growth by MSCs and CM was assessed by counting CFU. Briefly, MSCs plated in 24-well plates (3 × 104 hMSCs and 2 × 105 mMSCs per well) were infected with 500 CFU of C. albicans and incubated for 6 h under 37 °C at 5% CO2 condition. Then the plates were vigorously shaken, and aliquots of culture medium were taken from each well and serially diluted with sterile PBS and plated on LB-agar plates at 37 °C. Colonies were counted the next day. Anti-C. albicans activity of MSC CM (or hIL-17A) was subjected to a susceptibility test, according to a previous report42, with slight modifications. Briefly, CM was obtained from three-day cultured hMSCs and mMSCs by passing through a 0.22 μm filter (Millipore). The aliquots (380 μl) were transferred to a 24-well plate, inoculated with 500 CFU of C. albicans (in 20 μl of PBS) and incubated for 6 h at 37 °C. Then CFUs were analyzed as described earlier. To further evaluate anti-C. albicans activity of IL-17, MSCs were preincubated with IL-17 neutralizing antibody or isotype control antibody for 2 h, then added to 500 CFU of C. albicans and incubated for 6 h for CFU analysis.
MSC infusion into mice infected with C. albicans
C57BL/6 mice were infected intravenously with 2 × 105 C. albicans and then monitored daily. Bulk MSCs and single colony-derived MSCs (1 × 106 cells) were intravenously infused into infected mice (n = 6) 1 day after induction of C. albicans. In the control group, mice received PBS (n = 6). Samples from all mice were harvested on day 7 after infection. For fungal burden analysis, kidneys were removed, weighed and dissected to cell suspension, then serially diluted with sterile PBS and plated on LB-agar plates at 37 °C for CFU counting. For histological analysis, kidney samples were stained with PAS or hematoxylin and eosin.
Statistical analysis
P-values were analyzed from two-tailed Student's t-test or one-way analysis of variance using SPSS 13.0 software. P-values less than 0.05 were considered statistically significant.
Acknowledgments
This work was supported by grants from the National Institute of Dental and Craniofacial Research (NIDCR), National Institutes of Health (NIH), Department of Health and Human Services (R01DE017449, R01 DE019932, and R01 DE019413 to S S), the Ministry of Science and Technology of China (2010DFB32980 to Y Z), and the Intramural Research Program of NIDCR, NIH (to W J C).
Footnotes
(Supplementary information is linked to the online version of the paper on the Cell Research website.)
Supplementary Information
Flow cytometric analysis showed that hMSCs and mMSCs expressed stem cell markers CD146 and Sca1, but not hematopoietic markers CD34, CD3, CD4 and CD8a.
Immunofluorescent and immunocytochemical staining showed that hMSCs were positive for stem cell markers CD146 and Sca1, but negative for T cell markers CD3, CD4 and CD8a.
Flow cytometric analysis showed the efficacy of CD3 antibody/complement–mediated depletion of CD3+ T cells in hPBMCs.
IL-17 expression was regulated by IL-1β and IL-6 via activating Stat3 and RORγt.
Flow cytometric analysis showed that IL-17+ MSCs (#2, #3, and #4) expressed stem cell markers CD146 and Sca1, but not hematopoietic markers CD34, CD3, CD4 and CD8a.
Single colony-derived hMSCs possess stem cell properties.
Western blot analysis showed efficacy of IL-17, IL-17R, and IκB kinase α siRNA in hMSCs.
(A) Western blot analysis showed that recombinant IL-17 treatment increased p-IκBα and p-p65 in MSCs.
Schematic diagram shows the mechanism by which IL-17+ MSCs attenuate immunosuppressive capacities, but possess increased anti-C. albicans activity.
PCR primers
References
- Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;105:1815–1822. doi: 10.1182/blood-2004-04-1559. [DOI] [PubMed] [Google Scholar]
- Nauta AJ, Fibbe WE. Immunomodulatory properties of mesenchymal stromal cells. Blood. 2007;110:3499–3506. doi: 10.1182/blood-2007-02-069716. [DOI] [PubMed] [Google Scholar]
- Uccelli A, Moretta L, Pistoia V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8:726–736. doi: 10.1038/nri2395. [DOI] [PubMed] [Google Scholar]
- Batten P, Sarathchandra P, Antoniw JW, et al. Human mesenchymal stem cells induce T cell anergy and downregulate T cell allo-responses via the TH2 pathway: relevance to tissue engineering human heart valves. Tissue Eng. 2006;12:2263–2273. doi: 10.1089/ten.2006.12.2263. [DOI] [PubMed] [Google Scholar]
- Meisel R, Zibert A, Laryea M, Gobel U, Daubener W, Dilloo D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood. 2004;103:4619–4621. doi: 10.1182/blood-2003-11-3909. [DOI] [PubMed] [Google Scholar]
- Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-kappaB signaling in resident macrophages. Blood. 2011;118:330–338. doi: 10.1182/blood-2010-12-327353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y, Hu G, Su J, et al. Mesenchymal stem cells: a new strategy for immunosuppression and tissue repair. Cell Res. 2010;20:510–518. doi: 10.1038/cr.2010.44. [DOI] [PubMed] [Google Scholar]
- Sun L, Akiyama K, Zhang H, et al. Mesenchymal stem cell transplantation reverses multiorgan dysfunction in systemic lupus erythematosus mice and humans. Stem Cells. 2009;27:1421–1432. doi: 10.1002/stem.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Rey E, Anderson P, Gonzalez MA, Rico L, Buscher D, Delgado M. Human adult stem cells derived from adipose tissue protect against experimental colitis and sepsis. Gut. 2009;58:929–939. doi: 10.1136/gut.2008.168534. [DOI] [PubMed] [Google Scholar]
- Park MJ, Park HS, Cho ML, et al. Transforming growth factor beta-transduced mesenchymal stem cells ameliorate experimental autoimmune arthritis through reciprocal regulation of Treg/Th17 cells and osteoclastogenesis. Arthritis Rheum. 2011;63:1668–1680. doi: 10.1002/art.30326. [DOI] [PubMed] [Google Scholar]
- Liotta F, Angeli R, Cosmi L, et al. Toll-like receptors 3 and 4 are expressed by human bone marrow-derived mesenchymal stem cells and can inhibit their T-cell modulatory activity by impairing Notch signaling. Stem Cells. 2008;26:279–289. doi: 10.1634/stemcells.2007-0454. [DOI] [PubMed] [Google Scholar]
- Waterman RS, Tomchuck SL, Henkle SL, Betancourt AM. A new mesenchymal stem cell (MSC) paradigm: polarization into a pro-inflammatory MSC1 or an Immunosuppressive MSC2 phenotype. PLoS One. 2010;5:e10088. doi: 10.1371/journal.pone.0010088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Opitz CA, Litzenburger UM, Lutz C, et al. Toll-like receptor engagement enhances the immunosuppressive properties of human bone marrow-derived mesenchymal stem cells by inducing indoleamine-2,3-dioxygenase-1 via interferon-beta and protein kinase R. Stem Cells. 2009;27:909–919. doi: 10.1002/stem.7. [DOI] [PubMed] [Google Scholar]
- Li W, Ren G, Huang Y, et al. Mesenchymal stem cells: a double-edged sword in regulating immune responses. Cell Death Differ. 2012;19:1505–1513. doi: 10.1038/cdd.2012.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolls JK, Linden A. Interleukin-17 family members and inflammation. Immunity. 2004;21:467–476. doi: 10.1016/j.immuni.2004.08.018. [DOI] [PubMed] [Google Scholar]
- Matsuzaki G, Umemura M. Interleukin-17 as an effector molecule of innate and acquired immunity against infections. Microbiol Immunol. 2007;51:1139–1147. doi: 10.1111/j.1348-0421.2007.tb04008.x. [DOI] [PubMed] [Google Scholar]
- Zelante T, De Luca A, Bonifazi P, et al. IL-23 and the Th17 pathway promote inflammation and impair antifungal immune resistance. Eur J Immunol. 2007;37:2695–2706. doi: 10.1002/eji.200737409. [DOI] [PubMed] [Google Scholar]
- Michel ML, Keller AC, Paget C, et al. Identification of an IL-17-producing NK1.1(neg) iNKT cell population involved in airway neutrophilia. J Exp Med. 2007;204:995–1001. doi: 10.1084/jem.20061551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vykhovanets EV, Maclennan GT, Vykhovanets OV, Gupta S. IL-17 Expression by macrophages is associated with proliferative inflammatory atrophy lesions in prostate cancer patients. Int J Clin Exp Pathol. 2011;4:552–565. [PMC free article] [PubMed] [Google Scholar]
- Takahashi N, Vanlaere I, de Rycke R, et al. IL-17 produced by Paneth cells drives TNF-induced shock. J Exp Med. 2008;205:1755–1761. doi: 10.1084/jem.20080588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zepp J, Wu L, Li X. IL-17 receptor signaling and T helper 17-mediated autoimmune demyelinating disease. Trends Immunol. 2011;32:232–239. doi: 10.1016/j.it.2011.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beriou G, Costantino CM, Ashley CW, et al. IL-17-producing human peripheral regulatory T cells retain suppressive function. Blood. 2009;113:4240–4249. doi: 10.1182/blood-2008-10-183251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eljaafari A, Tartelin ML, Aissaoui H, et al. Bone marrow-derived and synovium-derived mesenchymal cells promote Th17 cell expansion and activation through caspase 1 activation: contribution to the chronicity of rheumatoid arthritis. Arthritis Rheum. 2012;64:2147–2157. doi: 10.1002/art.34391. [DOI] [PubMed] [Google Scholar]
- Mangan PR, Harrington LE, O'Quinn DB, et al. Transforming growth factor-beta induces development of the T(H)17 lineage. Nature. 2006;441:231–234. doi: 10.1038/nature04754. [DOI] [PubMed] [Google Scholar]
- Bettelli E, Carrier Y, Gao W, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- Harris TJ, Grosso JF, Yen HR, et al. Cutting edge: An in vivo requirement for STAT3 signaling in TH17 development and TH17-dependent autoimmunity. J Immunol. 2007;179:4313–4317. doi: 10.4049/jimmunol.179.7.4313. [DOI] [PubMed] [Google Scholar]
- Yang XO, Panopoulos AD, Nurieva R, et al. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J Biol Chem. 2007;282:9358–9363. doi: 10.1074/jbc.C600321200. [DOI] [PubMed] [Google Scholar]
- Morikawa S, Mabuchi Y, Kubota Y, et al. Prospective identification, isolation, and systemic transplantation of multipotent mesenchymal stem cells in murine bone marrow. J Exp Med. 2009;206:2483–2496. doi: 10.1084/jem.20091046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwakura Y, Ishigame H, Saijo S, Nakae S. Functional specialization of interleukin-17 family members. Immunity. 2011;34:149–162. doi: 10.1016/j.immuni.2011.02.012. [DOI] [PubMed] [Google Scholar]
- Alex P, Zachos NC, Nguyen T, et al. Distinct cytokine patterns identified from multiplex profiles of murine DSS and TNBS-induced colitis. Inflamm Bowel Dis. 2009;15:341–352. doi: 10.1002/ibd.20753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q, Shi S, Liu Y, Uyanne J, Shi Y, Le AD. Mesenchymal stem cells derived from human gingiva are capable of immunomodulatory functions and ameliorate inflammation-related tissue destruction in experimental colitis. J Immunol. 2009;183:7787–7798. doi: 10.4049/jimmunol.0902318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang W, Na L, Fidel PL, Schwarzenberger P. Requirement of interleukin-17A for systemic anti-Candida albicans host defense in mice. J Infect Dis. 2004;190:624–631. doi: 10.1086/422329. [DOI] [PubMed] [Google Scholar]
- Akiyama K, Chen C, Wang D, et al. Mesenchymal stem cell induced immunoregulation involves FAS-ligand/FAS-mediated T cell apoptosis. Cell Stem Cell. 2012;10:544–555. doi: 10.1016/j.stem.2012.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sudres M, Norol F, Trenado A, et al. Bone marrow mesenchymal stem cells suppress lymphocyte proliferation in vitro but fail to prevent graft-versus-host disease in mice. J Immunol. 2006;176:7761–7767. doi: 10.4049/jimmunol.176.12.7761. [DOI] [PubMed] [Google Scholar]
- Lindner U, Kramer J, Rohwedel J, Schlenke P. Mesenchymal stem or stromal cells: toward a better understanding of their biology. Transfus Med Hemother. 2010;37:75–83. doi: 10.1159/000290897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L, Lopes JE, Chong MM, et al. TGF-beta-induced Foxp3 inhibits T(H)17 cell differentiation by antagonizing RORgammat function. Nature. 2008;453:236–240. doi: 10.1038/nature06878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svobodova E, Krulova M, Zajicova A, et al. The role of mouse mesenchymal stem cells in differentiation of naive T-cells into anti-inflammatory regulatory T-cell or proinflammatory helper T-cell 17 population. Stem Cells Dev. 2012;21:901–910. doi: 10.1089/scd.2011.0157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu G, Zhang L, Ren G, et al. Immunosuppressive properties of cloned bone marrow mesenchymal stem cells. Cell Res. 2007;17:240–248. doi: 10.1038/cr.2007.4. [DOI] [PubMed] [Google Scholar]
- Le Blanc K, Frassoni F, Ball L, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet. 2008;371:1579–1586. doi: 10.1016/S0140-6736(08)60690-X. [DOI] [PubMed] [Google Scholar]
- Caccamo N, La Mendola C, Orlando V, et al. Differentiation, phenotype, and function of interleukin-17-producing human Vγ9Vδ2 T cells. Blood. 2011;118:129–138. doi: 10.1182/blood-2011-01-331298. [DOI] [PubMed] [Google Scholar]
- Auletta JJ, Deans RJ, Bartholomew AM. Emerging roles for multipotent, bone marrow-derived stromal cells in host defense. Blood. 2012;119:1801–1809. doi: 10.1182/blood-2011-10-384354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krasnodembskaya A, Song Y, Fang X, et al. Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37. Stem Cells. 2010;28:2229–2238. doi: 10.1002/stem.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Wang L, Kikuiri T, et al. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via IFN-gamma and TNF-alpha. Nat Med. 2011;17:1594–1601. doi: 10.1038/nm.2542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi S, Gronthos S, Chen S, et al. Bone formation by human postnatal bone marrow stromal stem cells is enhanced by telomerase expression. Nat Biotechnol. 2002;20:587–591. doi: 10.1038/nbt0602-587. [DOI] [PubMed] [Google Scholar]
- Yamaza T, Kentaro A, Chen C, et al. Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell Res Ther. 2010;1:5. doi: 10.1186/scrt5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hathcock KS.T cell depletion by cytotoxic elimination Curr Protoc Immunol 2001Chapter 3:Unit 3.4. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Flow cytometric analysis showed that hMSCs and mMSCs expressed stem cell markers CD146 and Sca1, but not hematopoietic markers CD34, CD3, CD4 and CD8a.
Immunofluorescent and immunocytochemical staining showed that hMSCs were positive for stem cell markers CD146 and Sca1, but negative for T cell markers CD3, CD4 and CD8a.
Flow cytometric analysis showed the efficacy of CD3 antibody/complement–mediated depletion of CD3+ T cells in hPBMCs.
IL-17 expression was regulated by IL-1β and IL-6 via activating Stat3 and RORγt.
Flow cytometric analysis showed that IL-17+ MSCs (#2, #3, and #4) expressed stem cell markers CD146 and Sca1, but not hematopoietic markers CD34, CD3, CD4 and CD8a.
Single colony-derived hMSCs possess stem cell properties.
Western blot analysis showed efficacy of IL-17, IL-17R, and IκB kinase α siRNA in hMSCs.
(A) Western blot analysis showed that recombinant IL-17 treatment increased p-IκBα and p-p65 in MSCs.
Schematic diagram shows the mechanism by which IL-17+ MSCs attenuate immunosuppressive capacities, but possess increased anti-C. albicans activity.
PCR primers